Abstract
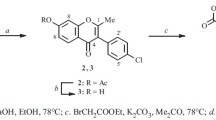
Conditions have been developed for an effective synthesis of 7-hydroxy- and 5,7-dihydroxyisoflavones and also of 3-aryloxy-7-hydroxychromones. The disadvantages and advantages of alternative pathways for the synthesis of these compounds are discussed. The proposed method of synthesis permits considerable simplification of the preparation of compounds of these series.
Similar content being viewed by others
Literature cited
A. L. Kazakov, V. P. Khilya, V. V. Mezheritskii, and Y. Litkei, Natural and Modified Isoflavonoids [in Russian], Izd. RGU, Rostov-on-Don (1985).
T. Namba, M. Hattori, et al., Phytochemistry,22, No. 4, 1057 (1983).
H. Takeno, M. Hashimoto, et al., J. Chem. Soc. Chem. Commun., 474 (1981).
P. E. Spoerri and A. S. Dubois, in: Organic Reactions, R. Adams (ed.), Vol. V (1949), pp. 387–412.
B. G. Pivovarenko, V. P. Khilya, and F. S. Babichev. Dokl. Akad. Nauk UkrSSR, Ser. B, No. 4, 59 (1985).
B. G. Pivovarenko and V. P. Khilya, Dokl. Akad. Nauk UkrSSR, Ser. B, No. 7, 44 (1985).
V. Szabo and A. Kiss, Magy. Kem. Foly.,85, No. 8, 353 (1979).
V. Szabo, J. Borbely, and J. Borda, in: Proceedings of an International Bioflavonoid Symposium, Munich, FRG (1981), p. 19.
V. R. Sethe and K. Venkataraman, Curr. Sci.,18, 373 (1949).
German Patent (Offenschrift) 2,640,617 (1977).
R. J. Bass, J. Chem. Soc., Chem. Commun., 78 (1976).
Additional information
T. G. Shevchenko Kiev State University. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 639–643, September–October, 1989.
Rights and permissions
About this article
Cite this article
Pivovarenko, V.G., Khilya, V.P. & Vasil'ev, S.A. Simple and effective synthesis of isoflavones and 3-aryloxychromones. Chem Nat Compd 25, 542–545 (1989). https://doi.org/10.1007/BF00598071
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00598071