Abstract
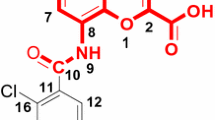
The three-dimensional (3D) quantitative structure-activity relationship method, comparative molecular similarity indices analysis (CoMSIA), was applied to a series of thioglitazones (or thiazolidinediones) to derive structure-activity relationships of their antihyperglycemic activity. Evaluation of 39 aligned compounds from the training set served to establish the various models. Predictions were obtained with CoMSIA steric, electrostatic, hydrophobic, and hydrogen bond acceptor and donor fields (q 2 = 0.51, r 2 = 0.942). The developed model was validated by an external test set of 11 compounds, with the satisfactory predictive r 2 value of 0.849. The information obtained from the present CoMSIA 3D contour map can be utilized for the design of specific PPAR-γ agonists.





Similar content being viewed by others
References
Chen HF, Li Q, Yao XJ, Fan BT, Yuan SG, Panaye A, Doucet JP (2003) 3D-QSAR and docking study of the binding mode of steroids to progesterone receptor in active site. QSAR Comb Sci 22:604–613
Chen HF, Li Q, Yao XJ, Fan BT, Yuan SG, Panaye A, Doucet JP (2004) CoMFA/CoMSIA/HQSAR and docking study of the binding mode of selective cyclooxygenase (Cox-2) inhibitors. QSAR Comb Sci 23:36–55
Clark M, Cramer RD III, Van Opdenbosch N (1989) Validation of the general purpose Tripos 5.2 force-field. J Comput Chem 10:982–1012
Clark M, Cramer RD III, Jones DM, Patterson DE, Simeroth PE (1990) Comparitive molecular field analysis (CoMFA). Toward its use with 3D-structural databases. Tetrahedron Comput Methodol 3:47–59
Cramer RD III, Bunce JD, Patterson DE, Frank LE (1988a) Crossvalidation, bootstrapping, and partial least squares compared with multiple regression in conventional QSAR studies. Quant Struct Act Relat 7:18–25
Cramer RD III, Patterson DE, Bunce JD (1988b) Comperative molecular field analysis (CoMFA): effect of shape on binding of steroids to carried protein. J Am Chem Soc 110:5959–5967
Delerive P, Fruchart J, Staels B (2001) Peroxisome proliferator-activated receptors in inflammation control. J Endocrinol 169:453–459
Evans RM, Barish GD, Wang YX (2004) PPARs and the complex journey to obesity. Nat Med 10:355–361
Gasteiger J, Marsili M (1980) Iterative partial equalization of orbital electronegativity a rapid access to atomic charges. Tetrahedron 36:3219–3228
James PT (2004) Obesity: the worldwide epidemic. Clin Dermatol 22:276–280
Kahn SE, Hull RL, Utzschneider (2006) Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444:840–846
Kanoh Y, Bandyopadhyay G, Sajan MP, Standaert ML, Farese RV (2000) Thiazolidinedione treatment enhances insulin effects on protein kinase C-zetz/lambda activation and glucose transport in adipocytes of nondiabetic and goto-kakizaki type II diabetic rats. J Biol Chem 275:16690–16696
Martens FM, Visseren FL, Lemay J, de Koning EJ, Rabelink TJ (2002) Metabolic and additional vascular effects of thiazolidinediones. Drugs 62:1463–1480
Miyazaki Y, Mahankali A, Matsuda M, Glass L, Mahankali S, Ferrannini E, Cusi K, Mandarino LJ, DeFronzo RA (2001) Improved glycemic control and enhanced insulin sensitivity in type 2 diabetic subjects treated with pioglitazone. Diabetes Care 24:710–719
Prashantha Kumar BR, Nanjan MJ (2008) QSAR study on thiazolidine-2,4-dione derivatives for their antihyperglycemic activity. Indian J Pharm Sci 70(5):565–571
Prashantha Kumar BR, Praveen TK, Nanjan MJ, Karvekar MD, Suresh B (2007) Serum glucose and triglyceride lowering activity of some novel glitazones against dexamethasone-induced hyperlipidemia and insulin resistance. Indian J Pharmacol 39:299–302
Prashantha Kumar BR, Bijoy JD, Jensen V, Praveen TK, Suresh B, Nanjan MJ (2008) CoMFA Study on thiazolidine-2,4-diones for their antihyperglycemic activity. Lett Drug Design Discov 5:79–87
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC (1998) Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 98:2088–2093
Standaert ML, Kanoh Y, Sajan MP, Bandyopadhyay G, Farese RV (2002) Cbl, IRS-1, and IRS-2 Mediate effects of rosiglitazone on PI3 K, PKC-lambda, and glucose transport in 3T3/L1 adipocytes. Endocrinology 143(5):1705–1716
Takashi S, Katsutoshi M, Eiko I, Yasuo S, Takeshi F, Yutaka K (1982) Studies on antidiabetic agents. II. Synthesis of 5-[4-(1-methylecyclohexylmethoxy)-benzyl]-thiazolidin-2,4-dione (ADD-3878) and its derivatives. Chem Pharm Bull 30(10):3580–3600
Weinstein SP, Holland A, O’Boyle E, Haber RS (1993) Effects of thiazolidinediones on glucocorticoid-induced insulin resistance and GLUT4 glucose transporter expression in rat skeletal muscle. Metabolism 42:1365–1369
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Prashantha Kumar, B.R., Nanjan, M.J. Comparative molecular similarity indices analysis for predicting the antihyperglycemic activity of thioglitazones. Med Chem Res 19, 1000–1010 (2010). https://doi.org/10.1007/s00044-009-9246-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-009-9246-0