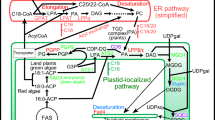
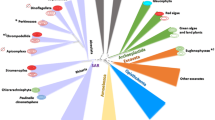
Abstract
The theory of endosymbiotic origin of chloroplasts has become basal in present-day biology. In this regard, the emergence of eukaryotic photosynthesis has been established as a result of phagocytal capture of cyanobacteria by some ancestral eukaryotic cell. The phenomenon of endosymbiosis is an indispensable element of cell evolution theory. The process of endosymbiogenesis belongs to the crucial factors of biological progress that gave rise to astonishing diversity and complexity of the eukaryotic organisms. The modern theory of evolution is built on the joint base of the phenomena of endosymbiosis, parallel gene transfer, the theory of natural selection, and the data of phylogenomics. Ultimately, eukaryogenesis has integrated different supramolecular complexes in the cells, created the diversity of organelles, and has led to biosphere oxygenation and modern diversity of living forms.




Similar content being viewed by others
REFERENCES
Mereschkowsky, C., Über Natur und Ursprung der Chromatophoren im Pflanzenreiche, Biol. Centralbl., 1905, vol. 25, p. 593.
Merezhkovskii, K.S., Teoriya dvukh plazm, kak osnova simbiogeneza, novogo ucheniya o proizkhozhdenii organizmov (Theory of Two Plasmas as the Basis of Symbiogenesis—A New Concept on Origin of Organisms), Kazan: Tipogr. Imper. Univ., 1909.
Khakhina, L.N., Problema simbiogeneza (A Problem of Symbiogenesis), Leningrad: Nauka, 1979.
Kolchinskii, E.I., Edinstvo evolyutsionnoi teorii v razdelennom mire XX veka (The Integration of Evolutionary Theory in the Divided World of the 20th Century), St. Petersburg: Nestor-Istoriya, 2015.
Gibbs S.P., The evolution of algal chloroplasts, in Origins of Plastids: Symbiogenesis, Prochlorophytes and the Origins of Chloroplasts, Lewin, R.A., Ed., New York: Springer-Verlag, 1993, p. 107.
Archibald, J.M., Endosymbiosis and eukaryotic cell evolution, Curr. Biol., 2015, vol. 25, p. 911.
Martin, W. and Kowallik, K.W., Annotated english translation of Mereschkowsky’s 1905 paper “Über Natur und Ursprung der Chromatophoren im Pflanzenreiche”, Eur. J. Phycol., 1999, vol. 34, p. 287.
Strel’nikova, N.I., Merezhkovskii K.S. and his research works on diatoms (to the 150th anniversary). http:// www.ksu.ru/conf/botan200/s183.rtf.
Zakharov, I.A., The 100 anniversary of the theory of symbiogenesis, Vestn. Vavilovsk. O-va Genet. Sel., 2009, vol. 13, p. 355.
Provorov, N.A., Merezhkovskii K.S. and origin of eukaryotic cell: 111 years of the theory of symbiogenesis, S-kh. Biol., 2016, vol. 51, p. 746.
Fedonkin, M.A., Geochemical impoverishment and eukaryotization of the biosphere: a causal link, Paleontol. J., 2003, vol. 37, no. 6, p. 592.
Rozanov, A.Yu., Bacterial paleontology, sedimentogenesis, and early evolution of biosphere, Tr. Geol. Inst., Ross. Akad. Nauk, 2004, no. 565, p. 448.
Woese, C.R., Kandler, O., and Wheelis, M.L., Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya, Proc. Natl. Acad. Sci. U.S.A., 1990, vol. 87, p. 4576.
MacLeod, F., Gareth, S., Kindler, G.S., Wong, H.L., Chen, R., and Burns, B.P., Asgard archaea: diversity, function, and evolutionary implications in a range of microbiomes, AIMS Microbiol., 2019, vol. 5, p. 48. https://doi.org/10.3934/microbiol.2019.1.48
Yutin, N., Wolf, M.Y., Wolf, Y.I., and Koonin, E.V., The origins of phagocytosis and eukaryogenesis, Biol. Direct., 2009, vol. 4, p. 9. https://doi.org/10.1186/1745-6150-4-9
Shestakov, S.V., The role of archaea in the origin of eukaryotes, Ekol. Genet., 2017, vol. 15, p. 52. https://doi.org/10.17816/ecogen15452-59
Martijn, J., Vosseberg, J., Guy, L., Offre, P., and Ettema, T.J.G., Deep mitochondrial origin outside the sampled alphaproteobacteria, Nature., 2018, vol. 557, p. 101.
López-García, P. and Móreira, D., The syntrophy hypothesis for the origin of eukaryotes revisited, Nat. Microbiol., 2020, vol. 5, p. 655.
Gorlenko, V.M., Anoxygenic phototrophic bacteria, Tr. Inst. Mikrobiol. im. S.N. Vinogradskogo, 2010, no. 15, p. 133.
Wachterschauser, G., From volcanic origins of chemoautotrophic life to Bacteria, Archaea and Eukarya, Philos. Trans. R. Soc. B., 2006, vol. 361, p. 1787.
Schopf, J.W., Kudrjavtsev, A.B., Agresti, D.G., Wdowiak, T., and Czaja, F.D., Laser-Raman imagery of Earth’s earliest fossils, Nature, 2003, vol. 416, p. 73.
Schirrmeister, B.E., Gugger, M., and Donoghue, P.C.J., Cyanobacteria and the great oxidation event: evidence from genes and fossils, Palaeontology, 2015, vol. 58, p. 769.
Nelson, N. and Junge, W., Structure and energy transfer in photosystems of oxygenic photosynthesis, Annu. Rev. Biochem., 2015, vol. 84, p. 659.
Stadnichuk, I.N., Krasilnikov, P.M., and Zlenko, D.V., Cyanobacterial phycobilisomes and phycobiliproteins, Microbiology (Moscow), 2015, vol. 84, p. 101.
Stadnichuk, I.N. and Tropin, I.V., Antenna replacement in the evolutionary origin of chloroplasts, Microbiology (Moscow), 2014, vol. 83, p. 299.
Keely, B.G., Geochemistry of chlorophylls, in Chlorophylls and Bacteriochlorophylls: Biochemistry, Biophysics, Functions and Applications, Grimm, B., Porra, R.J., Rüdiger, W., and Scheer, H., Eds., Dordrecht: Springer-Verlag, 2006, p. 535.
Granick, S., Speculations on the origins and evolution of photosynthesis, Ann. N.Y. Acad. Sci., 1957, vol. 69, p. 292.
Beale, S.I., Enzymes of chlorophyll biosynthesis, Photosynth. Res., 1999, vol. 60, p. 43.
Oster, U., Tanaka, R., Tanaka, A., and Rüdiger, W., Cloning and functional expression of the gene encoding the key enzyme for chlorophyll b biosynthesis (CAO) from Arabidopsis thaliana, Plant J., 2000, vol. 21, p. 305.
South, G.R. and Whittick, A., An Introduction to Phycology, Chichester: Wiley, 1987.
Kusakin, O.G. and Drozdov, A.L., Filema organicheskogo mira. Chast’ 2. Prokaryota, Eukaryota (Fileme of the Organic World, Part 2: Prokaryota, Eukaryota), St. Petersburg: Nauka, 1987.
Hausmann, K., Hülsmann, N., and Radek, R., Protistology, Stuttgart: Schweizerbart, 2003.
Cavalier-Smith, T., Kingdom Chromista and its eight phyla: a new synthesis emphasising periplastid protein targeting, cytoskeletal and periplastid evolution, and ancient divergences, Protoplasma, 2017, vol. 255, p. 297. https://doi.org/10.1007/s00709-017-1147-3
Burki, F., Okamoto, N., Pombert, J.F., and Keeling, P.J., The evolutionary history of haptophytes and cryptophytes: phylogenomic evidence for separate origins, Proc. R. Soc. B, 2012, vol. 279, p. 2246. https://doi.org/10.1098/rspb.2011.2301
Keeling, P.J., The endosymbiotic origin diversification and fate of plastids, Philos. Trans. R. Soc. B, 2010, vol. 365, p. 792.
Sánchez-Baracaldo, P., Raven, J.A., Pisani, D., and Knoll, A.H., Early photosynthetic eukaryotes inhabited low-salinity habitats, Proc. Natl. Acad. Sci. U.S.A., 2017, vol. 114, p. e7737.
Donoghue, P. and Paps, J., Plant evolution: assembling land plants, Curr. Biol., 2020, vol. 30, p. R81. https://doi.org/10.1016/j.cub.2019.11.084
Lhee, D., Ha, J.-S. Kim, S., Park, M.G., Bhattacharya, D., and Yoon, H.S., Evolutionary dynamics of the chromatophore genome in three photosynthetic Paulinella species, Sci. Rep., 2019, vol. 9. 1https://doi.org/10.1038/s41598-019-38621-8
Gibbs, S.P., The chloroplast of some algal groups may evolved from endosymbiotic eukaryotic algae, Ann. N.Y. Acad. Sci., 1981, vol. 361, p. 193.
Archibald, J.M., The puzzle of plastid evolution, Curr. Biol., 2009, vol. 19, p. R81.
Ludwig, M. and Gibbs, S.P., Evidence that the nucleomorphs of Chlororachnion reptans (Chlororachniophyceae) are vestigal nuclei: morphologi, division and DNA-DAPI fluorescence, J. Phycol., 1989, vol. 25, p. 385.
Luo, Z., Hu, Z., Tang, Y., Mertens, K.N., Leaw, C.P., Lim, P.T., Teng, S.T., Wang, L., and Gu, H., Morphology, ultrastructure, and molecular phylogeny of Wangodinium sinense Gen. et sp. nov. (Gymnodiniales, Dinophyceae) and revisiting of Gymnodinium dorsalisulcum and Gymnodinium impudicum, J. Phycol., 2018, vol. 54, p. 744. https://doi.org/10.1111/jpy.12780
Sheiner, L., Vaidya, A.B., and McFadden, G.I., The metabolic roles of the endosymbiotic organelles of Toxoplasma and Plasmodium spp., Curr. Opin. Microbiol., 2013, vol. 16, p. 452.
Striepen, B., The apicoplast: a red alga in human parasites, Essays Biochem., 2011, vol. 51, p. 111. https://doi.org/10.1042/BSE0510111
Smith, D.R. and Lee, R.W., A plastid without a genome: evidence from the nonphotosynthetic green algal genus Polytomella, Plant Physiol., 2014, vol. 164, p. 1812.
Larkum, A.W.D., Light-harvesting systems in algae, in Photosynthesis of Algae, Larkum, A.W.D., Douglas, S.E., and Raven, J.A., Eds., Dordrecht: Springer-Verlag, 2003, p. 277.
Tomo, T. and Allakhverdiev S.I., The divergence of chlorophyll and photosynthetic reactions in chlorophyll d-containing algae, in Sovremennye problemy fotosineza (Contemporary Problems of Photosynthesis), Allakhverdiev, S.I., Rubin, A.B., and Shuvalov, V.A., Eds., Moscow, 2014, vol. 2, p. 115.
Averina, S.G., Velichko, N.V., Pinevich, A.A., Senatskaya, E.V., and Pinevich, A.V., Non-a chlorophylls in cyanobacteria, Photosynthetica, 2019, vol. 57, p. 1109.
Raven, J.A., Photosynthesis in watercolours, Nature, 2007, vol. 448, p. 418.
Glazer, A.N., Light guides. Directional energy transfer in a photosynthetic antenna, J. Biol. Chem., 1989, vol. 264, p. 1.
Raven, J.A., A cost-benefit analysis of photon absorption by photosynthetic unicells, New Phytol., 1984, vol. 98, p. 274.
Gorshkova, T.A., Rastitel’naya kletochnaya stenka kak dinamichnaya sistema (The Plant Cell Wall as the Dynamics System), Moscow: Nauka, 2007.
Sarkar, P., Bosneaga, E., and Auer, M., Plant cell walls throughout evolution: towards a molecular understanding of their design principles, J. Exp. Bot., 2009, vol. 60, p. 3615.
Yong, W., O’Malley, R., and Link, B., Plant cell wall genomics, Planta., 2005, vol. 221, p. 745.
Kuznetsov, V.V., The structure and expression of chloroplast genome, Fiziol. Rast., 2018, vol. 65, p. 243.
Martin, W., Rujan, T., Richly, E., Hansen, A., Cornelsen, S., Lins, T., Leister, D., Stoebe, B., Hasegawa, M., and Penny, D., Evolutionary analysis of Arabidopsis, cyanobacterial, and chloroplast genomes reveals plastid phylogeny and thousands of cyanobacterial genes in the nucleus, Proc. Natl. Acad. Sci. U.S.A., 2002, vol. 99, p. 12246.
Nakayama, T., Archibald, J.M., Evolving a photosynthetic organelle, BMC Biol., 2012. https://doi.org/10.1186/1741-7007-10-35
Ponce-Toledo, R.I., López-Garcıa, P., and Moreira, D., Horizontal and endosymbiotic gene transfer in early plastid evolution, New Phytol., 2019, vol. 224, p. 618. https://doi.org/10.1111/nph.15965
Matsuo, M., Ito, Y., Yamauchi, R., and Obokata, J., The rice nuclear genome continuously integrates, shuffles, and eliminates the chloroplast genome to cause chloroplast–nuclear DNA flux, Plant Cell, 2005, vol. 17, p. 665.
Allen, J.F., The CoRR hypothesis for genes in organelles, J. Theor. Biol., 2017, vol. 434, p. 50. https://doi.org/10.1016/j.jtbi.2017.04.008
Tripathi, D., Oldenburg, D.J., Nam, A., and Bendich, A.J., Reactive oxygen species, antioxidant agents, and DNA damage in developing maize mitochondria and plastids, Front. Plant Sci., 2020, vol. 11, p. 596. https://doi.org/10.3389/fpls.2020.00596
Bendich, A.J., Why do chloroplasts and mitochondria contain so many copies of their genome? BioEssays, 1987, vol. 6, p. 279. https://doi.org/10.1002/bies.950060608
Danilenko, N.G. and Davydenko, O.G., Miry genomov organell (The World of Organelle Genomes), Minsk: Tekhnologiya, 2003.
Ling, Q. and Jarvis, P., Regulation of chloroplast protein import by the ubiquitin E3 ligase SP1 is important for stress tolerance in plants, Curr. Biol., 2015, vol. 25, p. 1.
Yurina, N.P. and Odintsova, M.S., Chloroplast retrograde signaling system, Russ. J. Plant Physiol., 2019, vol. 66, p. 509.
Pfannschmidt, T., Terry, M.J., van Aken, O., and Quiros, P.M., Retrograde signals from endosymbiotic organelles: a common control principle in eukaryotic cells, Philos. Trans. R. Soc. B, 2020, vol. 375, art. ID 20190396. https://doi.org/10.1098/rstb.2019.0396
Krupinska, K., Blanco, N.E., Oetke, S., and Zottini, M., Genome communication in plants mediated by organelle–nucleus-located proteins, Philos. Trans. R. Soc. B, 2020, vol. 375, art. ID e 20190397. https://doi.org/10.1098/rstb., 2019.0397
Sinetova, M.A. and Los, D.A., Lessons from cyanobacterial transcriptomics: universal genes and triggers of stress responses, Mol. Biol. (Moscow), 2016, vol. 50, p. 606.
Konstantinov, Yu.M., Dietrich, A., Weber-Lotfi, F., Ibrahim, N., Klimenko, E.S., Tarasenko, V.I., Bolotova, T.A., and Koulintchenko, M.V., DNA import into mitochondria, Biochemistry (Moscow), 2016, vol. 81, no. 10, p. 1044.
Zilberg-Rosenberg, J. and Rosenberg, E., Role of microorganisms in the evolution of animals and plants: the hologenome theory of evolution, FEMS Microbiol. Rev., 2008, vol. 32, p. 723.
Murakami, A., Miyashita, H., Iseki, M., Adachi, K., and Mimuro, M., Chlorophyll d in an epiphytic cyanobacterium of red algae, Science, 2004, vol. 303, p. 1633.
Nakayama, T., Kamikawa, R., Tanifuji, G., Kashiyama, Y., Ohkouchi, N., Archibald, J.M., and Inagaki, Y., Complete genome of a nonphotosynthetic cyanobacterium in a diatom reveals recent adaptations to an intracellular lifestyle, Proc. Natl. Acad. Sci. U.S.A., 2014, vol. 111, p. 11407.
Adams, D.G., Bergman, B., Nierzwicki-Bauer, S.A., Duggan, P.S., Rai, A.N., and Schüßler, A., Cyanobacterial-plant symbioses, in The Prokaryotes: Prokaryotic Biology and Symbiotic Associations, Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., and Thompson, F., Eds., New York: Springer-Verlag, 2002, p. 359.
Pinevich, A.V., Averina, S.G., and Velichko, N.V., Ocherki biologii prokhlorofitov (Biology of Prochlorophytes), St. Petersburg: S.-Peterb. Gos. Univ., 2010.
Tuovinen, V., Ekman, S., Thor, G., Vanderpool, D., Spribille, T., and Johannesson, H., Two basidiomycete fungi in the cortex of wolf lichens, Curr. Biol., 2019, vol. 29, p. 476.
Spribille, T., Lichen symbionts outside of symbiosis: how do they find their match? A commentary on: ‘A case study on the re-establishment of the cyanolichen symbiosis: where do the compatible photobionts come from?’ Ann. Bot., 2019, vol. 124, p. 379.
Johnson, M.D., The acquisition of phototrophy: adaptive strategies of hosting endosymbionts and organelles, Photosynth. Res., 2011, vol. 107, p. 117. https://doi.org/10.1007/s11120-010-9546-8
Stoecker, D.K., Hansen, P.J., Caron, D.A., and Mitra, A., Mixotrophy in the marine plankton, Ann. Rev. Mar. Sci., 2017, vol. 9, p. 311.
Klochkova, T.A., A review of the phenomenon of kleptoplasty in the marine posteriorbranch molluscs, Vestn. Kamchat. Gos. Tekh. Univ., 2016, no. 37, p. 57. https://doi.org/10.17217/2079-0333-2016-37-57-69
Sage, R.F. and Stata, M., Photosynthetic diversity meets biodiversity: the C4 plant example, Plant Physiol., 2015, vol. 172, p. 104.
Ivanishchev, V.V., Evolutionary aspects of C4 photosynthesis, Izv. Tul’sk. Gos. Univ., Estestv. Nauki, 2017, no. 3, p. 64.
Folk, R.A., Soltis, P.S., Soltis, D.E., and Guralnick, R., New prospects in the detection and comparative analysis of hybridization in the tree of life, Am. J. Bot., 2018, vol. 105, p. 364.
Kiu, L.-X., Du, Y.-X., Folk, R.A., Wang, S.-Y., Soltis, D.E., Shang, F.-D., and Li, P., Plastome evolution in Saxifragaceae and multiple plastid capture events involving Heuchera and Tiarella, Front. Plant Sci., 2020, vol. 11, p. 361. https://doi.org/10.3389/fpls.2020.0036183
Brillouet, J.M., Romieu, C., Schoefs, B., Solymosi, K., Cheynier, V., Fulcrand, H., Verdeil, J.L., and Conéjéro, G., The tannosome is an organelle forming condensed tannins in the chlorophyllous organs of Tracheophyta, Ann. Bot., 2013, vol. 112, p. 1003.
Sineshchekov, V.A. and Belyaeva, O.B., Regulation of chlorophyll biogenesis by phytochrome A, Biochemistry (Moscow), 2019, vol. 84, no. 5, p. 491.
Kuznetsov, V.V., doroshenko, A.S., Kudryakova, N.V., and Danilova, M.N., Role of phytohormones and light in de-etiolation, Russ. J. Plant Physiol., 2020, vol. 67, no. 6, p. 971.
Jarvis, P. and López-Juez, E., Biogenesis and homeostasis of chloroplasts and other plastids, Nat. Rev. Mol. Cell Biol., 2014, vol. 27, p. 787.
Westwood, J.H., Yoder, J.I., Timko, M.P., and de Pamphilis, C.W., Evolution of parasitism in plants, Trends Plant Sci., 2010, vol. 15, p. 227.
Filyushin, M.A., Kochieva, E.Z., Shchennikova, A.V., Beletsky, A.V., Mardanov, A.V., Ravin, N.V., and Skryabin, K.G., SWEET uniporter gene family expression profile in the pitcher development in the carnivorous plant Nepenthes sp., Russ. J. Genet., 2019, vol. 55, p. 692.
Funding
This work was supported by the Russian Foundation for Basic Research, project no. 19-14-50366.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflicts of interest.
Statement on the welfare of humans or animals. This article does not contain any studies involving animals performed by any of the authors.
Additional information
Translated by A. Aver’yanov
Abbreviations: Chl—chlorophyll.
Rights and permissions
About this article
Cite this article
Stadnichuk, I.N., Kusnetsov, V.V. Endosymbiotic Origin of Chloroplasts in Plant Cells’ Evolution. Russ J Plant Physiol 68, 1–16 (2021). https://doi.org/10.1134/S1021443721010179
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443721010179