Abstract
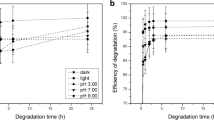
The hydrolysis rate of chlorpyrifos (the active ingredient of DURSBAN® and LORSBAN® insecticides, registered trademarks of The Dow Chemical Company, Midland, Michigan) in water followed simple first-order kinetics over the concentration range, 3×10−9 to 3×10−7 M. In buffered distilled water at 25°C and pH 8.1, 6.9, and 4.7, the half-life was 22.8, 35.3, and 62.7 days, respectively. Additional data were obtained for rates at 15° and 35°C. The activation energy for the reaction under these conditions was 21.2 kcal/mole. A 16-fold rate enhancement was demonstrated in canal and pond water at 25°C. Likewise, there was a catalytic effect on the hydrolysis rate in the presence of copper (II) ion.
The same information was generated for chlorpyrifos-methyl, the dimethyl analog of chlorpyrifos. In this case, the corresponding half-life values for hydrolysis at 25°C and pH 7.8, 6.7, and 4.2 were 12.7, 17.4, and 22.8 days, respectively. The activation energy was 20.8 kcal/mole, not significantly different from that for chlorpyrifos. A hydrolysis rate enhancement also occurred for chlorpyrifos-methyl in canal water.
Qualitatively, the products of chlorpyrifos hydrolysis were 3,5,6-trichloro-2-pyridinol (I),O-ethylO-hydrogenO-(3,5,6-trichloro-2-pyridyl) phosphorothioate (II), andO, O-dihydrogenO-(3,5,6-trichloro-2-pyridyl) phosphorothioate (III). In the case of chlorpyrifos-methyl, the hydrolysis products were compound I and the methyl analog of compound II.
Similar content being viewed by others
References
Bruno, G. A., and J. E. Christian: Determination of carbon-14 in aqueous bicarbonate solutions by liquid scintillation counting techniques. Application to biological fluids. Anal. Chem.33, 1216 (1961).
Christian, G. D., and W. C. Purdy: The residual current in ortho-phosphate medium. J. Electroanal. Chem.3, 363 (1962).
Collier, B. L., and C. D. Dieter: DURSBAN® … A new insecticide for chinch bug and sod webworm control in St. Augustinegrass. DOWN TO EARTH®21(3), 3, (1965).
Courtney, R. C., R. L. Gustafson, S. J. Westerback, H. Hyytiainen, S. Chaberek, and A. E. Martell: Metal chelate compounds as catalysts in the hydrolysis of isopropyl methyl phosphoro-fluoridate and diisopropylphosphorofluoridate. J. Amer. Chem. Soc.79, 3030 (1957).
Fukuto, T. R.: The chemistry and action of organic phosphorous insecticides. Adv. Pest Control Research1, 147 (1957).
Gray, H. E.: DURSBAN® … A new organo-phosphorus insecticide. DOWN TO EARTH®21(3), 1 (1965).
Kenaga, E. E., W. K. Whitney, J. L. Hardy, and A. E. Doty: Laboratory tests with DURSBAN® insecticide. J. Econ. Entomol.58, 1043 (1965).
Ketelaar, J. A. A., H. R. Gersman, and M. M. Beck: Metal-catalysed hydrolysis of thiophosphoric esters. Nature177, 392 (1956).
Martell, A. E., and M. Calvin: Chemistry of the metal chelate compounds, pp 336–432. New York: Prentice-Hall (1952).
Martell, A. E., S. Chaberek, R. C. Courtney, S. Westerback, and H. Hyytiainen: Hydrolytic tendencies of metal chelate compounds, I. Cu (II) chelates. J. Amer. Chem. Soc.79, 3036 (1957).
Mortland, M. M., and K. V. Raman: Catalytic hydrolysis of some organic phosphate pesticides by copper (II). J. Agr. Food Chem.15, 163 (1967).
Muelder, W. W., and M. N. Wass: A semimicro preparation ofO,O-diethylO-(3,5,6-trichloro-2-pyridyl-2,6-14C2) phosphorothioate. J. Agr. Food Chem.15, 508 (1967).
Schaefer, C. H., and E. F. Dupras, Jr.: The effects of water quality, temperature and light on the stability of organophosphorous larvicides used for mosquito control. Proc. Calif. Mosquito Control Assoc.37, 67 (1969).
Schaefer, C. H., and E. F. Dupras, Jr.: Factors affecting the stability of DURSBAN® in polluted waters. J. Econ. Entomol.63, 701 (1970).
Snedecor, G. W., and W. C. Cochran: Statistical Methods, 6th ed., p. 432. Ames, Iowa: The Iowa State University Press (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meikle, R.W., Youngson, C.R. The hydrolysis rate of chlorpyrifos,O-O-diethylO-(3,5,6-trichloro-2-pyridyl) phosphorothioate, and its dimethyl analog, chlorpyrifos-methyl, in dilute aqueous solution. Arch. Environ. Contam. Toxicol. 7, 13–22 (1978). https://doi.org/10.1007/BF02332034
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02332034