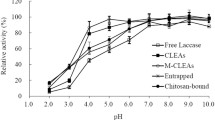
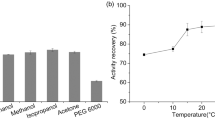
Abstract
A coordinatively immobilized laccase was prepared using a new cryogel type carrier. The support has a wide-pore texture facilitating diffusion of different substrates to the enzyme reaction center. The biocatalyst proved to be efficient in decolorization of two anthraquinone derivatives, namely Acid Blue 62 and bromaminic acid. After 24 h over 80% of the two substrates have been oxidated. The kinetic data (K m and V max) for the oxidation of the two anthraquinone derivatives, with the free and immobilized enzyme, have been determined and compared. Other parameters, like k cat and the specificity constant have been calculated and analyzed. The influence of substrate properties (hydrophobicity, polarity, etc.) has been discussed.





Similar content being viewed by others
References
Venkata Mohan, S., & Karthikeyan, J. (2004). Clean Technologies Environmental Policy, 6, 196–200.
Hai, F. I., Yamamoto, K., & Fukushi, K. (2007). Critical Reviews in Environmental Science and Technology, 37, 315–377.
Crini, G. (2006). Bioresource Technology, 97, 1061–1085.
Prigione, V., Tigini, V., Pezzella, C., Anastasi, A., Sannia, G., & Varese, G. C. (2008). Water Research, 42, 2911–2920.
Bes-Pia, A., Mendoza-Roca, J. A., Roig-Alcover, R., Iborra-Clar, A. I., Iborra-Clar, M. I., & Alcaina-Miranda, M. I. (2003). Desalination, 157, 73–80.
Lachheb, H., Puzenat, E., Houas, A., Ksibi, M., Elaloui, E., Guillard, C., et al. (2002). Applied Catalysis B: Environmental, 39, 75–90.
Van der Zee, F. (2002). Anaerobic azo dye reduction, Ph. D. Thesis Wageningen University, Wageningen, Netherlands
Gianfreda, L., Xu, F., & Bollag, J. (1999). Bioremediation Journal, 3, 4–25.
Zille, A. (2005) Lacasse reactions for Textile Applications, Ph. D. Thesis University of Minho, Guimaraes, Portugal
Desai, S. S., & Nityanand, C. (2011). Asian Journal of Biotechnology, 3, 98–124.
Niladevi, K. N., Niladevi, K. N., Sheejadevi, P. S., & Prema, P. (2008). Applied Biochemistry and Biotechnology, 151, 9–19.
Rodriguez Couto, S. (2007). Journal of Hazardous Materials, 149, 768–770.
Khlifi, R., Belbahri, L., Woodward, S., Ellouz, M., Dhouib, A., Sayadi, S., et al. (2010). Journal of Hazardous Materials, 175, 802–808.
Majeau, J.-A., Brar, S. K., & Tyagi, R. D. (2010). Bioresource Technology, 101, 2331–2350.
Vanhulle, S., Enaud, E., Trovaslet, M., Billottet, L., Kneipe, L., Jiwan, J.-L. H., et al. (2008). Chemosphere, 70, 1097–1107.
Baldrian, P., & Snajdr, J. (2006). Enzyme and Microbial Technology, 39, 1023–1029.
Michniewicz, A., Ledakowicz, S., Ullrich, R., & Hofrichter, M. (2008). Dyes and Pigments, 77, 295–302.
Neifar, M., Jaouani, A., Ellouze-Ghorbel, R., & Ellouze-Chaabouni, S. (2010). Journal of Molecular Catalysis B: Enzymatic, 64, 68–74. doi:10.1016/j.molcatb.2010.02.004.
Russo, M. E., Giardina, P., Marzocchella, A., Salatino, P., & Sannia, G. (2008). Enzyme and Microbial Technology, 42, 521–530.
Yang, X. Q., Zhao, X. X., Liu, C. Y., Zheng, Y., & Qian, S. J. (2009). Process Biochemistry, 44, 1185–1189.
Champagne, P.-P., & Ramsay, J. A. (2010). Applied Microbiology and Biotechnology, 101, 2230–2235.
Duran, N., Rosa, M. A., D’Annibale, A., & Gianfreda, L. (2002). Enzyme and Microbial Technology, 31, 907–931.
Rasera, K., Ferla, J., Dillon, A. J. P., Riveiros, R., & Zeni, M. (2009). Desalination, 245, 657–661.
Bayramoglu, G., Ylmaz, M., & Arica, M. Y. (2010). Bioresource Technology, 101, 6615–6621.
De Stefano, G., Piacquadio, P., & Sciancalepore, V. (1996). Biotechnology Techniques, 10, 857–860.
Piacquadio, P., De Stefano, G., Sammartino, M., & Sciancalepore, V. (1997). Biotechnology Techniques, 11, 515–517.
Lozinsky, V. I., Galaev, I. Y., Plieva, F. M., Savina, I. N., Jungvid, H., & Mattiasson, B. (2003). Trends in Biotechnology, 21, 445–451.
Lozinsky, V. I. (2008). Russian Chemical Bulletin, 57, 1015–1032.
Plieva, F. M., Galaev, I. Y., Noppe, W., & Mattison, B. (2008). Trends in Microbiology, 16, 543–551.
Bradford, M. M. (1976). Analytical Biochemistry, 72, 248–254.
Stanescu, M. D., Fogorasi, M., Shaskolskiy, B. L., Gavrilas, S., & Lozinsky, V. I. (2010). Applied Biochemistry and Biotechnology, 160, 1947–1954. doi:10.1007/s12010-009-8755-0.
Lozinsky, V. I., Morozova, S. A., Vainerman, E. S., Titova, E. F., Shtil’man, M. I., Belavtseva, E. M., et al. (1989). Acta Polymerica, 40, 8–15.
Arvidsson, P., Plieva, F. M., Savina, I. N., Lozinsky, V. I., Fexby, S., Bulow, L., et al. (2002). Journal of Chromatography. A, 977, 27–38.
Plieva, F. M., Karlsson, M., Aguilar, M.-R., Gomez, D., Mikhalovsky, S., & Galaev, I. Yu. (2005). Soft Matter, 1, 303–309.
Do Rosario Freixo, M., Karmali, A., Frazao, C., & Arteiro, J. M. (2008). Process Biochemistry, 43, 1265–1274.
Enaud, E., Trovaslet, M., Bruyneel, F., Billottet, L., Karaaslan, R., Sener, M. E., et al. (2010). Dyes and Pigments, 85, 99–108.
Northrop, D. B. (1999). Advances in Enzymology and Related Areas of Molecular Biology, 73, 25–55.
Rekuć, A., Jastrzembska, B., Liesiene, J., & Bryjak, J. (2009). Journal of Molecular Catalysis B: Enzymatic, 57, 216–223.
Acknowledgments
Thanks for financial support are due to the Romanian Academy (grant-1R-2008) and the Russian Foundation for Basic Research (Project no. 07-03-91682_RA_a)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stanescu, M.D., Sanislav, A., Ivanov, R.V. et al. Immobilized Laccase on a New Cryogel Carrier and Kinetics of Two Anthraquinone Derivatives Oxidation. Appl Biochem Biotechnol 165, 1789–1798 (2011). https://doi.org/10.1007/s12010-011-9395-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-011-9395-8