Summary
Blood ciclosporin (Cs) metabolite pattern in 58 liver grafted patients was routinely monitored by HPLC from the first Cs dose after transplantation until discharge from hospital. Eighteen patients with normal kidney function were allocated to Group I and 14 patients in Group II suffered Cs nephrotoxicity during their clinical course.
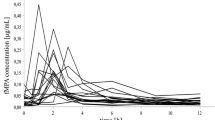
There were no significant differences between both groups in blood Cs level, kidney function before transplantation, liver function or co-administration of other potentially nephrotoxic drugs. A correlation matrix involving both groups showed a significant correlation between the blood concentration of metabolite M1c9 and serum creatinine and urea, and an inverse correlation with creatinine clearance. During a nephrotoxic episode the blood concentrations of metabolites M1c9 and M1A were significantly elevated in patients in Group II. Analysis of the time course revealed significantly higher blood levels of M19 and M1c9 in Group II patients compared with those in Group I for the first 10 days after transplantation. Serum creatinine and urea concentrations remained significantly elevated, the creatinine clearance being significantly reduced throughout the period of observation.
The elevated blood concentrations of ciclosporin metabolites M1c9 and M19 during nephrotoxic episodes suggest that these metabolites are associated with ciclosporin nephrotoxicity. It could not be decided if the elevated metabolite concentrations were the result of and/or the reason for impaired kidney function.
Similar content being viewed by others
References
Wallemacq PE, Lhoëst G, Dumont P (1989) Isolation, purification and structure elucidation of cyclosporine metabolites in rabbit and man. Biomed Environ Mass Spectrom 18: 48–56
Maurer G, Loosli R, Schreier E, Keller H (1984) Disposition of cyclosporine in several animal species and man-structural elucidation of its metabolites. Drug Metab Dispos 12: 120–126
Maurer G, Lemaire M (1986) Biotransformation of cyclosporine and distribution in blood. Transplant Proc 18: 25–34
Hartmann NR, Jardine I (1986) Mass spectrometric analysis of cyclosporine metabolites. Biomed Environ Mass Spectrom 13: 361–372
Kunzendorf U, Brockmöller J, Joachimsen F, Keller F, Walz G, Offermann G (1988) Cyclosporine metabolites and central-nervous-system toxicity Lancet I: 1223
Davenport A, Will EJ, Davison AM, Ironside JW (1988) Toxicity of cyclosporine metabolites. Lancet II: 333
Rosano TG, Pell MA, Freed BM, Dybas MT, Lempert N (1988) Cyclosporine and metabolites in blood from renal allograft recipients with nephrotoxicity, rejection, or good renal function: comparative high-performance liquid chromatography and monoclonal radioimmunoassay studies. Transplant Proc 20: 330–338
Leunissen K, Baumann G, Bosman R, Van Hoof JP (1986) Cyclosporin metabolites and nephrotoxicity. Lancet II: 1398
Ryffel B, Foxwell BMJ, Mihatsch MJ, Donatsch P, Maurer G (1988) Biological significance of cyclosporine metabolites. Transplant Proc 20: 575–584
Donatsch P, Rickenberger U, Ryffel B, Brouillard JF, Tschopp M, Wullschleger J (1990) Adverse reaction profiles of Sandimmune (CsA) metabolites. Transplant Proc 22 (3): 1137–1140
Christians U, Sewing KF (1989) Whole blood sample clean-up for chromatographic analysis. In: Zech K, Frei RW (eds) Selective sample handling and detection in high-performance liquid chromatography, Part B. Elsevier, Amsterdam, pp 82–132
Wonigeit K, Kohlhaw K, Winkler M, Schaefer O, Pichlmayr R (1990) Cyclosporine monitoring in liver allograft recipients. Two distinct patterns of blood level derangement associated with nephrotoxicity. Transplant Proc 22 (3): 1305–1311
Christians U, Zimmer KO, Wonigeit K, Sewing KF (1987) Measurement of cyclosporin A and four of its metabolites in whole blood by high-performance liquid chromatography. J Chromatogr 413: 121–129
Christians U, Zimmer KO, Wonigeit K, Maurer G, Sewing KF (1988) Liquid chromatographic measurement of cyclosporin A and its metabolites in blood, bile, and urine. Clin Chem 34: 34–39
Sewing KF, Christians U, Bleck JS, Schottmann R, Strohmeyer SS (1988) Measurement and disposition of cyclosporine and its metabolites. Bibliotheca Cardiol 43: 63–72
Kahan BD (1990) Consensus report of the ciclosporin expert meeting, Hawk's Cay, 1990. Transplant Proc 22 (3): 1357–1361
Wadhwa NK, Schroeder TJ, Amadeo JP, Myre SA, Clardy SW, First MR (1987) Cyclosporine drug interactions: a review. Ther Drug Monit 9: 399–406
Cockburn ITR, Krupp P (1989) An appraisal of drug interactions with SandimmunR. Transplant Proc 21: 3845–3850
Racusen LC, Solez K (1988) Cyclosporine nephrotoxicity. Int Rev Exp Pathol 30: 107–157
Mason J (1989) Pharmacology of cyclosporine (Sandimmune) VII. Pathophysiology and toxicology of cyclosporine in humans and animals. Pharmacol Rev 42: 423–434
Kahan BD (1989) Cyclosporine. N Engl J Med 321: 1725–1738
Cole E, Cheung F, Wong PY, Fung LS, Skorecki K, Levy GA (1989) Toxic effects on renal cells in culture- a comparison of cyclosporin A and its metabolites. Transplant Proc 21: 943–945
Bouis P, Brouillard JF, Fischer V, Donatsch P, Boelsterli UA (1990) Effect of enzyme induction on Sandimmun (Cyclosporin A) biotransformation and hepatotoxicity in cultured rat hepatocytes and in vivo. Biochem Pharmacol 39: 257–266
Bowers LD (1990) Studies of cyclosporine and metabolite toxicity in renal and hepatocyte culture systems. Transplant Proc 22: 1135–1136
Bleck JS, Schlitt HJ, Christians U, Thiesemann C, Strohmeyer S, Schottmann R, Kohlhaw K, Wonigeit K, Sewing KF (1989) Urinary excretion of ciclosporin and 17 of its metabolites in renal allograft recipients. Pharmacol 39: 160–164
Mihatsch MJ, Steiner K, Abeywickrama KH, Landmann J, Thiel G (1988) Risk factors for the development of chronic cyclosporine-nephrotoxicity. Clin Nephrol 29: 165–175
Lucey MR, Kolars JC, Merion RM, Campbell DA, Aldrich M, Watkins PB (1990) Cyclosporin toxicity at therapeutic blood levels and cytochrome P-450 IIIA. Lancet 335: 11–15
Kohlhaw K, Wonigeit K, Schäfer O, Ringe B, Bunzendahl H, Pichlmayr R (1989) Association of very high blood levels of cyclosporine metabolites with clinical complications after liver transplantation. Transplant Proc 21: 2232–2233
Nagelkerke FJ, Tijdens RB, Schwarz EP, Winters MFG, Paul LC, Mulder GJ (1987) The covalent binding of cyclosporin A to rat liver macromolecules in vivo and in vitro: the role of cytochrome P450. Toxicology 40: 277–284
Christians U, Schlitt HJ, Bleck JS, Kownatzki R, Schiebel HM, Maurer G, Strohmeyer SS, Schottmann R, Wonigeit K, Pichlmayr R, Sewing KF (1988) Measurement of cyclosporine and 18 metabolites in blood, bile and urine by high performance liquid chromatography. Transplant Proc 20: 609–613
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Christians, U., Kohlhaw, K., Budniak, J. et al. Ciclosporin metabolite pattern in blood and urine of liver graft recipients. Eur J Clin Pharmacol 41, 285–290 (1991). https://doi.org/10.1007/BF00314953
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00314953