Abstract
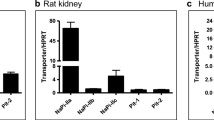
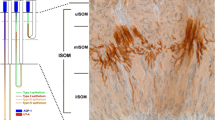
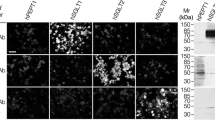
Polyclonal antibodies have been raised against a C-terminal peptide of NaPi-1, a recently cloned Na-Pi cotransport system of rabbit kidney cortex with a predicted (unglycosylated) molecular mass of 52 kDa. By Western blot analysis using brush-border membranes isolated from rabbit kidney cortex, two proteins with apparent molecular masses of 64 kDa and 35 kDa were specifically recognized (peptide protectable) by the antiserum obtained. The 64-kDa protein was found to migrate in parallel with the luminal membrane during separation by free-flow electrophoresis of brush-border and basolateral membranes. In immunofluorescence studies using cryostat sections of rabbit kidney, specific binding of antibodies was observed in proximal tubules (including S1, S2 and S3 segments) of superficial and deep nephrons. Anti-(NaPi-1)-antibody-mediated fluorescence was restricted to the brush border of proximal tubular cells. No specific immunoreaction was observed in other tubular segments. The results suggest that the native NaPi-1-related protein (Na-Pi cotransport system) has an apparent molecular mass of 64 kDa and is uniformly expressed in the apical membrane of proximal tubules of all nephron generations in the rabbit kidney. Immunohistochemical localization of the Na-Pi cotransport system NaPi-1 confirms the segmental localization within the nephron of NaPi-1-related mRNA as revealed by the reverse transcriptase/polymerase chain reaction (see preceding paper).
Similar content being viewed by others
References
Barany G, Merrifield RB (1980) Solid-phase peptide synthesis. In: Gross E, Meienhofer J (eds) The peptides, vol. 2. Academic Press, New York, pp 1–284
Berndt TJ, Knox FG (1992) Renal regulation of phosphate excretion. In: Seidin GW, Giebisch G (eds) The kidney: physiology and pathophysiology. Raven Press, New York, pp 2511–2532
Biber J, Stieger B, Haase B, Murer H (1981) A high yield preparation of rat kidney brush border membranes. Different behaviour of lysosomal markers. Biochim Biophys Acta 647:169–176
Calderhead DM, Kitagawa K, Tanner L, Holman GD, Lienhard GE (1990) Insulin regulation of the two glucose transporters in 3T3-L1 adipocytes. J Biol Chem 265:13 800–13 808
Custer M, Meier F, Schlatter E, Greger R, Garcia-Perez A, Biber J, Murer H (1993) Localization of NaPi-1, a putative Na-Pi cotransporter, in rabbit kidney proximal tubules. I. mRNA localization by RT-PCR. Pflügers Arch 424:203–209
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 227:680–685
Mizgala CL, Quamme GA (1985) Renal handling of phosphate. Physiol Rev 65:431–466
Murer H, Werner A, Reshkin S, Wuarin F, Biber J (1991) Cellular mechanisms in proximal tubular reabsorption of inorganic phosphate. Am J Physiol 260:C 885-C 899
Scalera V, Huang Y-K, Hildmann B, Murer H (1981) A simple isolation method for basolateral plasma membranes from rat kidney cortex. Membr Biochem 4:49–61
Stieger B, Murer H (1983) Heterogeneity of brush-border membrane vesicles from rat small intestine prepared by a precipitation method using Mg/EGTA. Eur J Biochem 135:95–101
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Werner A, Moore ML, Mantei N, Biber J, Semenza G, Murer H (1991) Cloning and expression of cDNA for a Na/Pi-cotransport system of kidney cortex. Proc Natl Acad Sci USA 88:9608–9612
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Biber, J., Custer, M., Werner, A. et al. Localization of NaPi-1, a Na/Pi cotransporter, in rabbit kidney proximal tubules. Pflügers Arch. 424, 210–215 (1993). https://doi.org/10.1007/BF00384344
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00384344