Abstract
Background and Objective
Selecting the most effective treatment for major depressive disorder (MDD) is a challenge for clinicians. The aim of this study was to compare the effects of sertraline with duloxetine on major depression signs and symptoms.
Methods
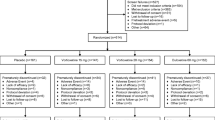
The trial was a 6-week, randomized, controlled, double-blind study. Sixty-three patients with diagnosis of MDD according to DSM-IV-TR criteria were randomly assigned to receive either duloxetine (31 patients) or sertraline (32 patients). The mean dosage of duloxetine was 55 mg/day (range 40–60 mg/day) and the mean dosage of sertraline was 146 mg/day (range 50–200 mg/day). Subjects were assessed at baseline, and at the end of week 6. Depression severity and symptoms were assessed by 21-item Hamilton Depression Rating Scale (HAM-D).
Results
Of 63 patients who were randomized to treatment, 54 patients including 28 in the sertraline group and 26 in the duloxetine group completed the trial. The HAM-D total score for both groups was significantly reduced at the end of the trial period without significant difference from each other (p = 0.463). Of the symptoms studied, psychomotor retardation, general somatic symptoms and sexual problems improved more in the duloxetine group. On the other hand, agitation, anxiety symptoms and hypochondriasis ameliorated better in the sertraline group. There was no difference between the two groups regarding the other symptoms.
Conclusions
Our study shows that the antidepressant mechanism of action has influence on its effects on different signs and symptoms. Clinician awareness of an antidepressant’s special effects can help in selecting appropriate medicine.
Similar content being viewed by others
References
Mendlewich J. Optimizing antidepressant use in clinical practice: towards criteria for antidepressant selection. Br J Psychiatry. 2001;179(suppl):1–3.
Nelson JC, Kennedy JS, Pollock BG, et al. Treatment of major depression with nortriptyline and paroxetine in patients with ischemic heart disease. Am J Psychiatry. 1999;156(7):1024–8.
Mowla A, Ghanizadeh A, Pani A. A comparison of effects of fluoxetine and nortriptyline on the symptoms of major depressive disorder. J Clin Psychopharmacol. 2006;26(2):209–11.
Kim HM, Zivin K, Choe HM. Predictors of start of different antidepressants in patient charts among patients with depression. J Manag Care Spec Pharm. 2015;21(5):424–30.
Montgomery SA. Is there a role for a pure noradrenergic drug in the treatment of depression? Eur Neuropsychopharmacol. 1997;7(suppl 1):S3–9.
Healy D, McMonagle T. The enhancement of social functioning as a therapeutic principle in the management of depression. J Psychopharmacol. 1997;11(suppl 4):S25–31.
Montgomery SA. Selective serotonin reuptake inhibitors in the acute treatment of depression. In: Bloom FE, Kupfer DJ, editors. Psychopharmacology: the fourth generation of progress. New York: Raven; 1995.
Montgomery SA. The efficacy of fluoxetine as an antidepressant in the short and long term. Int Clin Psychopharmacol. 1989;4(suppl 1):113–9.
Karpa KD, Cavanaugh JE, Lakoski JM. Duloxetine pharmacology: profile of a dual monoamine modulator. CNS Drug Rev. 2002;8(4):361–76.
Petersen T, Dording C, Neault NB, et al. A survey of prescribing practices in the treatment of depression. Prog Neuropsychopharmacol Biol Psychiatry. 2002;26(1):177–87.
Lee P, Shu L, Xu X, et al. Once-daily duloxetine 60 mg in the treatment of major depressive disorder: multicenter, double-blind, randomized, paroxetine-controlled, non-inferiority trial in China, Korea, Taiwan and Brazil. Psychiatry Clin Neurosci. 2007;61(3):295–307.
Pigott TA, Prakash A, Arnold LM, Aaronson ST, Mallinckrodt CH, Wohlreich MM. Duloxetine versus escitalopram and placebo: an 8-month, double-blind trial in patients with major depressive disorder. Curr Med Res Opin. 2007;23(6):1303–18.
Nierenberg AA, Greist JH, Mallinckrodt CH, et al. Duloxetine versus escitalopram and placebo in the treatment of patients with major depres-sive disorder: onset of antidepressant action, a non-inferiority study. Curr Med Res Opin. 2007;23(3):401–16.
Häuser W, Petzke F, Sommer C. Comparative efficacy and harms of duloxetine, milnacipran, and pregabalin in fibromyalgia syndrome. J Pain. 2010;11:505–21.
Jones CK, Peters SC, Shannon HE. Synergistic interactions between the dual serotonergic, noradrenergic reuptake inhibitor duloxetine and the non-steroidal anti-inflammatory drug ibuprofen in inflammatory pain in rodents. Eur J Pain. 2007;11(2):208–15.
Sanchez C, Reines EH, Montgomery SA. A comparative review of escitalopram, paroxetine, and sertraline: are they all alike? Int Clin Psychopharmacol. 2014;29(4):185–96.
Hamilton M. Rating depressive patients. J Clin Psychiatry. 1980;41(12 part 2):21–4.
Nelson JC, Clary CM, Leon AC, et al. Symptoms of late life depression: frequency and change during treatment. Am J Geriatr Psychiatry. 2005;13(6):520–6.
Nelson JC, Portera L, Leon AC. Are there differences in the symptoms that respond to a selective serotonin or norepinephrine reuptake inhibitor? Biol Psychiatry. 2005;57(12):1535–42.
Cipriani A, Koesters M, Furukawa TA. Duloxetine versus other anti-depressive agents for depression. Cochrane Database Syst Rev. 2012;17(10):CD006533. doi:10.1002/14651858.CD006533.
Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 12 new-generation antidepressants: a multiple treatments meta-analysis. Lancet. 2009;373:746–58.
Khoo AL, Zhou HJ, Teng M, et al. Network meta-analysis and cost-effectiveness analysis of new generation antidepressants. CNS Drugs. 2015;29(8):695–712.
Grover S, Shah R, Dutt A, et al. Prevalence and pattern of sexual dysfunction in married females receiving antidepressants: an exploratory study. J Pharmacol Pharmacother. 2012;3(3):259–65.
Montgomery SA, Baldwin DS, Riley A. Antidepressant medications: a review of the evidence for drug-induced sexual dysfunction. J Affect Disord. 2002;69:119–40.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by vice chancellor for research of Shiraz University of Medical Sciences with Grant Number 93-01-34-8919.
Conflicts of interest
AM, SAD and LRJ have no conflicts of interest that are directly relevant to the content of this study.
Ethical approval
The study was approved by the ethics committee of Shiraz University of Medical Sciences that adheres to the Declaration of Helsinki Ethical Principles, 1964 for Medical Research.
Informed consent
All patients provided written informed consent to participate in the study.
Rights and permissions
About this article
Cite this article
Mowla, A., Dastgheib, S.A. & Razeghian Jahromi, L. Comparing the Effects of Sertraline with Duloxetine for Depression Severity and Symptoms: A Double-Blind, Randomized Controlled Trial. Clin Drug Investig 36, 539–543 (2016). https://doi.org/10.1007/s40261-016-0399-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-016-0399-6