Abstract
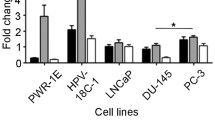
MicroRNAs (miRNAs) are small non-coding RNAs that function in transcriptional and post-transcriptional regulation of gene expression. Several miRNAs have been implicated in regulating prostate cancer (PCa) progression. Deregulations of miRNA regulatory networks have been reported in ERG positive PCa, which accounts for ~50 % of PCa and have been suggested to affect tumor aggressiveness. The function of miR338-3p, its prognostic significance, and its association with ERG positive PCa has not been fully investigated. Using microarray expression profiling, we identified miRNA338-3p as among the top deregulated miRNAs associated with ERG status in PCa. We investigated miR338-3p function using in vitro and in vivo experimental models and its expression was assessed and validated in clinical samples and a public cohort of localized and metastatic prostate cancer. miR338-3p was significantly down-regulated with disease progression from benign prostate tissue to primary and metastatic lesions. In localized disease, patients with lower miR338-3p expression levels showed increased association to biochemical recurrence and several adverse pathological parameters compared to patients with higher miRNA338-3p tissue expression levels. Using in vitro PCa cell models, overexpression of miR338-3p resulted in a decrease in cell invasion and expression of chemokine signalling genes CXCL12, CXCR4, and CXCR7. In vivo, orthotropic implantation of PC3 cells stably expressing miR338-3p was associated with a significant decrease in tumor weights compared to control cells. miR338-3p has anti-proliferative and anti-invasive properties. It affects CXCR4 axis, and its down-regulation is associated with adverse clinical outcomes in PCa patients.





Similar content being viewed by others
Abbreviations
- AR:
-
Androgen receptor
- CRPC:
-
Castration resistant prostate cancer
- GS:
-
Gleason score
- miRNA:
-
microRNA
- PCa:
-
Prostate cancer
- PSA:
-
Prostate specific antigen
- MSKCC:
-
Memorial Sloan-Kettering Cancer Center
- SCID:
-
Severe combined immunodeficiency
- TFs:
-
Transcription factors
References
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65(1):5–29. doi:10.3322/caac.21254
Fong MK, Hare R, Jarkowski A (2012) A new era for castrate resistant prostate cancer: a treatment review and update. J Oncol Pharm Pract. doi:10.1177/1078155212437599
Tomlins SA, Rhodes DR, Perner S, Dhanasekaran SM, Mehra R, Sun XW et al (2005) Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. Science 310(5748):644–648. doi:10.1126/science.1117679
Pettersson A, Graff RE, Bauer SR, Pitt MJ, Lis RT, Stack EC et al (2012) The TMPRSS2: ERG rearrangement, ERG expression, and prostate cancer outcomes: a cohort study and meta-analysis. Cancer Epidemiol Biomarker Prev 21(9):1497–1509. doi:10.1158/1055-9965.EPI-12-0042
Tomlins SA, Rhodes DR, Yu J, Varambally S, Mehra R, Perner S et al (2008) The role of SPINK1 in ETS rearrangement-negative prostate cancers. Cancer Cell 13(6):519–528. doi:10.1016/j.ccr.2008.04.016
Carver BS, Tran J, Gopalan A, Chen Z, Shaikh S, Carracedo A et al (2009) Aberrant ERG expression cooperates with loss of PTEN to promote cancer progression in the prostate. Nat Genet 41(5):619–624. doi:10.1038/ng.370
Leshem O, Madar S, Kogan-Sakin I, Kamer I, Goldstein I, Brosh R et al (2011) TMPRSS2/ERG promotes epithelial to mesenchymal transition through the ZEB1/ZEB2 axis in a prostate cancer model. PLoS ONE 6(7):e21650. doi:10.1371/journal.pone.0021650
Casey OM, Fang L, Hynes PG, Abou-Kheir WG, Martin PL, Tillman HS et al (2012) TMPRSS2- driven ERG expression in vivo increases self-renewal and maintains expression in a castration resistant subpopulation. PLoS ONE 7(7):e41668. doi:10.1371/journal.pone.0041668
Fayyaz S, Farooqi AA (2013) miRNA and TMPRSS2-ERG do not mind their own business in prostate cancer cells. Immunogenetics 65(5):315–332. doi:10.1007/s00251-012-0677-2
Wang G, Wang Y, Feng W, Wang X, Yang JY, Zhao Y et al (2008) Transcription factor and microRNA regulation in androgen-dependent and -independent prostate cancer cells. BMC Genomics 9(Suppl 2):S22. doi:10.1186/1471-2164-9-S2-S22
Selcuklu SD, Donoghue MT, Spillane C (2009) miR-21 as a key regulator of oncogenic processes. Biochem Soc Trans 37(Pt 4):918–925. doi:10.1042/BST0370918
Gordanpour A, Stanimirovic A, Nam RK, Moreno CS, Sherman C, Sugar L et al (2011) miR-221 Is down-regulated in TMPRSS2:ERG fusion-positive prostate cancer. Anticancer Res 31(2):403–410
Hart M, Wach S, Nolte E, Szczyrba J, Menon R, Taubert H et al (2013) The proto-oncogene ERG is a target of microRNA miR-145 in prostate cancer. FEBS J 280(9):2105–2116. doi:10.1111/febs.12236
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116(2):281–297
Lai EC (2002) Micro RNAs are complementary to 3′ UTR sequence motifs that mediate negative post-transcriptional regulation. Nat Genet 30(4):363–364. doi:10.1038/ng865
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120(1):15–20. doi:10.1016/j.cell.2004.12.035
Xie X, Lu J, Kulbokas EJ, Golub TR, Mootha V, Lindblad-Toh K et al (2005) Systematic discovery of regulatory motifs in human promoters and 3′ UTRs by comparison of several mammals. Nature 434(7031):338–345. doi:10.1038/nature03441
Chen X, Gong J, Zeng H, Chen N, Huang R, Huang Y et al (2010) MicroRNA145 targets BNIP3 and suppresses prostate cancer progression. Cancer Res 70(7):2728–2738. doi:10.1158/0008-5472.CAN-09-3718
Mezzanzanica D, Bagnoli M, De Cecco L, Valeri B, Canevari S (2010) Role of microRNAs in ovarian cancer pathogenesis and potential clinical implications. Int J Biochem Cell Biol 42(8):1262–1272. doi:10.1016/j.biocel.2009.12.017
Dong Q, Meng P, Wang T, Qin W, Qin W, Wang F et al (2010) MicroRNA let-7a inhibits proliferation of human prostate cancer cells in vitro and in vivo by targeting E2F2 and CCND2. PLoS ONE 5(4):e10147. doi:10.1371/journal.pone.0010147
Barik S (2008) An intronic microRNA silences genes that are functionally antagonistic to its host gene. Nucleic Acids Res 36(16):5232–5241. doi:10.1093/nar/gkn513
Tomomura M, Fernandez-Gonzales A, Yano R, Yuzaki M (2001) Characterization of the apoptosis-associated tyrosine kinase (AATYK) expressed in the CNS. Oncogene 20(9):1022–1032. doi:10.1038/sj.onc.1204210
Wienholds E, Kloosterman WP, Miska E, Alvarez-Saavedra E, Berezikov E, de Bruijn E et al (2005) MicroRNA expression in zebrafish embryonic development. Science 309(5732):310–311. doi:10.1126/science.1114519
Kos A, Olde Loohuis NF, Wieczorek ML, Glennon JC, Martens GJ, Kolk SM et al (2012) A potential regulatory role for intronic microRNA-338-3p for its host gene encoding apoptosis-associated tyrosine kinase. PLoS ONE 7(2):e31022. doi:10.1371/journal.pone.0031022
Tsuchiya S, Oku M, Imanaka Y, Kunimoto R, Okuno Y, Terasawa K et al (2009) MicroRNA-338-3p and microRNA-451 contribute to the formation of basolateral polarity in epithelial cells. Nucleic Acids Res 37(11):3821–3827. doi:10.1093/nar/gkp255
Murphy PM, Baggiolini M, Charo IF, Hebert CA, Horuk R, Matsushima K et al (2000) International union of pharmacology. XXII. Nomenclature for chemokine receptors. Pharmacol Rev 52(1):145–176
Fredriksson R, Lagerstrom MC, Lundin LG, Schioth HB (2003) The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol Pharmacol 63(6):1256–1272. doi:10.1124/mol.63.6.1256
Zlotnik A (2006) Involvement of chemokine receptors in organ-specific metastasis. Contrib Microbiol 13:191–199. doi:10.1159/000092973
Begley LA, MacDonald JW, Day ML, Macoska JA (2007) CXCL12 activates a robust transcriptional response in human prostate epithelial cells. J Biol Chem 282(37):26767–26774. doi:10.1074/jbc.M700440200
Uygur B, Wu WS (2011) SLUG promotes prostate cancer cell migration and invasion via CXCR4/CXCL12 axis. Mol Cancer 10:139. doi:10.1186/1476-4598-10-139
Sun YX, Wang J, Shelburne CE, Lopatin DE, Chinnaiyan AM, Rubin MA et al (2003) Expression of CXCR4 and CXCL12 (SDF-1) in human prostate cancers (PCa) in vivo. J Cell Biochem 89(3):462–473. doi:10.1002/jcb.10522
Singareddy R, Semaan L, Conley-Lacomb MK, St John J, Powell K, Iyer M et al (2013) Transcriptional regulation of CXCR4 in prostate cancer: significance of TMPRSS2-ERG fusions. Mol Cancer Res 11(11):1349–1361. doi:10.1158/1541-7786.MCR-12-0705
Taylor BS, Schultz N, Hieronymus H, Gopalan A, Xiao Y, Carver BS et al (2010) Integrative genomic profiling of human prostate cancer. Cancer Cell 18(1):11–22. doi:10.1016/j.ccr.2010.05.026
Pettaway CA, Pathak S, Greene G, Ramirez E, Wilson MR, Killion JJ et al (1996) Selection of highly metastatic variants of different human prostatic carcinomas using orthotopic implantation in nude mice. Clin Cancer Res 2(9):1627–1636
Yang JH, Li JH, Jiang S, Zhou H, Qu LH (2013). ChIPBase: a database for decoding the transcriptional regulation of long non-coding RNA and microRNA genes from ChIP-Seq data. Nucleic Acids Res 41(Database issue):D177–D187. doi:10.1093/nar/gks1060
Guo B, Liu L, Yao J, Ma R, Chang D, Li Z et al (2014) miR-338-3p suppresses gastric cancer progression through a PTEN-AKT axis by targeting P-REX2a. Mol Cancer Res 12(3):313–321. doi:10.1158/1541-7786.MCR-13-0507
Xue Q, Sun K, Deng HJ, Lei ST, Dong JQ, Li GX (2014) MicroRNA-338-3p inhibits colorectal carcinoma cell invasion and migration by targeting smoothened. Jpn J Clin Oncol 44(1):13–21. doi:10.1093/jjco/hyt181
Huang XH, Wang Q, Chen JS, Fu XH, Chen XL, Chen LZ et al (2009) Bead-based microarray analysis of microRNA expression in hepatocellular carcinoma: miR-338 is downregulated. Hepatol Res 39(8):786–794. doi:10.1111/j.1872-034X.2009.00502.x
Krol J, Loedige I, Filipowicz W (2010) The widespread regulation of microRNA biogenesis, function and decay. Nat Rev Genet 11(9):597–610. doi:10.1038/nrg2843
Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E et al (2007) A MicroRNA feedback circuit in midbrain dopamine neurons. Science 317(5842):1220–1224. doi:10.1126/science.1140481
Walter BA, Valera VA, Pinto PA, Merino MJ (2013) Comprehensive microRNA Profiling of Prostate Cancer. J Cancer 4(5):350–357. doi:10.7150/jca.6394
Keeley EC, Mehrad B, Strieter RM (2010) CXC chemokines in cancer angiogenesis and metastases. Adv Cancer Res 106:91–111. doi:10.1016/S0065-230X(10)06003-3
Koizumi K, Hojo S, Akashi T, Yasumoto K, Saiki I (2007) Chemokine receptors in cancer metastasis and cancer cell-derived chemokines in host immune response. Cancer Sci 98(11):1652–1658. doi:10.1111/j.1349-7006.2007.00606.x
Wagner PL, Hyjek E, Vazquez MF, Meherally D, Liu YF, Chadwick PA et al (2009) CXCL12 and CXCR4 in adenocarcinoma of the lung: association with metastasis and survival. J Thorac Cardiovasc Surg 137(3):615–621. doi:10.1016/j.jtcvs.2008.07.039
Smith MC, Luker KE, Garbow JR, Prior JL, Jackson E, Piwnica-Worms D et al (2004) CXCR4 regulates growth of both primary and metastatic breast cancer. Cancer Res 64(23):8604–8612. doi:10.1158/0008-5472.CAN-04-1844
Cai J, Kandagatla P, Singareddy R, Kropinski A, Sheng S, Cher ML et al (2010) Androgens Induce Functional CXCR4 through ERG Factor Expression in TMPRSS2-ERG Fusion-Positive Prostate Cancer Cells. Transl Oncol 3(3):195–203
Carlisle AJ, Lyttle CA, Carlisle RY, Maris JM (2009) CXCR4 expression heterogeneity in neuroblastoma cells due to ligand-independent regulation. Mol Cancer 8:126. doi:10.1186/1476-4598-8-126
Li X, Ma Q, Xu Q, Liu H, Lei J, Duan W et al (2012) SDF-1/CXCR4 signaling induces pancreatic cancer cell invasion and epithelial-mesenchymal transition in vitro through non-canonical activation of Hedgehog pathway. Cancer Lett 322(2):169–176. doi:10.1016/j.canlet.2012.02.035
Huang XH, Chen JS, Wang Q, Chen XL, Wen L, Chen LZ et al (2011) miR-338-3p suppresses invasion of liver cancer cell by targeting smoothened. J Pathol 225(3):463–472. doi:10.1002/path.2877
Sun K, Deng HJ, Lei ST, Dong JQ, Li GX (2013) miRNA-338-3p suppresses cell growth of human colorectal carcinoma by targeting smoothened. World J Gastroenterol 19(14):2197–2207. doi:10.3748/wjg.v19.i14.2197
Ok S, Kim SM, Kim C, Nam D, Shim BS, Kim SH et al (2012) Emodin inhibits invasion and migration of prostate and lung cancer cells by downregulating the expression of chemokine receptor CXCR4. Immunopharmacol Immunotoxicol 34(5):768–778. doi:10.3109/08923973.2012.654494
Shanmugam MK, Manu KA, Ong TH, Ramachandran L, Surana R, Bist P et al (2011) Inhibition of CXCR4/CXCL12 signaling axis by ursolic acid leads to suppression of metastasis in transgenic adenocarcinoma of mouse prostate model. Int J Cancer 129(7):1552–1563. doi:10.1002/ijc.26120
Weinberg RA (2014) The Biology of Cancer Garland Science, 2nd edn. Taylor & Francis Group, LLC, New York
Acknowledgments
The authors would like to thank Shuhong Liu, Sabrina Daniela Silva, Samir Al Bashir, Tingting Wang and Liang Hong Teng for technical assistance in this study.
Author contribution
AB; executed and planned in vitro experiments and drafted the manuscript. MA and RA; carried out all bioinformatics work in this study and assisted in drafting manuscript. LP; carried out ERG cloning experiments and participated in the preparation of figures; HA helped in performing qPCR experiments, writing the manuscript and preparing of figures; and AA and TW performed western blots, SH performed Q-PCR and contributed to manuscript writing, KB carried out in vitro experiments. MAJ designed and supervised in vitro and in vivo experiments of the study and revised manuscript. TAB; carried out pathological analysis, and contributed to manuscript drafting.
Funding
This work was supported by the Prostate Cancer Foundation Young Investigator Award (T.A.B) and in part by the Canadian Institutes for Health Research, the Canadian Cancer Society, and Quebec Breast Cancer Foundation (MAAJ). This work was also supported by Prostate cancer Canada and is proudly funded by the Movember Foundation-Grant #B2013-01.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare in this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bakkar, A., Alshalalfa, M., Petersen, L.F. et al. microRNA 338-3p exhibits tumor suppressor role and its down-regulation is associated with adverse clinical outcome in prostate cancer patients. Mol Biol Rep 43, 229–240 (2016). https://doi.org/10.1007/s11033-016-3948-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-016-3948-4