Abstract
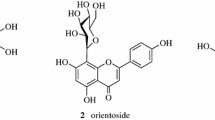
A preparative high-speed counter-current chromatography method for isolation and purification of three acetophenones, baishouwubenzophenone, 4-hydroxyacetophenone and 2,4-dihydroxyacetophenone from the Chinese medicinal plant Cynanchum auriculatum Royle ex Wight was successfully established. With a two-phase solvent system composed of light petroleum (b.p. 60–90 °C)–ethyl acetate–methanol–water (4:9:6:6, v/v), about 20.2 mg compound 1, 35.0 mg compound 2 and 8.3 mg compound 3, each at over 95% purity as determined by LC, were obtained in one-step elution from 400 mg of the ethanol extract. The structures of these compounds were identified by UV, IR, ESI-MS, 1H NMR and 13C NMR spectroscopy. Among them, compounds 2,4-dihydroxyacetophenone and 4-hydroxyacetophenone were obtained from C. auriculatum for the first time.



Similar content being viewed by others
References
Zhang RS, Liu YL, Wang YQ, Ye YP, Li XY (2007) Chem Biodivers 4:887–892. doi:10.1002/cbdv.200790076
Chen JJ, Zhang ZX, Zhou J (1990) Acta Bot Yunnan 12:197–210
Gong SS, Liu CD, Liu SL, Du YR, Kang W, Dong XQ (1988) Acta Pharmacol Sin 23:276–280
Zhang JF, Li YB, Qian SH, Li CL, Jiang JQ (2006) Chin J Chin Mater Med 31:814–816
Peng YR, Li YB, Liu XD, Zhang JF, Duan JA (2008) Phytomedicine 15:391–395. doi:10.1016/j.phymed.2008.02.021
Zhang RS, Ye YP, Shen YM, Liang HL (2000) Acta Pharmacol Sin 35:431–437
Zhang RS, Ye YP, Liu XL (2000) Chin Tradit Herbal Drugs 31:599–601
Wang DY, Zhang HQ, Li X (2007) Acta Pharmacol Sin 42:366–370
Song JM, Wang YX, Ding XL (2001) Food Sci 22:22–25
Song JM, Ding XL (1998) Chin Wild Plant Resour 17:1–4
Shan L, Liu RH, Shen YH, Zhang WD, Zhang C, Wu DZ, Min L, Su J, Xu XK (2006) J Ethnopharmacol 107:389–394. doi:10.1016/j.jep.2006.03.022
Zhang SX, Li X, Yin JL, Chen LL, Zhang HQ (2007) Pract Geriatr 21:104–107
Yin L, Li X, Zhang SX, Zhang HQ (2007) Anhui Med Pharm J 11:198–200
Zhang YH, Wen YY, Kuang TY (1999) Nat Prod Res Dev 12:80–87
Li J, Kadota S, Kawata Y, Hattori M, Xu GJ, Namba T (1992) Chem Pharm Bull (Tokyo) 40:3133–3137
Ito Y (1991) J Chromatogr A 538:3–25. doi:10.1016/S0021-9673(01)91617-6
Wang X, Liu JH, Zhang TY, Ito Y (2007) J Liq Chromatogr Relat Technol 30:2585–2592. doi:10.1080/10826070701540522
Li L, Tsao R, Yang R, Liu CM, Young JC, Zhu HH (2008) Food Chem 108:702–710. doi:10.1016/j.foodchem.2007.10.082
Liu ZL, Yu Y, Shen PN, Wang J, Wang CY, Shen YJ (2008) Sep Purif Technol 58:343–346. doi:10.1016/j.seppur.2007.05.011
Kohler N, Wray V, Winterhalter P (2008) J Chromatogr A 1177:114–125. doi:10.1016/j.chroma.2007.11.028
Liu R, Lu YB, Wu TX, Pan YJ (2008) Chromatographia 68:95–99. doi:10.1365/s10337-008-0629-z
Gu DY, Yang Y, Zhong J, Aisa HA, Zhang TY (2007) Chromatographia 66:949–951. doi:10.1365/s10337-007-0432-2
Zhang JJ (1997) Master’s Thesis, Beijing University of Chinese Medicine, pp 31–35
Acknowledgments
Financial support from the Ministry of Science & Technology of China (No. 2005DKA21000) and Shandong Agricultural University (No. 23489) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sun, Y., Liu, Z., Wang, J. et al. Separation and Purification of Baishouwubenzophenone, 4-Hydroxyacetophenone and 2,4-Dihydroxyacetophenone from Cynanchum auriculatum Royle ex Wight by HSCCC. Chroma 70, 1–6 (2009). https://doi.org/10.1365/s10337-009-1107-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1365/s10337-009-1107-y