Summary
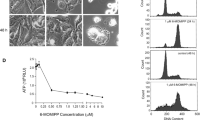
S 12363, a new vinca alkaloid derivative, was considerably more cytotoxic to murine L1210 cells and five human tumor cell lines (HL60, HT-29, COLO 320DM, NCI-H460, and PANC-1) than was vincristine (VCR) or vinblastine (VLB). S 12363 bound to tubulin in crude extracts from brain or L1210 cells with an affinity similar to that of VLB and VCR (apparentK d value: 1.1–1.6, 1.2–1.7, and 0.6–0.8 μM, respectively). After 1 h exposure, the accumulation of 20 nM [3H]-S 12363 by L1210 cells was 4-to 18-fold that of [3H]-VLB and [3H]-VCR, respectively. After the cells had been preloaded for 1 h with the labeled drugs and then incubated for 3 h in drug-free medium, 37%–55% of the [3H]-S 12363 was retained by the cells vs 36%–47% of the [3H]-VCR and <6% of the [3H]-VLB. Similar results were obtained for the five human cell lines tested. The accumulation factors (intracellular vs extracellular concentrations) found for [3H]-S 12363 (54- to 167-fold) were significantly higher than those observed for [3H]-VCR (5- to 14-fold) or [3H]-VLB (19- to 41-fold). >90% of the radioactivity extracted from L1210 cells that had been treated with [3H]-S 12363 was recovered as unmodified drug, demonstrating that [3H]-S 12363 was not metabolized by these cells. S 12362, which differs from S 12363 only in the absolute configuration of the asymmetric carbon atom of its α-aminophosphonic side chain, was 300 times less cytotoxie, bound to tubulin with a lower affinity (apparentK d value, 4.9–9.6 μm), and was neither accumulated nor retained by the cells. Taken together, these results demonstrate that the potency of S 12363 is due at least in part to its cellular accumulation and retention.
Similar content being viewed by others
References
Alley MC, Scudiero DA, Monks A, Hursey ML, Czerwinski A, Fine D, Abbott BJ, Mayo JF, Shoemaker RK, Boyd MR (1988) Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res 48: 589–611
Beck WT, Cirtain MC, Lefko JL (1983) Energy-dependent reduced drug binding as a mechanism of vinca alkaloid resistance in human leukemic lymphoblasts. Mol Pharmacol 24: 484–492
Bleyer WA, Frisby SA, Oliverio VT (1975) Uptake and binding of vincristine by murine leukemia cells. Biochem Pharmacol 24: 633–639
Bowman LC, Houghton JA, Houghton PJ (1988) Formation and stability of vincristine-tubulin complexes in kidney cytosols. Biochem Pharmacol 37: 1251–1257
Cheng YC, Prusof WH (1973) Relationship between the inhibition constant (Ki) and the concentration of inhibitor which causes 50 per cent inhibition (150) of an enzymatic reaction. Biochem Pharmacol 22: 3099–3108
Ferguson PJ, Cass CE (1985) Differential cellular retention of vincristine and vinblastine by cultured human promyelocytic leukemia HL-60/CI cells. The basis of differential toxicity. Cancer Res 45: 5480–5488
Ferguson PJ, Phillips JR, Selner M, Cass CE (1984) Differential activity of vicristine and vinblastine against cultured cells. Cancer Res 44: 3307–3312
Gout PW, Noble RL, Bruchovski N, Beer CT (1984) Vinblastine and vincristine: growth inhibitory effects correlate with their retention by cultured Nb2 node lymphoma cells. Int J Cancer 34: 245–248
Hammond JR, Johnstone RM, Gros P (1989) enhanced efflux of [3H]vinblastine from Chinese hamster ovary cells transfected with a full-length complementary DNA clone for the MDRI gene. Cancer Res 49: 3867–3871
Himes RH, Kersey RN, Heller-Bettinger I, Samson FE (1976) Action of the vinca alkaloids vincristine, vinblastine, trans desacetyl vinblastine amide on microtubules in vitro. Cancer Res 36: 3798–3802
Horton JK, Houghton PJ, Houghton JA (1988) Relationships between tumor responsiveness, vincristine pharmacokinetics and arrest of mitosis in human tumor xenografts. Biochem Pharmacol 37: 3995–4000
Houghton JA, Williams LG, Torrance PM, Houghton PJ (1984) Determinants of intrinsic sensitivity to vinca alkaloids in xenografts of pediatric rhabdomyosarcomas. Cancer Res 44: 582–590
Houghton JA, Williams LG, Houghton PJ (1985) Stability of vincristine complexes in cytosols derived from xenografts of human rhabdomyosarcoma and normal tissues of the mouse. Cancer Res 45: 3761–3767
Houghton JA, Williams LG, Dodge RK, George SL, Hazelton RJ, Houghton PJ (1987) Relationship between binding affinity, retention and sensitivity of human rhabdomyosarcoma xenografts to vinca alkaloids. Biochem Pharmacol 36: 81–88
Jordan MA, Himes RH, Wilson L (1985) Comparison of the effects of vinblastine, vincristine, vindesine and vinepidine on microtubule dynamics and cell proliferation in vitro. Cancer Res 45: 2741–2747
Kraus-Berthier L, Visalli M, Seurre G, Bizzari JP, Pierré A (1990) Effect of a new vinca alkaloid, S 12363, on human tumor xenografts in nude mice. Proc Am Assoc Cancer Res 31: 415
Lavielle G, Hautefaye P, Schaeffer G, Boutin JA, Cudennec CA, Pierré A (1991) New α-aminophosphonate derivatives of vinblastine: chemistry and antitumor activity. J Med Chem 37: 1998–2003
Lengsfeld AM, Dietrich J, Schultze-Maurer B (1982) Accumulation and release of vinblastine and vincristine by HeLa cells: light microscopic, cinematographic and biochemical study. Cancer Res 42: 3798–3805
Léonce S, Anstett M, Combe-Pérez V, Pierré A (1991) Flow cytometric evaluation of the cell cycle perturbations induced by S 12363, a new vinca alkaloid derivative. Anticancer Drugs 1: 179–183
Lowry OH, Rosebrough NJ, Fall AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193: 265–275
Na GC, Timasheff SN (1986) Interaction of vinblastine with calf brain tubulin: multiple equilibria. Biochemistry 25: 6214–6222
Pierré A, Léonce S, Anstett M, Hautefaye P, Lavielle G, Cudennec CA (1989) Cytotoxic properties of a new potent vinca alkaloid derivative on human solid tumors in vitro. Proc Am Assoc Cancer Res 30: 581
Pierré A, Lavielle G, Hautefaye P, Seurre G, Léonce S, Saint-Dizier D, Boutin JA, Cudennec CA (1990) Pharmacological properties of a new α-aminophosphonate acid derivative of vinblastine. Anticancer Res 10: 139–144
Pierré A, Kraus-Berthier L, Atassi G, Cros S, Poupon MF, Lavielle G, Berlion M, Bizzari JP (1991) Preclinical antitumor activity of a new vinca alkaloid derivative, S 12363. Cancer Res 51: 2312–2318
Rivera-Fillat MP, Pallarés-Trujillo J, Doménech C, Grau-Oliete MR (1988) Comparative uptake, rention and action of vincristine, vinblastine and vindesine on murine leukaemic lymphoblasts sensitive and resistant to vincristine. Br J Pharmacol 93: 902–908
Shelanski ML, Gaskin E, Cantor CR (1973) Microtubule assembly in the absence of added nucleotides. Proc Natl Acad Sci USA 70: 765–769
Singer WD, Hersh RT, Himes RH (1988) Effect of solution variables on the binding of vinblastine to tubulin. Biochem Pharmacol 37: 2691–2696
Sirotnak FM, Yang CH, Mines LS, Oribe E, Biedler JL (1986) Markedly altered membrane transport and intracellular binding of vincristine in multidrug-resistant Chinese hamster cells selected for resistance to vinca alkaloids. J Cell Physiol 126: 266–274
Skovsgaard T (1978) Mechanism of cross-resistance between vincristine and daunorubicin in Ehrlich ascites tumor cells. Cancer Res 39: 4722–4727
Zweig MH, Chignell CF (1973) Interaction of some colchicine analogs, vinblastine and podophyllotoxin with rat brain microtubule protein. Biochem Pharmacol 22: 2141–2150
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pierré, A., Pérez, V., Léonce, S. et al. Relationship between the cellular accumulation and the cytotoxicity of S12363, a new vinca alkaloid derivative. Cancer Chemother. Pharmacol. 29, 367–374 (1992). https://doi.org/10.1007/BF00686005
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00686005