Abstract
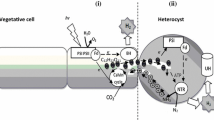
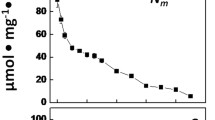
A method is described for the preparation of cyanobacterial heterocysts with high nitrogen-fixation (acetylene-reduction) activity supported by endogenous reductants. The starting material was Anabaena variabilis ATCC 29413 grown in the light in the presence of fructose. Heterocysts produced from such cyanobacteria were more active than those from photoautotrophically-grown A. variabilis, presumably because higher reserves of carbohydrate were stored within the heterocysts. It proved important to avoid subjecting the cyanobacteria to low temperatures under aerobic conditions, as inhibition of respiration appeared to lead to inactivation of nitrogenase. Low temperatures were not harmful in the absence of O2. A number of potential osmoregulators at various concentrations were tested for use in heterocyst isolation. The optimal concentration (0.2M sucrose) proved to be a compromise between adequate osmotic protection for isolated heterocysts and avoidance of inhibition of nitrogenase by high osmotic strength. Isolated heterocysts without added reductants such as H2 had about half the nitrogen-fixation activity expected on the basis of intact filaments. H2 did not increase the rate of acetylene reduction, suggesting that the supply of reductant from heterocyst metabolism did not limit nitrogen fixation under these conditions. Such heterocysts had linear rates of acetylene reduction for at least 2 h, and retained their full potential for at least 12 h when stored at 0°C under N2.
Similar content being viewed by others
References
Blumwald E, Tel-Or E (1982) Osmoregulation and cell composition in salt-adaptation of Nostoc muscorum. Arch Microbiol 132:168–172
Burris RH (1974) Methodology. In: Quispel A (ed) The biology of nitrogen fixation. North-Holland Publishing Company, Amsterdam, pp 9–33
Dubois JD, Kapustka LA (1981) Osmotic stress effects on the N2(C2H2)ase activity of aquatic cyanobacteria. Aquat Bot 11:11–20
Eisbrenner G, Bothe H (1979) Modes of electron transfer from molecular hydrogen in Anabaena cylindrica. Arch Microbiol 123:37–45
Fay P (1980) Heterocyst isolation. Methods Enzymol 69:801–812
Fogg GE (1949) Growth and heterocyst production in Anabaena cylindrica Lemm. II. In relation to carbon and nitrogen metabolism. Ann Bot 13:241–259
Giddings TH, Wolk CP (1981) Factors influencing the stability of nitrogenase activity in isolated cyanobacterial heterocysts. FEMS Microbiol Lett 10:299–302
Goa J (1953) A micro biuret method for protein determination. Scand J Clin Invest 5:218–222
Kumar A, Tabita FA, van Baalen C (1982) Isolation and characterization of heterocysts from Anabaena sp strain CA. Arch Microbiol 133:103–109
Kumar A, Tabita FR, van Baalen C (1983) High endogenous nitrogenase activity in isolated heterocysts of Anabaena sp strain CA after nitrogen starvation. J Bact 155:493–497
Li JL, Burris RH (1983) Influence of pN2 and pD2 on HD formation by various nitrogenases. Biochemistry 22:4472–4480
MacKinney G (1941) Absorption of light by chlorophyll solutions. J Biol Chem 140:315–322
Peterson RB, Burris RH (1978) Hydrogen metabolism in isolated heterocysts of Anabaena 7120. Arch Microbiol 116:125–132
Peterson RB, Wolk CP (1978) High recovery of nitrogenase activity and of 55Fe-labeled nitrogenase in heterocysts isolated from Anabaena variabilis. Proc Natl Acad Sci USA 75:6271–6275
Privalle LS, Burris RH (1984) d-Erythrose supports nitrogenase activity in isolated Anabaena sp strain 7120 heterocysts. J Bact 157:350–356
Reed RH, Richardson DL, Warr SRC, Stewart WDP (1984) Carbohydrate accumulation and osmotic stress in cyanobacteria. J Gen Microbiol 130:1–4
Rippka R, Deruelles J, Waterbury JB, Herdman M, Stainer RY (1979) Generic assignments, strain histories and properties of pure cultures of cyanobacteria. J Gen Microbiol 111:1–61
Smith GD, Mackay EJ, Daday A (1983) Buffer and samage to the filamentous cyanobacterium Anabaena cylindrica. J Gen Microbiol 129:3099–3102
Stacey G, van Baalen C, Tabita FR (1977) Isolation and characterization of a marine Anabaena sp capable of rapid growth on molecular nitrogen. Arch Microbiol 114:197–201
Tel-Or E, Stewart WDP (1977) Photosynthetic components and activities of nitrogen-fixing isolated heterocysts of Anabaena cylindrica. Proc R Soc Lond Ser B 198:61–86
Winkenbach F, Wolk CP (1973) Activities of enzymes of the reductive and oxidative pentose phosphate pathway in heterocysts of a blue-green alga. Plant Physiol 52:480–493
Wolk CP (1968) Movement of carbon from vegetative cells to heterocysts in Anabaena cylindrica. J Bact 96:2138–2143
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jensen, B.B., Cox, R.P. & Burris, R.H. Isolation of cyanobacterial heterocysts with high and sustained dinitrogen-fixation capacity supported by endogenous reductants. Arch. Microbiol. 145, 241–247 (1986). https://doi.org/10.1007/BF00443652
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00443652