Conclusions
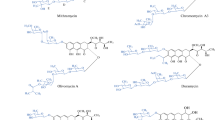
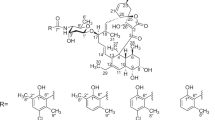
It has been shown that olivomose, which forms part of the carbohydrate moiety of olivomycins A, B, C, and D, possesses the structure of 4-O-methyl-2, 6-dideoxy-D-lyxo-hexose (VI).
Similar content being viewed by others
References
Yu. A. Berlin, S. E. Esipov, M. N. Kolosov, M. M. Shemyakin, and M. G. Brazhnikova, Tetrah. Let.,1323, 3513, 1964.
Yu. A. Berlin, S. E. Esipov, M. N. Kolosov, and V. A. Krivoruchko, KhPS [Chemistry of Natural Compounds],3, 405, 1967.
Yu. A. Berlin, S. E. Esipov, M. N. Kolosov, and M. M. Shemyakin, Tetrah. Let.,143, 1643, 1966.
Yu. A. Berlin, S. E. Esipov, O. A. Kiseleva, and M. N. Kolosov, KhPS [Chemistry of Natural Compounds],3, 331, 1967.
A. I. Scott, “Interpretation of the Ultraviolet Spectra of Natural Products”, Pergamon Press, 58, 1964.
N. S. Bhacca, L. F. Johnson, and J. N. Shoolery, NMR Spectra Catalog, Varian Associates, Palo Alto, 1, no. 11, 1962.
A. Rassat, C. W. Jefford, J. M. Lehn, and B. Waegell, Tetrah. Let.,233, 1964.
P. J. Beynon, P. M. Collins, P. T. Doganges, and W. G. Overend, J. Chem. Soc., Sec. C, 1131, 1966.
R. U. Lemieux and J. D. Stevens, Can. J. Chem.,44, 249, 1966.
K. Heyns and H. Paulsen, Adv. Carbohydrate Chem.,17, 169, 1962.
N. Bhacca and D. Williams, Applications of NMR Spectroscopy in Organic Chemistry [Russian translation], p. 231, 1966.
Yu. A. Berlin, D. V. Borisova, S. E. Esipov, M. N. Kolosov, and V. A. Krivoruchko, KhPS [Chemistry of Natural Compounds], 5, 109, 1969 [following paper].
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 5, No. 2, pp. 103–109, 1969
Rights and permissions
About this article
Cite this article
Berlin, Y.A., Esipov, S.E., Kolosov, M.N. et al. Olivomycin and related antibiotics. Chem Nat Compd 5, 89–93 (1969). https://doi.org/10.1007/BF00633285
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00633285