Summary
Presynaptic α2-autoreceptors in rat and rabbit brain cortex were compared by means of antagonists and agonists. Brain cortex slices were preincubated with [3H]-noradrenaline and then superfused and stimulated by 3 (rat) or 4 (rabbit) pulses at a frequency of 100 Hz.
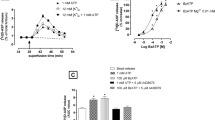
The α2-adrenoceptor agonist bromoxidine (UK 14 304) reduced the electrically evoked overflow of tritium with EC50 values of 4.5 nmol/l in the rat and 0.7 nmol/l in the rabbit. The antagonists phentolamine, 2-[2H-(1-methyl-1,3-dihydroisoindole)methyl]-4,5-dihydroimidazole (BRL 44408), rauwolscine, 1,2-dimethyl-2,3,9,13b-tetrahydro-1H-dibenzo(c,f)imidazo(1,5-a)azepine (BRL 41992), 2-(2,6-dimethoxyphenoxyethyl)aminomethyl-1,4-benzodioxane (WB 4101), 6-chloro-9-[(3-methyl-2-butenyl)oxy]-3-methyl-1H-2,3,4, 5-tetrahydro-3-benzazepine (SKF 104078), imiloxan, prazosin and corynanthine did not per se increase the evoked overflow of tritium but shifted the concentration-inhibition curve of bromoxidine to the right in a manner compatible with competitive antagonism. Up to 4 concentrations of each antagonist were used to determine its dissociation constant KD. The KD values correlated only weakly between the rat and the rabbit. Dissociation constants KA of bromoxidine were calculated from equieffective concentrations in unpretreated brain slices and slices in which part of the α2-adrenoceptors had been irreversibly blocked by phenoxybenzamine. The KA value was 123 nmol/l in the rat and 7.2 nmol/l in the rabbit.
The results confirm the species difference between rat and rabbit brain presynaptic α2-autoreceptors. Comparison with data from the literature indicates that the rat brain autoreceptors can be equated with the α2D subtype as defined by radioligand binding, whereas the rabbit brain autoreceptors conform to the α2A subtype. For example, the antagonist affinities for the rat autoreceptors correlate with their binding affinities for the gene product of α2-RG20, the putative rat α2D-adrenoceptor gene (r = 0.97; P<0.01), but not with their binding affinities for the gene product of α2-C10, the putative human α2A-adrenoceptor gene. Conversely, the rabbit autoreceptors correlate with the α2-C10 (r = 0.98; P<0.001) but not with the α2-RG20 gene product. Since presynaptic α2-autoreceptors are also α2D in rat submaxillary gland and perhaps vas deferens and α2A in rabbit pulmonary artery, the possibility arises that the majority of α2-autoreceptors generally are α2D in the rat and α2A in the rabbit. Moreover, receptors of the α2A/D group generally may be the main mammalian α2-autoreceptors.
Similar content being viewed by others
References
Adler CH, Meller E, Goldstein M (1987) Receptor reserve at the alpha-2 adrenergic receptor in the rat cerebral cortex. J Pharmacol Exp Ther 240:508–515
Agneter E, Drobny H, Singer EA (1991) Estimation of agonist dissociation constants at central presynaptic alpha-2 autoreceptors in the absence of autoinhibition. J Pharmacol Exp Ther 257:19–25
Alabaster VA, Keir RF, Peters CJ (1986) Comparison of potency of α2-adrenoceptor antagonists in vitro: evidence for heterogeneity of α2-adrenoceptors. Br J Pharmacol 88:607–614
Alberts P (1992) Subtype classification of the presynaptic α-adrenoceptors which regulate [3H]-noradrenaline secretion in guinea-pig isolated urethra. Br J Pharmacol 105:142–146
Angus JA, Lew MJ (1984) Phentolamine — an unexpected agonist in the rabbit. Br J Pharmacol 81:423–425
Arribas S, Galvan R, Ferrer M, Herguido MJ, Marin J, Balfagón G (1991) Characterization of the subtype of presynaptic α2-adrenoceptors modulating noradrenaline release in cat and bovine cerebral arteries. J Pharm Pharmacol 43:855–859
Arunlakshana O, Schild HO (1959) Some quantitative uses of drug antagonists. Br J Pharmacol 14:48–58
Blaxall HS, Murphy TJ, Baker JC, Ray C, Bylund DB (1991) Characterization of the alpha-2C adrenergic receptor subtype in the opossum kidney and in the OK cell line. J Pharmacol Exp Ther 259:323–329
Bohmann C, Schollmeyer P, Rump LC (1993) α2-Autoreceptor subclassification in rat isolated kidney by use of short trains of electrical stimulation. Br J Pharmacol 108:262–268
Bylund DB (1992) Subtypes of α1- and α2-adrenergic receptors. FASEB J 6:832–839
Bylund DB, Blaxall HS, Iversen LJ, Caron MG, Lefkowitz RJ, Lomasney JW (1992) Pharmacological characteristics of α2-adrenergic receptors: comparison of pharmacologically defined subtypes with subtypes identified by molecular cloning. Mol Pharmacol 42:1–5
Cheung YD, Barnett DB, Nahorski SR (1982) [3H]Rauwolscine and [3H]yohimbine binding to rat cerebral and human platelet membranes: possible heterogeneity of α2-adrenoceptors. Eur J Pharmacol 84:79–85
Connaughton S, Docherty JR (1990) Functional evidence for heterogeneity of peripheral prejunctional α2-adrenoceptors. Br J Pharmacol 101:285–290
Ennis C (1985) Comparison of the α2-adrenoceptors which modulate noradrenaline release in rabbits and rat occipital cortex. Br J Pharmacol 85:318P
Ennis C, Lattimer N (1984) Presynaptic agonist effect of phentolamine in the rabbit vas deferens and rat cerebral cortex. J Pharm Pharmacol 36:753–757
Furchgott RF (1966) The use of β-haloalkylamines in the differentiation of receptors and in the determination of dissociation constants of receptor-agonist complexes. Adv Drug Res 3:21–55
Furchgott RF (1972) The classification of adrenoceptors (adrenergic receptors). An evaluation from the standpoint of receptor theory. In: Blaschko H, Muscholl E (eds) Catecholamines. Handbook of experimental pharmacology, vol 33, Springer, Berlin, Heidelberg, New York, pp 283–335
Gleason MM, Hieble JP (1991) Ability of SK&F 104078 and SK&F 104856 to identify alpha-2 adrenoceptor subtypes in NCB20 cells and guinea pig lung. J Pharmacol Exp Ther 259:1124–1132
Gobbi M, Frittoli E, Mennini T (1990) The modulation of [3H]noradrenaline and [3H]serotonin release from rat brain synaptosomes is not mediated by the α2B-adrenoceptor subtype. Naunyn-Schmiedeberg's Arch Pharmacol 342:382–386
Harrison JK, D'Angelo DD, Zeng D, Lynch KR (1991) Pharmacological characterization of rat α2-adrenergic receptors. Mol Pharmacol 40:407–412
Heepe P, Starke K (1985) α-Adrenoceptor antagonists and the release of noradrenaline in rabbit cerebral cortex slices: support for the α-autoreceptor hypothesis. Br J Pharmacol 84:147–155
Konno F, Kusunoki M, Takayanagi I (1987) Receptor interactions of a series of imidazolines: comparison of the alpha2-adrenoceptors between the rabbit vas deferens and guinea pig ileum. Jpn J Pharmacol 44:171–178
Lanier SM, Downing S, Duzic E, Homcy CJ (1991) Isolation of rat genomic clones encoding subtypes of the α2-adrenergic receptor. J Biol Chem 266:10470–10478
Lattimer N, Rhodes KF (1985) A difference in the affinity of some selective α2-adrenoceptor antagonists when compared on isolated vasa deferentia of rat and rabbit. Naunyn-Schmiedeberg's Arch Pharmacol 329:278–281
Limberger N, Starke K (1984) Further study of prerequisites for the enhancement by α-adrenoceptor antagonists of the release of noradrenaline. Naunyn-Schmiedeberg's Arch Pharmacol 325:240–246
Limberger N, Mayer A, Zier G, Valenta B, Starke K, Singer EA (1989) Estimation of pA2 values at presynaptic α2-autoreceptors in rabbit and rat brain cortex in the absence of autoinhibition. Naunyn-Schmiedeberg's Arch Pharmacol 340:639–647
Limberger N, Spath L, Starke K (1991) Subclassification of the presynaptic α2-autoreceptors in rabbit brain cortex. Br J Pharmacol 103:1251–1255
Limberger N, Trendelenburg AU, Starke K (1992) Pharmacological characterization of presynaptic α2-autoreceptors in rat submaxillary gland and heart atrium. Br J Pharmacol 107:246–255
Mackay D (1990) Agonist potency and apparent affinity: interpretation using classical and steady-state ternary-complex models. Trends Pharmacol Sci 11:17–22
MacKinnon AC, Kilpatrick AT, Kenny BA, Spedding M, Brown CM (1992) [3H]-RS-15385–197, a selective and high affinity radioligand for α2-adrenoceptors: implications for receptor classification. Br J Pharmacol 106:1011–1018
Maura G, Bonanno G, Raiteri M (1992) Presynaptic α2-adrenoceptors mediating inhibition of noradrenaline and 5-hydroxytryptamine release in rat cerebral cortex: further characterization as different α2-adrenoceptor subtypes. Naunyn-Schmiedeberg's Arch Pharmacol 345:410–416
Michel AD, Loury DN, Whiting RL (1989) Differences between the α2-adrenoceptor in rat submaxillary gland and the α2A- and α2B-adrenoceptor subtypes. Br J Pharmacol 98:890–897
Molderings G, Göthert M (1992) Pharmacological characterization of the inhibitory α2-autoreceptors on the sympathetic nerves of the rabbit pulmonary artery. Naunyn-Schmiedeberg's Arch Pharmacol 346:R64
Motulsky HJ, Ransnas LA (1987) Fitting curves to data using nonlinear regression: a practical and nonmathematical review. FASEB J 1:365–374
Nasseri A, Minneman KP (1987) Relationship between α2-adrenergic receptor binding sites and the functional receptors inhibiting norepinephrine release in rat cerebral cortex. Mol Pharmacol 32:655–662
Parker RB, Wand DR (1971) Pharmacological estimation of drug-receptor dissociation constants. Statistical evaluation. I. Agonists. J Pharmacol Exp Ther 177:1–12
Raiteri M, Bonanno G, Maura G, Pende M, Andrioli GC, Ruelle A (1992) Subclassification of release-regulating α2-autoreceptors in human brain cortex. Br J Pharmacol 107:1146–1151
Reichenbacher D, Reimann W, Starke K (1982) α-Adrenoceptor-mediated inhibition of noradrenaline release in rabbit brain cortex slices. Naunyn-Schmiedeberg's Arch Pharmacol 319:71–77
Ruffolo RR, Nichols AJ, Stadel JM, Hieble JP (1991) Structure and function of α-adrenoceptors. Pharmacol Rev 43:475–505
Schloos J, Wellstein A, Palm D (1987) Agonist binding at alpha2-adrenoceptors of human platelets using 3H-UK-14, 304: regulation by Gpp(NH)p and cations. Naunyn-Schmiedeberg's Arch Pharmacol 336:48–59
Schwartz DD, Malik KU (1992) Characterization of prejunctional alpha-2 adrenergic receptors involved in modulation of adrenergic transmitter release in the isolated perfused rat kidney. J Pharmacol Exp Ther 261:1050–1055
Shen KZ, Barajas-Lopez C, Surprenant A (1990) Functional characterization of neuronal pre and postsynaptic α2-adrenoceptor subtypes in guinea-pig submucosal plexus. Br J Pharmacol 101:925–931
Simonneaux V, Ebadi M, Bylund DB (1991) Identification and characterization of α2D-adrenergic receptors in bovine pineal gland. Mol Pharmacol 40:235–241
Singer EA (1988) Transmitter release from brain slices elicited by single pulses: a powerful method to study presynaptic mechanisms. Trends Pharmacol Sci 9:274–276
Smith K, Docherty JR (1992) Are the prejunctional α2-adrenoceptors of the rat vas deferens and submandibular gland of the α2A- or α2D-subtype? Eur J Pharmacol 219:203–210
Smith K, Connaughton S, Docherty JR (1992) Investigation of prejunctional α2-adrenoceptors in rat atrium, vas deferens and submandibular gland. Eur J Pharmacol 211:251–256
Starke K (1987) Presynaptic α-autoreceptors. Rev Physiol Biochem Pharmacol 107:73–146
Summers RJ, McMartin LR (1993) Adrenoceptors and their second messenger systems. J Neurochem 60:10–23
Taube HD, Starke K, Borowski E (1977) Presynaptic receptor systems on the noradrenergic neurones of rat brain. Naunyn-Schmiedeberg's Arch Pharmacol 299:123–141
Thron CD (1970) Graphical and weighted regression analyses for the determination of agonist dissociation constants. J Pharmacol Exp Ther 175:541–553
Uhlén S, Xia Y, Chhajlani V, Felder CC, Wikberg JES (1992) [3H]-MK 912 binding delineates two α2-adrenoceptor subtypes in rat CNS one of which is identical with the cloned pA2d α2-adrenoceptor. Br J Pharmacol 106:986–995
Waud DR (1976) Analysis of dose-response relationships. In: Narahashi T, Bianchi CP (eds) Advances in general and cellular pharmacology, vol 1, Plenum, New York, London, pp 145–178
Young P, Berge J, Chapman H, Cawthorne MA (1989) Novel α2-adrenoceptor antagonists show selectivity for α2A- and α2B-adrenoceptor subtypes. Eur J Pharmacol 168:381–386
Author information
Authors and Affiliations
Additional information
Correspondence to: N. Limberger at the above address
Rights and permissions
About this article
Cite this article
Trendelenburg, AU., Limberger, N. & Starke, K. Presynaptic α2-autoreceptors in brain cortex: α2D in the rat and α2A in the rabbit. Naunyn-Schmiedeberg's Arch Pharmacol 348, 35–45 (1993). https://doi.org/10.1007/BF00168534
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00168534