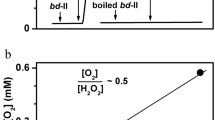
Abstract
After uptake of microbial ferrisiderophores, iron is assumed to be released by reduction. Two ferrisiderophore-reductase activities were identified in Escherichia coli K-12. They differed in cellular location, susceptibility to amytal, and competition between oxygen and ferrichrome-iron(III) reduction. The ferrisiderophore reductase associated with the 40,000×g sediment (membrane-bound enzyme) was inhibited by 10 mM amytal in contrast to the ferrisiderophore reductase present in the 100,000×g supernatant (soluble enzyme). Reduction by the membrane-bound enzyme followed sigmoid kinetics, but was biphasic in the case of the soluble enzyme. The soluble reductase could be assigned to a protein consisting of a single polypeptide of M r 26000. Reduction of iron(III) by the purified enzyme depended on the addition of NADH or NADPH which were equally active reductants. The cofactor FMN and to a lesser degree FAD stimulated the reaction. Substrate specificity of the soluble reductase was low. In addition to the hydroxamate siderophores arthrobactin, schizokinen, fusigen, aerobactin, ferrichrome, ferrioxamine B, coprogen, and ferrichrome A, the iron(III) complexes of synthetic catecholates, dihydroxy benzoic acid, and dicitrate, as well as carrier-free iron(III) were accepted as substrates. Both ferrisiderophore reductases were not controlled by the fur regulatory system and were not suppressed by anaerobic growth.
Similar content being viewed by others
Abbreviations
- DHB:
-
dihydroxybenzoic acid
- MECAM:
-
1,3,5-N,N′,N″-tris-(2,3-dihydroxybenzoyl)-triamino-methylbenzene
- MECAMS:
-
2,3-dihydroxy-5-sulfonyl-derivative of MECAM
References
Arcenaux JEL, Byers BR (1980) Ferrisiderophore reductase activity in Bacillus megaterium. J Bacteriol 141: 715–721
Braun V, Hantke K, Eick-Helmerich K, Köster W, Pressler U, Sauer M, Schäffer S, Schöffler H, Staudenmaier H, Zimmermann L (1987) Iron transport systems in Escherichia coli, In: Winkelmann G, Helm D van der, Neilands JB (eds) Iron transport in microbes, plants, and animals. Verlag Chemie, Weinheim, pp 35–51
Brown KA, Ratledge C (1975) Iron transport in Mycobacterium smegmatis: ferrimycobactin reductase [NAD(P)H: ferrimycobactin oxidoreductase], the enzyme releasing iron from its carrier. FEBS Lett 53: 262–266
Bryce CFA, Crichton RR (1973) The catalytic activity of horse spleen apoferritin. Preliminary kinetic studies and the effect of chemical modification. Biochem J 133: 301–309
Cox CD (1980) Iron reductase in Pseudomonas aeruginosa. J Bacteriol 141: 199–204
Dailey HA, Lacelles J (1977) Reduction of iron and synthesis of protoheme by Spirillum itersonii and other organisms. J Bacteriol 129: 815–820
Eick-Helmerich K, Hantke K, Braun V (1987) Cloning and expression of the exbB gene of Escherichia coli K-12. Mol Gen Genet 206: 246–251
Ernst JF, Winkelmann G (1977) Enzymatic release of iron from sideramines in fungi. NADH: sideramine oxidoreductase in Neurospora crassa. Biochim Biophys Acta 500: 27–41
Fecker L, Braun V (1983) Cloning and expression of the fhu genes involved in iron(III)-hydroxamate uptake by Escherichia coli. J Bacteriol 156: 1301–1314
Gaines CG, Lodge JS, Arcenaux JEL, Byers BR (1981) Ferrisiderophore reductase activity associated with an aromatic biosynthetic enzyme complex in Bacillus subtilis. J Bacteriol 148: 527–533
Garibaldi JA, Neilands JB (1955) Isolation and properties of ferrichrome A. J Am Chem Soc 77: 2429–2430
Hancock REW, Hantke K, Braun V (1977) Iron transport in Escherichia coli K-12. 2,3-Dihydroxy-benzoate-promoted iron uptake. Arch Microbiol 114: 231–239
Hantke K (1981) Regulation of ferric iron transport in Escherichia coli K-12: Isolation of a constitutive mutant. Mol Gen Genet 182: 288–292
Heidinger S, Braun V, Pecoraro VL, Raymond KN (1983). Iron supply to Escherichia coli by synthetic analogs of enterochelin. J Bacteriol 153: 109–115
Jaworowski A, Mayo G, Shaw DC, Campbell HD, Young IG (1981). Characterization of the respiratory NADH dehydrogenase of Escherichia coli and reconstitution of NADH oxidase in ndh mutant membrane vesicles. Biochemistry 20: 3621–3628
Lugtenberg B, Meijers J, Peters R, van der Hoeck P, van Alphen L (1975). Electrophoretic resolution of the major outer membrane protein of Escherichia coli K-12 into four bands. FEBS Lett 58: 254–258
Mann BJ, Holroyd CD, Bradbeer C, Kadner RJ (1986) Reduced activity of TonB-dependent functions in strains of Escherichia coli. FEMS Lett 33: 255–260
Matzanke BF, Müller GI, Bill E, Trautwein AX (1989) Iron metabolism of Escherichia coli studied by Mössbauer spectroscopy and biochemical methods. Eur J Biochem 183: 371–379
Moody MD, Dailey HA (1983) Aerobic ferrisiderophore reductase assay and activity stain for native polyacrylamide gels. Anal Biochem 134: 235–239
Morrissey JH (1981) Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem 117: 307–310
Neilands JB (1981) Microbial iron compounds. Ann Rev Biochem 50: 715–731
Raymond KN, Müller GN, Matzanke BF (1984) Complexation of iron by siderophores. A review of their solution and structural chemistry and biological function. In: FL Boschke (ed) Topics in current chemistry, vol 123. Springer, Berlin Heidelberg New York, pp 49–102
Stookey LL (1970) Ferrozine: a new spectrophotometric reagent for iron. Anal Chem 42: 779–781
Tait GH (1975) The identification and biosynthesis of siderochromes formed by Micrococcus denitrificans. Biochem J 146: 191–204
Udenfried S, Stein S, Böhlen P, Dairman W, Leimgruber W, Weigele M (1972) Fluorescamin: a reagent for assay of amino acids, peptides, proteins, and primary amines in the picomole range. Science 178: 871–872
Williams HD, Poole RK (1987) Reduction of iron(III) by Escherichia coli K12: Lack of involvement of the respiratory chains. Curr Microbiol 15: 319–324
Young IG, Jaworowski A, Poulis MI (1978) Amplification of the respiratory NADH dehydrogenase of Escherichia coli by gene cloning. Biochim Biophys Acta 449: 376–385
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fischer, E., Strehlow, B., Hartz, D. et al. Soluble and membrane-bound ferrisiderophore reductases of Escherichia coli K-12. Arch. Microbiol. 153, 329–336 (1990). https://doi.org/10.1007/BF00249001
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00249001