Abstract
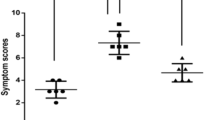
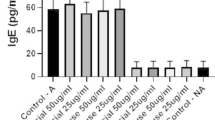
Allergic diseases are affecting public health and have increased over the last decade. Sensitization to mite allergens is a considerable trigger for allergy development. Storage mite-Tyrophagus putrescentiae shows great significance of allergenic potential and clinical relevance. The fungal immunomodulatory peptide FIP-fve has been reported to possess immunomodulatory activity. We aimed to determine whether T. putrescentiae-induced sensitization and airway inflammation in mice could be downregulated by FIP-fve in conjunction with denatured T. putrescentiae (FIP-fve and DN-Tp). Immune responses and physiologic variations in immunoglobulins, leukocyte subpopulations, cytokine productions, pulmonary function, lung pathology, cytokines in CD4+ and Treg cells were evaluated after local nasal immunotherapy (LNIT). After the LNIT with FIP-fve and DN-Tp, levels of specific IgE, IgG1, and IgG2a in the sera and IgA in the bronchoalveolar lavage fluid (BALF) were significantly reduced. Infiltrations of inflammatory leukocytes (eosinophils, neutrophils, and lymphocytes) in the airway decreased significantly. Production of proinflammatory cytokines (IL-5, IL-13, IL-17F and IL-23) and chemokine (IL-8) were significantly reduced, and Th1-cytokine (IL-12) increased in the airway BALF after LNIT. Pulmonary functions of Penh values were significantly decreased after the methacholine challenge, which resulted in a reduction of airway hypersensitivity after LNIT. Bronchus pathology showed a reduction of inflammatory cell infiltration and epithelium damage after LNIT. The IL-4+/CD4+ T cells could be downregulated and the IFN-γ+/CD4+ T cells upregulated. The Treg-related immunity of IL-10 and Foxp3 expressions in CD4+CD25+ cells were both upregulated after LNIT. In conclusion, LNIT with FIP-fve and DN-Tp had an anti-inflammatory effect on mite-induced airway inflammations and possesses potential as an immunomodulatory therapy agent for allergic airway diseases.









Similar content being viewed by others
References
Aida F, Shuhaimi M, Yazid M et al (2009) Mushroom as a potential source of prebiotics: a review. Trends Food Sci Technol 20:567–575. https://doi.org/10.1016/j.tifs.2009.07.007
Akdis CA, Akdis M (2014) Mechanisms of immune tolerance to allergens: role of IL-10 and Tregs. J Clin Invest 124:4678–4680. https://doi.org/10.1172/JCI78891
Akdis CA, Akdis M (2015) Mechanisms of allergen-specific immunotherapy and immune tolerance to allergens. World Allergy Organ J 8:17. https://doi.org/10.1186/s40413-015-0063-2
Arlian L, Geis D, Vyszenski-Moher D et al (1984) Antigenic and allergenic properties of the storage mite Tyrophagus putrescentiae. J Allergy Clin Immunol 74:166–171. https://doi.org/10.1016/0091-6749(84)90281-1
Casale TB, Stokes JR (2011) Future forms of immunotherapy. J Allergy Clin Immunol 127:8–15. https://doi.org/10.1016/j.jaci.2010.10.034
Cates EC, Fattouh R, Wattie J et al (2004) Intranasal exposure of mice to house dust mite elicits allergic airway inflammation via a GM-CSF-mediated mechanism. J Immunol 173:6384–6392. https://doi.org/10.4049/jimmunol.173.10.6384
Chang YC, Hsiao YM, Hung SC et al (2015) Alleviation of Dermatophagoides microceras-induced allergy by an immunomodulatory protein, FIP-fve, from Flammulina velutipes in mice. Biosci Biotechnol Biochem 79:88–96. https://doi.org/10.1080/09168451.2014.956682
Cho SW, Han DH, Kim JW et al (2018) House dust mite sublingual immunotherapy in allergic rhinitis. Immunotherapy 10:567–578. https://doi.org/10.2217/imt-2018-0013
Chu PY, Sun HL, Ko JL et al (2017) Oral fungal immunomodulatory protein-Flammulina velutipes has influence on pulmonary inflammatory process and potential treatment for allergic airway disease: a mouse model. J Microbiol Immunol Infect 50:297–306. https://doi.org/10.1016/j.jmii.2015.07.013
Creticos PS, Esch RE, Couroux P et al (2014) Randomized, double-blind, placebo-controlled trial of standardized ragweed sublingual-liquid immunotherapy for allergic rhinoconjunctivitis. J Allergy Clin Immunol 133:751–758. https://doi.org/10.1016/j.jaci.2013.10.041
Cui Y, Yu L, Teng F et al (2016) Transcriptomic/proteomic identification of allergens in the mite Tyrophagus putrescentiae. Allergy 71:1635–1639. https://doi.org/10.1111/all.12999
Custovic A (2017) Epidemiology of allergic diseases. Middleton’s allergy essentials. Elsevier, Amsterdam, pp 51–72. https://doi.org/10.1016/B978-0-323-37579-5.00003-9
D’Amato G, Holgate ST, Pawankar R et al (2015) Meteorological conditions, climate change, new emerging factors, and asthma and related allergic disorders. A statement of the World Allergy Organization. World Allergy Organ J 8:25. https://doi.org/10.1186/s40413-015-0073-0
de la Torre MV, Baeza ML, Nájera L et al (2018) Comparative study of adjuvants for allergen-specific immunotherapy in a murine model. Immunotherapy 10:1219–1228. https://doi.org/10.2217/imt-2018-0072
Dzoro S, Mittermann I, Resch-Marat Y et al (2018) House dust mites as potential carriers for IgE sensitization to bacterial antigens. Allergy 73:115–124. https://doi.org/10.1111/all.13260
El Enshasy HA, Hatti-Kaul R (2013) Mushroom immunomodulators: unique molecules with unlimited applications. Trends Biotechnol 31:668–677. https://doi.org/10.1016/j.tibtech.2013.09.003
Erban T, Rybanska D, Harant K et al (2016) Feces derived allergens of Tyrophagus putrescentiae reared on dried dog food and evidence of the strong nutritional interaction between the mite and Bacillus cereus producing protease bacillolysins and exo-chitinases. Front Physiol 7:53. https://doi.org/10.3389/fphys.2016.00053
Erban T, Klimov P, Molva V et al (2020) Whole genomic sequencing and sex-dependent abundance estimation of Cardinium sp., a common and hyperabundant bacterial endosymbiont of the American house dust mite, Dermatophagoides farinae. Exp Appl Acarol 80:363–380. https://doi.org/10.1007/s10493-020-00475-5
Erban T, Klimov PB, Harant K et al (2021) Label-free proteomic analysis reveals differentially expressed Wolbachia proteins in Tyrophagus putrescentiae: mite allergens and markers reflecting population-related proteome differences. J Proteom 249:104356. https://doi.org/10.1016/j.jprot.2021.104356
Fernández-Caldas E, Puerta L, Caraballo L (2014) Mites and allergy. In: Bergmann KC, Ring J (eds) History of allergy. Karger, Basel, pp 234–242. https://doi.org/10.1159/000358860
Frew AJ (2008) Sublingual Immunotherapy. New Engl J Med 358:2259–2264. https://doi.org/10.1056/NEJMct0708337
Gessner A, Mohrs K, Mohrs M (2005) Mast cells, basophils, and eosinophils acquire constitutive IL-4 and IL-13 transcripts during lineage differentiation that are sufficient for rapid cytokine production. J Immunol 174:1063–1072. https://doi.org/10.4049/jimmunol.174.2.1063
Hsieh CW, Lan JL, Meng Q et al (2007) Eosinophil apoptosis induced by fungal immunomodulatory peptide-fve via reducing IL-5α receptor. J Formos Med Assoc 106:36–43. https://doi.org/10.1016/S0929-6646(09)60214-X
Hubert J, Nesvorna M, Klimov P et al (2019) Differential allergen expression in three Tyrophagus putrescentiae strains inhabited by distinct microbiome. Allergy 74:2502–2507. https://doi.org/10.1111/all.13921
Jeong KY, Park JW, Hong CS (2012) House dust mite allergy in Korea: the most important inhalant allergen in current and future. Allergy Asthma Immunol Res 4:313–325. https://doi.org/10.4168/aair.2012.4.6.313
Jõgi NO, Kleppe Olsen R, Svanes C et al (2020) Prevalence of allergic sensitization to storage mites in Northern Europe. Clin Exp Allergy 50:372–382. https://doi.org/10.1111/cea.13536
Kim YK, Lee MH, Jee YK et al (1999) Spider mite allergy in apple-cultivating farmers: European red mite (Panonychus ulmi) and two-spotted spider mite (Tetranychus urticae) may be important allergens in the development of work-related asthma and rhinitis symptoms. J Allergy Clin Immunol 104:1285–1292. https://doi.org/10.1016/s0091-6749(99)70026-6
Kim J, Yi MH, Hwang Y et al (2018a) 16S rRNA profiling of the Dermatophagoides farinae core microbiome: Enterococcus and Bartonella. Clin Exp Allergy 48:607–610. https://doi.org/10.1111/cea.13104
Kim JH, Shin JU, Kim SH et al (2018b) Successful transdermal allergen delivery and allergen-specific immunotherapy using biodegradable microneedle patches. Biomaterials 150:38–48. https://doi.org/10.1016/j.biomaterials.2017.10.013
Ko JL, Hsu CI, Lin RH et al (1995) A new fungal immunomodulatory protein, FIP-fve isolated from the edible mushroom, Flammulina velutipes and its complete amino acid sequence. Eur J Biochem 228:244–249. https://doi.org/10.1111/j.1432-1033.1995.0244n.x
Komai M, Tanaka H, Masuda T et al (2003) Role of Th2 responses in the development of allergen-induced airway remodelling in a murine model of allergic asthma. Br J Pharmacol 138:912–920. https://doi.org/10.1038/sj.bjp.0705105
Kubo M (2018) Mast cells and basophils in allergic inflammation. Curr Opin Immunol 54:74–79. https://doi.org/10.1016/j.coi.2018.06.006
Lee YT, Lee SS, Sun HL et al (2013) Effect of the fungal immunomodulatory protein FIP-fve on airway inflammation and cytokine production in mouse asthma model. Cytokine 61:237–244. https://doi.org/10.1016/j.cyto.2012.09.024
Liao EC, Hsu EL, Tsai JJ et al (2009) Immunologic characterization and allergenicity of recombinant Tyr p 3 allergen from the storage mite Tyrophagus putrescentiae. Int Arch Allergy Immunol 150:15–24. https://doi.org/10.1159/000210376
Liao EC, Ho CM, Lin MY et al (2010a) Dermatophagoides pteronyssinus and Tyrophagus putrescentiae allergy in allergic rhinitis caused by cross-reactivity not dual-sensitization. J Clin Immunol 30:830–839. https://doi.org/10.1007/s10875-010-9446-x
Liao EC, Ho CM, Tsai JJ (2010b) Prevalence of Tyrophagus putrescentiae hypersensitivity in subjects over 70 years of age in a veterans’ nursing home in Taiwan. Int Arch Allergy Immunol 152:368–377. https://doi.org/10.1159/000288290
Liao E, Ho C, Yin S et al (2013) Immune responses to Tyrophagus putrescentiae-induced airway in áammation in mice. J Investig Allergol Clin Immunol 23:20–29
Lin YL, Liang YC, Tseng YS et al (2009) An immunomodulatory protein, Ling Zhi-8, induced activation and maturation of human monocyte-derived dendritic cells by the NF-κB and MAPK pathways. J Leukoc Biol 86:877–889. https://doi.org/10.1189/jlb.0708441
Liu YH, Tsai JJ (2005) Production of salivary immunoglobulin A and suppression of Dermatophagoides pteronyssinus-induced airway inflammation by local nasal immunotherapy. Int Arch Allergy Immunol 138:161–168. https://doi.org/10.1159/000088438
Liu YH, Kao MC, Lai YL et al (2003) Efficacy of local nasal immunotherapy for Dp2-induced airway inflammation in mice: using Dp2 peptide and fungal immunomodulatory peptide. J Allergy Clin Immunol 112:301–310. https://doi.org/10.1067/mai.2003.1619
Liu Y, Bastiaan-Net S, Wichers HJ (2020) Current understanding of the structure and function of fungal immunomodulatory proteins. Front Nutr 7:132. https://doi.org/10.3389/fnut.2020.00132
Malling HJ, Abreu-Nogueira J, Alvarez-Cuesta E et al (1998) Local immunotherapy. Allergy 53:933–943. https://doi.org/10.1111/j.1398-9995.1998.tb03793.x
Miller JD (2019) The role of dust mites in allergy. Clin Rev Allergy Immunol 57:312–329. https://doi.org/10.1007/s12016-018-8693-0
Moldaver D, Larché M (2011) Immunotherapy with peptides. Allergy 66:784–791. https://doi.org/10.1111/j.1398-9995.2011.02610.x
Moldaver D, Bharhani M, Wattie J et al (2014) Amelioration of ovalbumin-induced allergic airway disease following Der p 1 peptide immunotherapy is not associated with induction of IL-35. Mucosal Immunol 7:379–390. https://doi.org/10.1038/mi.2013.56
Moote W, Kim H (2011) Allergen-specific immunotherapy. Allergy Asthma Clin Immunol 7:1–7. https://doi.org/10.1186/1710-1492-7-S1-S5
Müsken H, Fernández-Caldas E, Marañón F et al (2002) In vivo and in vitro sensitization to domestic mites in German urban and rural allergic patients. J Investig Allergol Clin Immunol 12:177–181
Nakajima H, Takatsu K (2007) Role of cytokines in allergic airway inflammation. Int Arch Allergy Immunol 142:265–273. https://doi.org/10.1159/000097357
Nelson HS, Nolte H, Creticos P et al (2011) Efficacy and safety of timothy grass allergy immunotherapy tablet treatment in North American adults. J Allergy Clin Immunol 127:72-80.e2. https://doi.org/10.1016/j.jaci.2010.11.035
Nurieva RI, Chung Y (2010) Understanding the development and function of T follicular helper cells. Cell Mol Immunol 7:190–197. https://doi.org/10.1038/cmi.2010.24
Pajno GB, Fernandez-Rivas M, Arasi S et al (2018) EAACI Guidelines on allergen immunotherapy: IgE-mediated food allergy. Allergy 73:799–815. https://doi.org/10.1111/all.13319
Passalacqua G, Canonica GW (2006) Local nasal specific immunotherapy for allergic rhinitis. Allergy Asthma Clin Immunol 2:1–7. https://doi.org/10.1186/1710-1492-2-3-117
Pomés A, Chapman MD, Wünschmann S (2016) Indoor allergens and allergic respiratory disease. Curr Allergy Asthma Rep 16:43. https://doi.org/10.1007/s11882-016-0622-9
Pope SM, Brandt EB, Mishra A et al (2001) IL-13 induces eosinophil recruitment into the lung by an IL-5–and eotaxin-dependent mechanism. J Allergy Clin Immunol 108:594–601. https://doi.org/10.1067/mai.2001.118600
Prescott S, Nowak-Węgrzyn A (2011) Strategies to prevent or reduce allergic disease. Ann Nutr Metab 59:28–42. https://doi.org/10.1159/000334150
Reddy AT, Lakshmi SP, Reddy RC (2012) Murine model of allergen induced asthma. J vis Exp 63:e3771. https://doi.org/10.3791/3771
Rezvani M, Bernstein DI (2007) Anaphylactic reactions during immunotherapy. Immunol Allergy Clin N Am 27:295–307. https://doi.org/10.1016/j.iac.2007.03.010
Rivas MN, Chatila TA (2016) Regulatory T cells in allergic diseases. J Allergy Clin Immunol 138:639–652. https://doi.org/10.1016/j.jaci.2016.06.003
Sakaguchi S (2005) Naturally arising Foxp3-expressing CD25+ CD4+ regulatory T cells in immunological tolerance to self and non-self. Nat Immunol 6:345–352. https://doi.org/10.1038/ni1178
Sánchez-Borges M, Suárez-Chacon R, Capriles-Hulett A et al (2009) Pancake syndrome (oral mite anaphylaxis). World Allergy Organ J 2:91–96. https://doi.org/10.1186/1939-4551-2-5-91
Shamji M, Durham S (2011) Mechanisms of immunotherapy to aeroallergens. Clin Exp Allergy 41:1235–1246. https://doi.org/10.1111/j.1365-2222.2011.03804.x
Shen CY, Yen CY, Chien DK et al (2020) Influence of storage conditions on the infestation of Tyrophagus putrescentiae and prevalence of mite hypersensitivity in Taiwan. Exp Appl Acarol 80:381–398. https://doi.org/10.1007/s10493-019-00453-6
Steinke JW, Borish L (2001) Th2 cytokines and asthma. Interleukin-4: Its role in the pathogenesis of asthma, and targeting it for asthma treatment with interleukin-4 receptor antagonists. Respir Res 2:66–70. https://doi.org/10.1186/rr40
Takahashi K, Taniguchi M, Fukutomi Y et al (2014) Oral mite anaphylaxis caused by mite-contaminated okonomiyaki/pancake-mix in Japan: 8 case reports and a review of 28 reported cases. Allergol Int 63:51–56. https://doi.org/10.2332/allergolint.13-OA-0575
Taylor A, Verhagen J, Blaser K et al (2006) Mechanisms of immune suppression by interleukin-10 and transforming growth factor-β: the role of T regulatory cells. Immunology 117:433–442. https://doi.org/10.1111/j.1365-2567.2006.02321.x
Thomas WR, Smith WA, Hales BJ (2004) The allergenic specificities of the house dust mite. Chang Gung Med J 27:563–569
Tsai JJ, Liao EC, Tsai FH et al (2009) The effect of local nasal immunotherapy in allergic rhinitis: using strips of the allergen Dermatophagoides pteronyssinus. J Asthma 46:165–170. https://doi.org/10.1080/02770900802553110
Valerio C, Arlian L, Slater J (2009) Bacterial DNA sequences isolated from standardized dust mite extracts and wild mites. J Allergy Clin Immunol 123:S216. https://doi.org/10.1016/j.jaci.2008.12.826
von Mutius E (2010) Epidemiology of allergic diseases. Pediatr Allergy 1:1–8. https://doi.org/10.1016/B978-1-4377-0271-2.00001-8
Wang H, Ng TB, Ooi VE (1998) Lectins from mushrooms. Mycol Res 102:897–906. https://doi.org/10.1017/S0953756298006200
Wang PH, Hsu CI, Tang SC et al (2004) Fungal immunomodulatory protein from Flammulina velutipes induces interferon-γ production through p38 mitogen-activated protein kinase signaling pathway. J Agric Food Chem 52:2721–2725. https://doi.org/10.1021/jf034556s
Wasser SP (2010) Medicinal mushroom science: history, current status, future trends, and unsolved problems. Int J Med Mushrooms 12:1–16. https://doi.org/10.1615/IntJMedMushr.v12.i1.10
Wasser SP (2017) Medicinal mushrooms in human clinical studies. Part I. Anticancer, oncoimmunological, and immunomodulatory activities: a review. Int J Med Mushrooms 19:279–317. https://doi.org/10.1615/IntJMedMushrooms.v19.i4.10
Webb DC, McKenzie AN, Koskinen AM et al (2000) Integrated signals between IL-13, IL-4, and IL-5 regulate airways hyperreactivity. J Immunol 165:108–113. https://doi.org/10.4049/jimmunol.165.1.108
Wu CC, Liao EC, Lee MF et al (2009) Augmentation of regulatory T cells in allergic individuals by recombinant Der f 2 peptide with fungal immunomodulatory peptide fve. Ann Allergy Asthma Immunol 102:216–222. https://doi.org/10.1016/S1081-1206(10)60084-1
Xu X, Yan H, Chen J et al (2011) Bioactive proteins from mushrooms. Biotechnol Adv 29:667–674. https://doi.org/10.1016/j.biotechadv.2011.05.003
Yu SJ, Liao EC, Tsai JJ (2015) Effects of local nasal immunotherapy in allergic airway inflammation: using urea denatured Dermatophagoides pteronyssinus. Hum Vaccin Immunother 11:915–921. https://doi.org/10.1080/21645515.2015.1012029
Acknowledgements
This study was supported in part by the research grant (MOST109-2320-B-715-004) by the Ministry of Science and Technology, Taiwan, Republic of China. This project was also supported by the grants (MMC-RD-1091-B25 and MMC-RD-110-1E-P004) from MacKay Medical College, and by the grant (MMH-MM-10809) from the MacKay Memorial Hospital, New Taipei City, Taiwan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors have no commercial association with any sponsoring companies/organization that might pose a conflict of interest with the submitted manuscript. All authors have no financial and personal relationships with other people or organization that could influence this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Yen, CY., Yu, CH., Tsai, JJ. et al. Effects of Local Nasal Immunotherapy with FIP-fve Peptide and Denatured Tyrophagus putrescentiae for Storage Mite-Induced Airway Inflammation. Arch. Immunol. Ther. Exp. 70, 6 (2022). https://doi.org/10.1007/s00005-022-00645-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00005-022-00645-w