Abstract
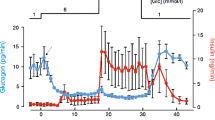
ATP-sensitive K-channels in the cloned β-cell line HIT T15 were studied by patch-clamp methods; by measurement of 86Rb efflux; and by [3H]glibenclamide binding to isolated membrane preparations. In inside-out patches a 50 pS K-channel was found which was blocked by ATP or tolbutamide applied to the intracellular membrane surface. A minimum estimate of about 500 channels per β-cell was obtained by combining whole-cell and single-channel data. The rate of efflux of 86Rb from 86RbCl-loaded HIT cells was markedly increased by intracellular ATP-depletion; 86Rb-efflux was progressively inhibited by increasing concentrations of glibenclamide or tolbutamide. In non-ATP-depleted cells, diazoxide elicited a concentration-dependent stimulation of 86Rb-efflux which was completely blocked by 1 μM glibenclamide. Isolated membranes showed dose-dependent saturable binding of [3H]glibenclamide to both high (K d=1.12 nM) and low (K d=136 nM) affinity binding sites. We estimate about 5000 high-affinity binding sites per cell. [3H]-glibenclamide binding was inhibited by tolbutamide (IC50=125 μM) but was not affected by diazoxide. ADP (0.5 or 1.0 mM) markedly reduced binding; other nucleotides tested were ineffective.
Similar content being viewed by others
References
Ashcroft FM (1988) Adenosine 5-triphosphate-sensitive potassium channels. Annu Rev Neurosci 11:97–118
Ashcroft SJH, Stubbs M (1987) The glucose sensor in HIT cells is the glucose transporter. FEBS Lett 219:311–315
Ashcroft FM, Harrison DE, Ashcroft SJH (1984) Glucose induces closure of single potassium channels in isolated rat pancreatic β-cells. Nature 312:446–448
Ashcroft SJH, Hammonds P, Harrison DE (1986) Insulin secretory responses of a clonal cell line of Syrian virus 40-transformed B-cells. Diabetologia 29:727–733
Ashcroft FM, Ashcroft SJH, Harrison DE (1988a) Properties of single potassium channels modulated by glucose in rat pancreatic β-cells. J Physiol 400:501–527
Ashcroft SJH, Hughes SJ, Kerr AJ (1988b) Detection of the sulphonylurea-binding component of the ATP-sensitive K-channel in HIT T15 B-cells by electrophoresis on nondenaturing gels. Diabetologia 31:466A
Ashcroft FM, Kakei M, Kelly RP (1989) Rubidium and sodium permeability of the ATP-sensitive K+ channel in single rat pancreatic β-cells. J Physiol 408:413–430
Bernadi H, Fosset M, Lazdunski M (1988) Characterization, purification, and affinity labelling of the brain [3H] glibenclamidebinding protein, a putative neuronal ATP-regulated K+ channel. Proc Natl Acad Sci USA 85:9816–9820
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brass LF, Shattil SJ (1989) Interaction of extracellular Ca with the surface membrane of human platelets. Methods Enzymol 169:355–371
Campbell KP, Leung AT, Sharp AH (1988) The biochemistry and molecular biology of the dihydropyridine-sensitive calcium channel. TINS 11:425–430
Catterall W (1982) The emerging molecular view of the sodium channel. TINS 5:303–306
Cook DL, Hales CN (1984) Intracellular ATP directly blocks K-channels in pancreatic B-cells. Nature 311:271–273
Cook DL, Satin LS, Ashford MJL, Hales N (1988) ATP-sensitive K-channels in pancreatic β-cells. Spare channel hypothesis. Diabetes 37:495–498
Dunne MJ, Petersen OH (1986) Intracellular ADP activates K+ channels that are inhibited by ATP in an insulin-secreting cell line. FEBS Letts 208:59–63
Dunne MJ, Ilott MC, Petersen OH (1987) Interaction of diazoxide, tolbutamide and ATP4− on nucleotide-dependent K+ channels in an insulin-secreting cell line. J Membr Biol 99:215–224
Eddlestone GT, Ribalet B, Ciani C (1989) Differences in K channel behaviour in glucose-responsive and glucose-unresponsive insulin-secreting tumor cell lines. Biophys J 55:541a
Findlay I (1987) The effects of magnesium ions upon adenosine triphosphate sensitive potassium channels in a rat insulin-secreting cell line. J Physiol 391:611–629
Findlay I, Dunne MJ (1986) ATP maintains ATP-inhibited K+ channels in an operational state. Pflügers Arch 407:238–240
Findlay I, Dunne MJ, Petersen OH (1985) High conductance K+ channel in pancreatic islet cells can be activated and inactivated by internal calcium. J Membr Biol 83:169–175
Fosset M, De Weille JR, Green RD, Schmid-Antomarchi H, Lazdunski M (1988) Antidiabetic sulphonylureas control action potential properties in heart cells via high affinity receptors that are linked to ATP-dependent K-channels. J Biol Chem 263:7933–7936
Gaines KL, Hamilton S, Boyd III AE (1988) Characterization of the sulfonylurea receptor on beta cell membranes. J Biol Chem 263:2589–2592
Gylfe E, Hellman B, Sehlin J, Täljedal I-B (1984) Interaction of sulfonylurea with the pancreatic β-cell. Experientia 40:1126–1134
Hamill OP, Marty A, Neher E, Sakmann B, Sigworth FJ (1981) Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflügers Arch 391:85–100
Henquin JC (1988) ATP-sensitive K+ channels may control glucose-induced electrical activity in pancreatic B-cells. Biochem Biophys Res Comm 156:769–775
Henquin JC, Charles S, Nenquin M, Mathot F, Tamagawa T (1982) Diazoxide and D600 inhibition of insulin release. Distinct mechanisms explain the specificity for different stimuli. Diabetes 31:776–784
Isenberg C, Schröter K, Trube G (1988) Kinetics of single ATP-dependent K-channels in the membrane of murine pancreatic β-cells. Pflügers Arch 411: R111
Kakei M, Kelly RP, Ashcroft SJH, Ashcroft FM (1986) The ATP-sensitivity of K+ channels in rat pancreatic B-cells is modulated by ADP. FEBS Lett 208:63–66
Kelly R, Ashcroft FM (1988) ATP-sensitive K-channels in HIT-T15 insulin-secreting cells. Diabetologia 31:507A
Kramer W, Oekonomopoulos R, Punter J, Summ H-D (1988) Direct photoaffinity labelling of the putative sulphonylurea receptor in rat β-cell tumor membranes by [3H]glibenclamide. FEBS Lett 229:355–359
Lupo B, Bataille D (1987) A binding site for [3H]glipizide in the rat cerebral cortex. Eur J Pharmacol 140:157–169
Martell AE, Smith RM (1974) Critical stability constants, vol 1, Amino acids, vol 2, Amines. Plenum Press, New York
Misler DS, Falke LC, Gillis K, McDaniel ML (1986) A metabolite regulated potassium channel in rat pancreatic β-cells. Proc Natl Acad Sci USA 83:7119–7123
Ohno-Shosaku T, Zünkler BJ, Trube G (1987) Dual effects of ATP on K+ currents in mouse pancreatic B-cells. Pflügers Arch 408:133–138
Petersen OH, Findlay I (1987) Electrophysiology of the pancreas. Physiol Rev 67:1054–1116
Pruitt AW, Dayton PG, Patterson JH (1973) Disposition of diazoxide in children. Clin Pharmacol Ther 14:73–82
Rorsman P, Trube G (1985) Glucose-dependent K+-channels in pancreatic β-cells are regulated by intracellular ATP. Pflügers Arch 405:305–309
Rorsman P, Trube G (1986) Calcium and delayed potassium currents in mouse pancreatic beta-cells under voltage-clamp conditions. J Physiol 374:531–550
Santerre RF, Cook RA, Crisel RMD, Sharp JD, Schmidt RJ, Williams DC, Wilson CP (1981) Insulin synthesis in a clonal cell line of SV40 transformed hamster B cells. Proc Natl Acad Sci USA 78:4339–4343
Schmid-Antomarchi H, De Weille J, Fosset M, Lazdunski M (1987) The receptor for antidiabetic sulfonylureas controls the activity of the ATP-modulated K+ channel in insulin secreting cells. J Biol Chem 262:15840–15844
Schwarz LM, McClesky EW, Almers W (1985) Dihydropyridine receptors in muscle are voltage-dependent but most are not functional calcium channels. Nature 314:747–750
Sturgess NC, Kozlowski RZ, Carrington CA, Hales CN, Ashford MLJ (1988) Effects of sulphonylureas and diazoxide on insulin secretion and nucleotide-sensitive channels in an insulin-secreting cell line. Br J Pharmacol 95:83–94
Trube G, Rorsman P, Ohno-Shosaku T (1986) Opposite effects of tolbutamide and diazoxide on the ATP-dependent K+ channel in mouse pancreatic β-cells. Pflügers Arch 407:493–499
Trube G, Heschler J, Schroter K (1989) Regulation and function of ATP-dependent channels in pancreatic β-cells. J Gen Physiol (in press)
Zünkler BJ, Lenzen S, Manner K, Panten U, Trube G (1988a) Concentration-dependent effects of tolbutamide, meglitinide, glipizide, glibenclamide and diazoxide on ATP-regulated K currents in pancreatic B-cells. Naunyn-Schmiedeberg's Arch Pharmacol 337:225–230
Zünkler BJ, Lins S, Ohno-Shosaku T, Trube G, Panten U (1988b) Cytosolic ADP enhances the sensitivity to tolbutamide of ATP-dependent K+ channels from pancreatic B-cells. FEBS Lett 239:241–244
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Niki, I., Kelly, R.P., Ashcroft, S.J.H. et al. ATP-sensitive K-channels in HIT T15 β-cells studied by patch-clamp methods, 86Rb efflux and glibenclamide binding. Pflugers Arch. 415, 47–55 (1989). https://doi.org/10.1007/BF00373140
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00373140