Abstract
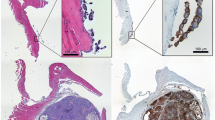
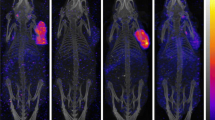
Murine monoclonal antibody 196-14 recognizes the ovarian-cancer-associated antigen CA 125, but the epitope it recognizes is different from that of monoclonal antibody OC125. We developed a human/mouse chimeric 196-14 using the variable regions of the murine 196-14 and human heavy-chain (γl) and light-chain (κ) constant regions. Cell binding and competitive inhibition assays using chimeric 196-14 labeled with125I,111In or99mTc demonstrated that the in vitro immunoreactivity of the chimeric antibody was identical to that of the parental murine monoclonal antibody. However, in mice bearing human ovarian cancer xenografts, the clearance from blood was faster and absolute levels of accumulation in the tumor were lower for the125I-labeled or99mTc-labeled chimeric antibody than for the murine antibody labeled with the corresponding radionuclides. The tumor-to-blood radioactivity ratio was not significantly different between the chimeric antibody and the murine antibody, regardless of the radionuclide used for labeling. Chimeric antibody 196-14 labeled with131I,111In or99mTc is promising for the radioimmunoimaging of ovarian cancer.
Similar content being viewed by others
References
Abdel-Nabi HH, Schwartz AN, Higano CS, Wechter DG, Unger MW (1987) Colorectal carcinoma: detection with indium-111 anticarcinoembryonic-antigen monoclonal antibody ZCE-025. Radiology 164:617
Bast RC Jr, Feeney M, Lazarus H, Nadler LM, Colvin RB, Knapp RC (1981) Reactivity of a monoclonal antibody with human ovarian carcinoma. J Clin Invest 68: 1331
Berzen G, Mayr AC, Langer M, Becker R, Cordes M, Zwicker C, Koppenhagen K, Felix R (1989) Radioimmunoscintigraphy of ovarian cancer with 131-iodine labeled OC-125 antibody fragments. Eur J Nucl Med 15:42
Boulianne GL, Hozumi N, Shulman MJ (1984) Production of functional chimeric mouse/human antibody. Nature 312: 643
Brown BA, Davis GL, Saltzgaber-Mullaer J, Simon P, Ho M-K, Shaw PS, Stone BA, Sands H, Moore GP (1987) Tumor-specific genetically engineered murine/human chimeric monoclonal antibody. Cancer Res 47: 3577
Carrasquillo JA, Mulshine JL, Bunn PA Jr, Reynolds JC, Foon KA, Schroff RW, Perentesis P, Steis RG, Keenan AM, Horowitz M, Larson SM (1987) Indium-111-T101 monoclonal antibody is superior to iodine-131-T101 in imaging of cutaneous T-cell lymphoma. J Nucl Med 28: 281
Chatal JF, Fumoleau P, Saccavini JC, Thedrez P, Curtet C, Bianco-Arco A, Chetanneau A, Peltier P, Kremer M (1987) Immunoscintigraphy of recurrences of gynecologic carcinomas. J Nucl Med 28: 1807
Davis HM, Zurawski VR Jr Bast RC Jr, Klug TL (1986) Characterization of the CA 125 antigen associated with human epitherial ovarian carcinomas. Cancer Res 46: 6143
Dutin JP, Peltier P, Chatal JF (1991) Bayesian analysis of the utility of In-111 OC 125 immunoscintigraphy (IS) in diagnosing ovarian cancer (abstract). J Nucl Med 32: 941
Hamilton TC, Young RC, McKoy WM, Grotzinger KR, Green JA, Chu EW, Whang-Peng, J, Rogan AM, Green WR, Ozols RF (1983) Characterization of a human ovarian carcinoma cell line (NIH: OVCAR3) with androgen and estrogen receptors. Cancer Res 43: 5379
Heal A, Tyson I, Robb S, Docs J, Courey M (1990) Erroneous CA-125 values due to HAMA in serum of patients treated with mouse MoAbs (abstract). J Nucl Med 31: 715
Hirano T, Tomiyoshi K, Watanabe N, Tateno M, Oriuchi N, Inoue T, Endo K (1992) Technetium-99m-labeled anti-fibrin monoclonal antibody accumulation in an inflammatory focus. J Nucl Med 33: 1181
Ishiwata I, Soma M, Ono I, Nakaguchi T, Ishiwata C, Nozawa S, Ishikawa H (1989) Biological characteristics of adenocarcinoma cell line (HOUA-I) and B-lymphoblastoid cell line (HOUA-II) established from the same tissue of endometrial adenocarcinoma. Jikeikai Med J 36: 303
Khazaeli MB, Saleh MN, Liu TP, Meredith RF, Wheeler RH, Baker TS, King D, Secher D, Allen L, Rogers K, Colcher D, Schlom J Shochat D, LoBuglio AF (1991) Pharmacokinetics and immune response of131I-chimeric mouse/human B72.3 (human γ4) monoclonal antibody in humans. Cancer Res 51: 5461
Lindmo T, Boven E, Cuttitta F, Fedorko J, Bunn PA Jr (1978) Determination of immunoreactive fraction of radiolabeled monoclonal antibodies by linear extrapolation to binding at infinite antigen excess. J Immunol Methods 72: 101
Mather SJ, Ellison D (1990) Reduction-mediated technetium-99m labeling of monoclonal antibodies. J Nucl Med 31: 692
Meredith RF, LoBuglio AF, Plott WE, Orr RA, Brezovich IA, Russell CD, Harvey EB, Yester MV, Wagner AJ, Spencer SA, Wheeler RH, Saleh MN, Rogers KJ, Polansky A, Salter MM, Khazaeli MB (1991) Pharmacokinetics, immune response, and biodistribution of iodine-131-labeled chimeric mouse/human IgG, κ 17-1A monoclonal antibody. J Nucl Med 32: 1162
Morrison SL, Johnson MJ, Herzenberg LA, Oi TV (1984) Chimeric human antibody molecules: mouse antigen-binding domains with human constant region domains. Proc Natl Acad Sci USA81: 6851
Muto MG, Lepisto EM, Van den Abbeele AD, Knapp RC, Kassis AI (1989) Influence of human antimurine antibody on CA 125 levels in patients with ovarian cancer undergoing radioimmunotherapy or immunoscintigraphy with murine monoclonal antibody OC125. Am J Obstet Gynecol 161: 1206
Nishimura Y, Yokoyama M, Araki K, Ueda R, Kudo A, Watanabe T (1987) Recombinant human-mouse chimeric monoclonal antibody specific for common acute lymphocytic leukemia antigen. Cancer Res 47: 999
Phillips J, Sikora K, Watson JV (1982) Localization of glioma by a human monoclonal antibody. Lancet2: 1214
Reuland P, Winker KH, Heuchert T, Ruck P, Mueller-Schauenburg W, Weller S, Feine U (1991) Detection of infection in postoperative orthopedic patients with technetium-99m-labeled monoclonal antibodies against granulocytes. J Nucl Med 32: 2209
Saga T, Endo K, Koizumi M, Kawamura Y, Watanabe Y, Konishi J, Ueda R, Nishimura Y, Yokoyama, M, Watanabe T (1990) In vitro and in vivo properties of human/mouse chimeric monoclonal antibody specific for common acute lymphocytic leukemia antigen. J Nucl Med31: 1077
Saga T, Ishiwata I, Endo K, Sakahara H, Koizumi M, Watanabe Y, Nakai T, Hosono M, Ishikawa H, Sawada M, Konishi J (1990) An antibody-tumor model for the targeting of CA 125-producing gynecologic malignancies. Jpn J Cancer Res 81: 1141
Sahagan BG, Dorai H, Saltzgaber-Mullaer J, Toneguzzo F, Guindon CA, Lilly SP, McDonald KW, Morrissey DV, Stone BA Davis GL, McIntosh PK, Moore GP (1986) A genetically engineered murinc/human chimeric antibody retains specificity for human tumor associated antigen. J Immunol 137: 1066
Sakahara H, Endo K, Nakashima T, Koizumi M, Ohta H, Torizuka K, Furukawa T, Ohmomo Y, Yokoyama A, Okada K, Yoshida O, Nishi S (1985) Effect of DTPA conjugation on the binding activity and biodistribution of monoclonal antibodies against alpha-fetoprotein. J Nucl Med 26: 750
Sakahara H, Endo K, Nakashima T, Koizumi M, Kunimatsu M, Kawamura Y, Ohta H, Nakamura T, Tanaka H, Kotoura Y, Yamamuro T, Hosoi S, Toyama S, Torizuka K (1987) Localization of human osteogenic sarcoma xenografts in nude mice by a monoclonal antibody labeled with radioiodine and indium-111. J Nucl Med 28: 342
Sakahara H, Reynolds JA, Carrasquillo JA, Lor ME, Maloney PJ, Lotze MT, Larson SM, Neumann RD (1989) In vitro complex formation and biodistribution of mouse antitumor monoclonal antibody in cancer patients. J Nucl Med 30: 1311
Sakamoto M (1988) Cyto-biological and immunological characterization of a newly established cell line (TMCC-1) derived from human uterine cervical adenocarcinoma. J Tokyo Med Coll46: 925
Scatchard G (1949) The attraction of proteins for small molecules and ions. Ann NY Acad Sci 51: 660
Schwarz A, Steinstrasser A (1987) A novel approach to Tc-99m-labeled monoclonal antibodies (abstract). J Nucl Med28: 721
Sikora K (1987) Human monoclonal antibodies to cancer cells. In: Human hybridomas. Dekker, New York Basel, p 159
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kobavashi, H., Sakahara, H., Saga, T. et al. A human/mouse chimeric monoclonal antibody against CA125 for radioimmunoimaging of ovarian cancer. Cancer Immunol Immunother 37, 143–149 (1993). https://doi.org/10.1007/BF01525427
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01525427