Abstract
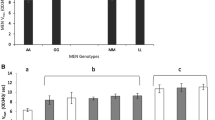
Among the progeny of Drosophila flies heterozygous for two noncomplementing Adh-negative alleles, two individuals were found that had recovered appreciable alcohol dehydrogenase activity, thereby surviving the ethanol medium used as a screen. The most likely explanation is that these Adh-positive flies are the product of intracistronic recombination within the Adh locus. Judging by the distribution of outside markers, one of the crossovers would have been a conventional reciprocal exchange while the other appears to have been an instance of nonreciprocal recombination. The enzymes produced in strains derived from the original survivors can be easily distinguished from wild-type enzymes ADH-S and ADH-F on the basis of their sensitivity to denaturing agents. None of various physical and catalytic properties tested revealed differences between the enzymes of the survivor strains except that in one of them the level of activity is 55–65% of the other. Quantitative immunological determinations of ADH gave estimates of enzyme protein which are proportional to the measured activity levels. These results are interpreted to indicate that different amounts of ADH protein are being accumulated in the two strains.
Similar content being viewed by others
References
Abraham, I., and Doane, W. W. (1976). Temporal gene controlling midgut amylase activity patterns in Drosophila melanogaster. Genetics 83:S1.
Chovnick, A., Ballantyne, G., and Holm, D. (1971). Studies on gene conversion and its relationship to linked exchange in Drosophila melanogaster. Genetics 69179.
Chovnick, A., Gelbart, W., McCarron, M., Osmond, B., Candido, E. P. M., and Baillie, D. L. (1976). Organization of the rosy locus in Drosophila melangaster: Evidence for a control element adjacent to the xanthine dehydrogenase structural element. Genetics 84233.
Cleland, W. W. (1967). The statistical analysis of enzyme kinetic data. Adv. Enzymol. 291.
Davis, B. J. (1964). Disc gel electrophoresis. II. Method and application to human serum proteins. Ann. N. Y. Acad. Sci. 121404.
Dickinson, W. J. (1975). A genetic locus affecting the developmental expression of an enzyme in Drosophila melanogaster. Dev. Biol. 42131.
Elgin, S. C. R. (1975). Methods for isolation and characterization of nonhistone chromosomal proteins. Methods Enzymol. 40144.
Greenleaf, A., and Bautz, E. (1975). RNA polymerase B from Drosophila melanogaster larvae: Purification and partial characterization. Eur. J. Biochem. 60169.
Grell, E. H., Jacobson, K. B., and Murphy, J. B. (1968). Alterations of genetic material for analysis of alcohol dehydrogenase isozymes of Drosophila melanogaster. Ann. N. Y. Acad. Sci. 151441.
Lindsley, D. L., and Grell, E. H. (1968). Genetic Variations of Drosophila melanogaster, Carnegie Inst. Wash. Publ. N627, Washington, D.C.
Lowry, O. H., Rosebrough, N. J., Farr, A. C., and Randell, R. J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. chem. 193265.
Maroni, G. (1977). Genetic control of ADH levels in Drosophila melanogaster. Genetics (Suppl.) 86:s41.
O'Donnell, J., Gerace, L., Leister, F., and Sofer, W. (1975). Chemical selection of mutants that affect alcohol dehydrogenase in Drosophila. II. Use of 1-pentyne-3-ol. Genetics 7973.
Ouchterlony, E. (1970). Handbook of Immunodiffusion and Immunoelectrophoresis Ann Arbor Science Publishers, Ann Arbor, Mich.
Schwartz, M., and Sofer, W. (1976). Alcohol dehydrogenase-negative mutants in Drosophila: Defects at the structural locus. Genetics 83125.
Schwartz, M., Gerace, L., O'Donnell, J., and Sofer, W. (1975). Drosophila alcohol dehydrogenase: Origin of the multiple forms. In Markert, C. L. (ed.), Isozymes I: Molecular Structure Academic Press, New York, 725 pp.
Sofer, W., and Hatkoff, M. A. (1972). Chemical selection of alcohol dehydrogenase negative mutants in Drosophila. Genetics 72545.
Ursprung, H., Smith, K. E., Sofer, W. H., and Sullivan, D. T. (1968). Assay systems for the study of gene function. Science 1601075.
Vigue, C., and Sofer, W. (1974). Adh n5: A temperature-sensitive mutant at the Adh locus in Drosophila. Biochem. Genet. 11387.
Vigue, C., and Sofer, W. (1976). Chemical selection of mutants that affect ADH activity in Drosophila. III. Effects of ethanol. Biochem. Genet. 14127.
Author information
Authors and Affiliations
Additional information
This work was supported in part by NSF Grant PCM 76-19563.
Rights and permissions
About this article
Cite this article
Maroni, G. Genetic control of alcohol dehydrogenase levels in Drosophila . Biochem Genet 16, 509–523 (1978). https://doi.org/10.1007/BF00484215
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00484215