Abstract
Background
The published results on expression of c-kit in benign and malignant breast tissues vary.
Materials and Methods
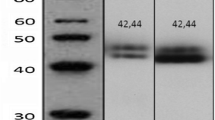
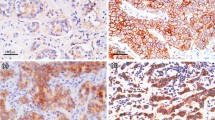
The immunohistochemical expression of c-kit proto-oncogene product in 52 invasive breast cancer tissues and 16 benign breast tumor (fibroadenoma) tissues was studied using anti-c-kit proto-oncogene product antibody. Its expression was evaluated by immunoreactive score (IRS).
Results
In breast cancer tissues, the mean IRS of c-kit proto-oncogene product expression was significantly increased compared to those of fibroadenoma (3.4 ± 2 and 2.19 ± 1.8, respectively,p = 0.035). The mean IRS of c-kit expression was higher in the group comprising estrogen (ER) positive tumor than in the group of ER negative (4.1 ± 2.1 and 2.7 ± 1.8, respectively,p = 0.012) but no statistically significant relationship was seen between the expression of c-kit proto-oncogene product and other clinicopathological parameters of breast cancer, including histologic type, tumor size, lymph node metastasis, distant metastasis, stage, progesterone receptor, c-erbB-2 expression, menopausal status and age of the patient (p > 0.05).
Conclusions
Our results show that a high level of c-kit expression occurs frequently in invasive breast cancer, and its expression is associated with ER but unrelated to other clinico-pathological variables.
Similar content being viewed by others
References
Yarden Y, et al. Human proto-oncogene c-kit: a new cell surface receptor tyrosine kinase for an unidentified ligand.EMBO J 1987;6: 3341–3351.
Lammie A, Drobnjak M, Gerald W, Saad A, Cote R, Cordon-Cordo C. Expression of c-kit and kit ligand proteins in normal human tissues.J Histochem Cytochem 1994;42: 1417–1425.
Huang E, et al. The hematopoietic growth factor KL is encoded by the SI locus and is the ligand of the c-kit receptor, the gene product of the W locus.Cell 1990;63: 225–233.
Natali PG, Nicotra MR, Sures I, Santoro E, Bigotti A, Ullrich A. Expression of c-kit receptor in normal transformed human nonlymphoid tissues.Cancer Res 1992;52: 6139–6143.
Escribano L, Ocqueteau M, Almeida J, Orfao A, San Miguel JF. Expression of the c-kit (CD117) molecule in normal and malignant hematopoiesis.Leuk Lymphoma 1998;30: 459–466.
Hirota S, et al. Gain-of function mutations of c-kit in human gastrointestinal stromal tumors.Science 1998;279: 577–580.
Tsuura Y, et al. Preferential localization of c-kit product in tissue mast cells, basal cells of skin, epithelial cells of breast, small cell lung carcinoma and seminoma/dysgerminoma in human: immunohistochemical study on formalin-fixed, paraffin-embedded tissues.Virchows Arch 1994;424: 135–141.
Woenckhaus C, Giebel J, Failing K, Fenic I, Dittberner T, Poetsch M. Expression of AP-2α, c-kit, and cleaved caspase- 6 and -3 in naevi and malignant melanomas of the skin. A possible role for caspases in melanoma progression?J Pathol 2003;201: 278–287.
Lux ML, et al. KIT extracellular and kinase domain mutations in gastrointestinal stromal tumors.Am J Pathol 2000;156: 791–795.
Natali PG, Nicotra MR, Sures I, Mottolese M, Botti C, Ullrich A. Breast cancer is associated with loss of the c-kit oncogene product.Int J Cancer 1992;52: 713–717.
Chui X, et al. Immunohistochemical expression of the c-kit proto-oncogene product in human malignant and nonmalignant breast tissues.Br J Cancer 1996;73: 1233–1236.
Ko CD, et al. The meaning of the c-kit proto-oncogene product in malignant transformation in human mammary epithelium.Clin Exp Metastasis 2003;20: 593–597.
Yared MA, Middleton LP, Bernstam FM, Cristofanilli M, Sahin AA. Expression of c-kit proto-oncogene product in breast tissue.Breast J 2004;10: 323–327.
Ulivi P, et al. c-kit and SCF expression in normal and tumor breast tissue.Breast Cancer Res Treat 2004;83: 33–42.
Palmu S, Soderstrom KO, Quazi K, Isola J, Salminen E. Expression of C-KIT and HER-2 tyrosine kinase receptors in poor-prognosis breast cancer.Anticancer Res 2002;22: 411–414.
Hines SJ, Organ C, Kornstein MJ, Krystal GW. Coexpression of the c-kit and stem cell factor genes in breast carcinomas.Cell Growth Differ 1995;6: 769–779.
Tsuda H, et al. Frequent KIT and epidermal growth factor receptor overexpressions in undifferentiated-type breast carcinomas with “stem-cell-like” features.Cancer Sci 2005;96: 333–339.
Simon R, et al. KIT (CD117)-positive breast cancers are infrequent and lack KIT gene mutations.Clin Cancer Res 2004;10: 178–183.
Crisi GM, Marconi SA, Makari-Judson G, Goulart RA. Expression of c-kit in adenoid cystic carcinoma of the breast.Am J Clin Pathol 2005;124: 733–739.
Mastropasqua MG, et al. Immunoreactivity for c-kit and p63 as an adjunct in the diagnosis of adenoid cystic carcinoma of the breast.Mod Pathol 2005;18: 1277–1282.
Remmele W, Stegner HE. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue.Pathology 1987;8: 138–140.
Mauro M, Druker B. STI 571: targeting BCR-ABL as therapy for CML.Oncologist 2001;6: 233–238.
Joensuu H, et al. Effect of the tyrosine kinase inhibitor STI 571 in a patient with a metastatic gastrointestinal stromal tumor.N Eng J Med 2001;344: 1052–1056.
Matsuda R, et al. Expression of the c-kit protein in human solid tumors and in corresponding fetal and adult normal tissues.Am J Pathol 1993;142: 339–346.
Strohmeyer T, et al. Expression of the hst-1 and c-kit protooncogenes in human testicular germ cell tumors.Cancer Res 1991;51: 1811–1816.
Tsuura Y, Suzuki T, Honma K, Sano M. Expression of c-kit protein in proliferative lesions of human breast: sexual difference and close association with phosphotyrosine status.J Cancer Res Clin Oncol 2002;128: 239–246.
Hornick JL, Fletcher CDM. Immunohistochemical staining for KIT (CD117) in soft tissue sarcomas is very limited in distribution.Am J Clin Pathol 2002;117: 188–193.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eroğlu, A., Sari, A. Expression of c-kit proto-oncogene product in breast cancer tissues. Med Oncol 24, 169–174 (2007). https://doi.org/10.1007/BF02698036
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02698036