Abstract
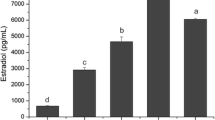
Recently, it has been shown that granulosa-cell secretion of plasminogen activator (PA) is responsive to luteinizing hormone (LH)/human chorionic gonadotropin (hCG) as well as follicle stimulating hormone (FSH) in the rat and the pig. Accordingly, we asked whether PA activity in follicular fluid from exogenously stimulated human follicles was different from that of normal cycles and whether or not these activities correlated with follicular maturation as determined by follicular fluid steroid concentration. Follicular aspirates were obtained from women who were participating in an in vitro fertilization protocol. Follicular fluid concentrations of estradiol and progesterone were determined by established radioimmunoassay. PA activity, determined using a modified indirect solid-phase radioassay, was significantly less in follicles from patients treated with human menopausal gonadotropin (hMG) plus clomiphene (P<0.05) compared to untreated patients or those receiving hMG or clomiphene alone. Correlations of PA activity and follicular fluid steroid concentrations demonstrated no significant correlation in samples from treated patients. In contrast, untreated spontaneously cycling patients had a significant (r=0.89,P<0.05), positive correlation between follicular fluid estradiol levels. There was no correlation between PA activity and follicular fluid progesterone levels in any of the groups. These results suggest that a subtle balance in granulosa-cell secretion of PA and steroids exists, which appears to be disrupted by follicular hyperstimulation during treatment of patients participating in in vitro fertilization protocols.
Similar content being viewed by others
References
Granelli-Piperno A, Reich E: A study of proteases and protease-inhibitor complexes in biological fluids. J Exp Med 1978;148:223–234
Strickland S, Beers W: Studies on the role of plasminogen activator in ovulation. J Biol Chem 1976;251:5694–5702
Beers W, Strickland S: A cell culture assay for follicle-stimulating hormone. J Biol Chem 1978;253:3877–3881
Liu WK, Burleigh BD, Ward D: Steroid and plasminogen activator production by cultured rat granulosa cells in response to hormone treatment. Mol Cell Endocrinol 1981;21:63–73
Beers W: Follicular plasminogen and plasminogen activator and the effect of plasmin on ovarian follicle wall. Cell 1975;6:379–386
Beers W, Strickland S, Reich E: Ovarian plasminogen activator: Relationship to ovulation and hormonal regulation. Cell 1975;6:387–394
Wang C: Gonadotropins regulate plasminogen activator production by rat granulosa cells. Endocrinology 1983; 112:1201–1207
Too SKL, Bryant-Greenwood GD: The role of relaxin and the differential production, in vitro, of plasminogen activator by periovulatory rat granulosa cells. 65th Annual Meeting of the Endocrine Society, Abstr No. 856, 1983
Shaw KJ, Campeau JC, Roche PC, diZerega GS: Porcine granulosa cell production of plasminogen activator: Disparity between the effects of hCG and pFSH. Endokrinologie (in press), 1985
Marrs RP, Vargyas JM, Saito H, Gibbons WE, Berger T, Mishell DR, Jr: Clinical application of techniques used in human in vitro fertilization research. Am J Obstet Gynecol 1983;477–483
Mishell DR, Nakamura RM, Stone SG, Kharma K, Nagata Y, Thorneycroft IH: Serum gonadtoropin and steroid paterns during the normal menstrual cycle. Am J Obstet Gynecol 1971;111:60–65
Roche PC, Campeau JD, Shaw ST Jr: Comparative electrophoretic analysis of human and porcine plasminogen activators in SDS-polyacrylamide gels containing plasminogen and casein. Biochim Biophys Acta 1983;745:82–89
Roche PC, Campeau JD, Shaw ST Jr: A rapid and highly sensitive solid-phase radioassay for plasminogen activators. Thromb Res (in press), 1984
Wijnegaards G, Kluft C, Groenveld E: Demonstration of urokinase-related fibrinolytic activity in human serum. Br J Haematol 1982;51:165–169
Croyton FE, Cowdon DJ: Applied General Statistics, 2nd ed. Englewood Cliffs, Prentice Hall, 1965, p 722
Channing CP, Anderson LD, Hoover DJ, Kolena J, Osteen KG, Pomgrantz SH, Tanabe K: The role of non steroidal regulators in control of oocyte and follicular maturation. Res Prog Horm Res 1982;38:331–408
diZerega GS, Campeau JD, Nakamura RM, Ujita EL, Lobo R, Marrs RP: Activity of a human follicular fluid protein(s) in spontaneous and induced ovarian cycles. J Clin Endocrinol Metab 1983;57:838–846
diZerega GS, Marrs RP, Lobo R, Ujita EL, Brown J, Campeau JD: Correlation of inhibin and follicle regulatory protein activities with follicular fluid steroid levels in anovulatory patients. Fertil Steril 1984;41:849–855
Fowler RE, Edwards RG, Walters DE, Chan STH, Steptoe PC: Steroidogenesis in preovulatory follicles of patients given human meopausal and chorionic gonadotropins as judged by radioimmunoassay of steroids in follicular fluids J Endocrinol 1978;77:161–168
Channing CP, Liv CQ, Jones GS, Jones H: Decline of feilicular oocyte maturation inhibitor coincident with maturation and achieveentk of fertilizability of oocytes recovered at midcycle of gonadotropin-treated women. Proc Natl Acad Sci USA 1983;80:4184–4188
Marrs RP, Vargyas JM, Lobo R: Comparison of the intrafollicular hormonal mileau with fertilization of human oocytes in vitro (submitted for publication)
Marrs RP, Lobo R, Campeau JD, Nakamura RM, Brown H, Ujita EL, diZerega GS: Correlation of human follicular fluid inhibin activity with spontaneous and induced follicle maturation. J Clin Endocrinol Metab 1984;58:704–709
Author information
Authors and Affiliations
Additional information
Supported in part by Clinical Investigator Award HD-00401 and by the University of Southern California Faculty Research and Innovation Award.
Rights and permissions
About this article
Cite this article
Weimer, S.L., Campeau, J.D., Marrs, R.P. et al. Alteration of human follicular fluid plasminogen activator activity by ovarian hyperstimulation. J Assist Reprod Genet 1, 263–266 (1984). https://doi.org/10.1007/BF01131626
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01131626