Summary
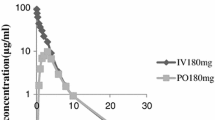
The pharmacokinetics of cefoperazone after i.p. and/or i.v. administration were studied in 12 CAPD patients. After i.v. injection, the plasma half-life was 2.65±0.4 h, the total clearance amounting to 70.1±19.2 ml/min. Peritoneal clearance was calculated to be 6.9±1 ml/min. After peritoneal instillation, the bioavailability was 63.9±5%. After repeated i.p. administration, no accumulation of the drug in the body was observed. Thus, cefoperazone can be safely administered for the treatment of peritonitis in CAPD patients.
Similar content being viewed by others
References
Benet LZ, Galeazzi RL (1979) Non compartmental determination of the steady-state volume of distribution. J Pharm Sci 68: 1071–1074
Dayer P, Balant L, Fabre J, Rudhardt M, Allaz A-F (1981) Renal and extrarenal clearances of cefoperazone in patients with renal failure. Int J Clin Pharm Res 1: 233–244
Fabre J, Blanchard P, Rudhardt M (1977) Pharmacokinetics of ampicillin, cephalothin and doxycycline in various tissues of the rat. Chemotherapy 23: 129–141
Hodler J, Frey B, Rudhardt M, Seiler AJ (1983) Treatment of peritonitis in patients undergoing chronic ambulatory peritoneal dialysis (CAPD) by cefoperazone: clinical and pathophysiological aspects. Spitzy KH, Karrer K (eds) Proc. 13th International Congress of Chemotherapy. Egermann, Vienna, 1983
Keller E, Jansen A, Pelz K, Schollmeyer P, Hoppe-Seyler G (1983) Plasma levels of cefoperazone after intraperitoneal application. Clin Pharmacol Ther 33: 235
Wideröe T-E, Smeby LC, Berg KJ, Jörstad S, Svartas TM (1983) Intraperitoneal (125I) insulin absorption during intermittent and continuous peritoneal dialysis. Kidney Int 23: 22–28
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hodler, J.E., Galeazzi, R.L., Frey, B. et al. Pharmacokinetics of cefoperazone in patients undergoing chronic ambulatory peritoneal dialysis: Clinical and pathophysiological implications. Eur J Clin Pharmacol 26, 609–612 (1984). https://doi.org/10.1007/BF00543494
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00543494