Abstract
Purpose
Infections with Enterohaemorrhagic Escherichia coli typically occur in children causing haemolytic uraemic syndrome (HUS) and neurological symptoms in 20–50 %. Little information is available on the morphology of brain manifestations in adults. The purpose of this study was to identify a characteristic magnetic resonance imaging (MRI) pattern during the outbreak of a novel mutation of Escherichia coli O104:H4.
Methods
Patients were recruited from two hospitals between May and July 2011. The MRI protocol included standard anatomical, diffusion-weighted, and susceptibility-sensitive sequences.
Results
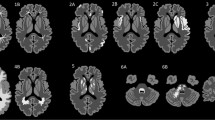
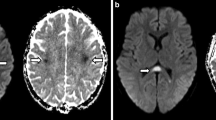
A total of 104 MRIs of 57 (32 female, 25 male) patients (mean 45.5 ± 18.4 years) showed abnormal signal intensity on 51 MRIs (49 %). Bilateral thalamus (39 %), bilateral pons (35 %), centrum semiovale and splenium of corpus callosum (33 %) were most often involved. Acute lesions were reversible in 81 % of cases. There was no statistically significant association between symptom onset and the MRI findings (P = 0.2).
Conclusions
Neuroimaging findings in this adult patient cohort were non-specific and similar to previous findings in children. A characteristic neuroimaging pattern of an infection with Escherichia coli O104:H4 was not identified. However, bilateral symmetric T2 hyperintense lesions of the thalami and dorsal pons characterized by restricted diffusion suggest a metabolic toxic effect of the disease on the brain.






Similar content being viewed by others
References
Bale JF Jr., Brasher C, Siegler RL. CNS manifestations of the haemolytic uraemic syndrome. Relationship to metabolic alterations and prognosis. Am J Dis Child. 1980;134(9):869–72.
Eriksson KJ, Boyd SG, Tasker RC. Acute neurology and neurophysiology of haemolytic uraemic syndrome. Arch Dis Child. 2001;84(5):434–5.
Sheth KJ, Swick HM, Haworth N. Neurological involvement in haemolytic uraemic syndrome. Ann Neurol. 1986;19(1):90–3.
Ellchuk TN, Shah LM, Hewlett RH, Osborn AG. Suspicious neuroimaging pattern of thrombotic micro-angiopathy. AJNR Am J Neuroradiol. 2011;32(4):734–8.
Nathanson S, Kwon T, Elmaleh M, Charbit M, Launay EA, Harambat J, et al. Acute neurological involvement in diarrhoea-associated haemolytic uraemic syndrome. Clin J Am Soc Nephrol. 2010;5(7):1218–28.
Magnus T, Rother J, Simova O, Meier-Cillien M, Repenthin J, Moller F, et al. The neurological syndrome in adults during the 2011 northern German E. coli serotype O104:H4 outbreak. Brain. 2012;135(6):1850–9.
Ahrens F, Ludwig K, Terstegge K, Querfeld U. Encephalopathy and exposure to Shiga toxin without evidence of haemolytic uraemic syndrome. Eur J Pediatr. 2002;161(8):462–3.
Goldstein J, Loidl CF, Creydt VP, Boccoli J, Ibarra C. Intracerebroventricular administration of Shiga toxin type 2 induces striatal neuronal death and glial alterations: an ultrastructural study. Brain Res. 2007;1161:106–15.
Garcia-Monco JC, Cortina IE, Ferreira E, Martinez A, Ruiz L, Cabrera A, et al. Reversible splenial lesion syndrome (RESLES): what’s in a name? J Neuroimaging. 2011;21(2):e1–14.
Ogura H, Takaoka M, Kishi M, Kimoto M, Shimazu T, Yoshioka T, et al. Reversible MR findings of haemolytic uraemic syndrome with mild encephalopathy. AJNR Am J Neuroradiol. 1998;19(6):1144–5.
Steinborn M, Leiz S, Rudisser K, Griebel M, Harder T, Hahn H. CT and MRI in haemolytic uraemic syndrome with central nervous system involvement: distribution of lesions and prognostic value of imaging findings. Pediatr Radiol. 2004;34(10):805–10.
Nakamura H, Takaba H, Inoue T, Saku Y, Saito F, Ibayashi S, et al. MRI findings of haemolytic uraemic syndrome with encephalopathy: widespread symmetrical distribution. J Neuroimaging. 2003;13(1):75–8.
Meuth SG, Gobel K, Kanyshkova T, Ehling P, Ritter MA, Schwindt W, et al. Thalamic involvement in patients with neurologic impairment due to Shiga toxin 2. Ann Neurol. 2013;73(3):419–29. doi:10.1002/ana.23814.
Wengenroth M, Hoeltje J, Repenthin J, Meyer TN, Bonk F, Becker H, et al. Central nervous system involvement in adults with epidemic haemolytic uraemic syndrome. AJNR Am J Neuroradiol. 2013;34(5):1016–21. doi:10.3174/ajnr.A3336.
Weissenborn K, Donnerstag F, Kielstein JT, Heeren M, Worthmann H, Hecker H, et al. Neurologic manifestations of E coli infection-induced haemolytic uraemic syndrome in adults. Neurology. 2012;79(14):1466–73. doi:10.1212/WNL.0b013e31826d5f26.
Gallo EG, Gianantonio CA. Extrarenal involvement in diarrhoea-associated haemolytic uraemic syndrome. Pediatr Nephrol. 1995;9(1):117–9.
Barnett ND, Kaplan AM, Bernes SM, Cohen ML. Haemolytic uraemic syndrome with particular involvement of basal ganglia and favourable outcome. Pediatr Neurol. 1995;12(2):155–8.
Schmidt S, Gudinchet F, Meagher-Villemure K, Maeder P. Brain involvement in haemolytic uraemic syndrome: MRI features of coagulative necrosis. Neuroradiology. 2001;43(7):581–5.
Hamano S. Neurological manifestation of haemorrhagic colitis in the outbreak of Escherichia coli O157:H7 infection in Japan. Acta Paedatr. 1993;82(5):454–8.
Karch H, Bockemühl J, Huppertz H-I. Erkrankungen durch enterohämorrhagische Escherichia coli (EHEC). Dtsch Arztebl. 2000;97(36):A2314–38.
Karch H, Schmidt H, Brunder W. Plasmid-encoded determinants of Escherichia coli O157:H7. In: Kaper JB, O’Brian AD, editors. Escherichia Coli O157: H7 and other Shiga Toxin-producing E. Coli strains. Washington, D.C.: American Society for Microbiology; 1998. pp. 183–94.
Steele BT, Murphy N, Chuang SH, McGreal D, Arbus GS. Recovery from prolonged coma in haemolytic uraemic syndrome.J Pediatr. 1983;102(3):402–4.
Jeong YK, Kim IO, Kim WS, Hwang YS, Choi Y, Yeon KM. Haemolytic uraemic syndrome: MR findings of CNS complications. Pediatr Radiol. 1994;24(8):585–6.
Vergouwen MD, Adriani KS, Roos YB, Groothoff JW, Majoie CB. Proximal cerebral artery stenosis in a patient with haemolytic uraemic syndrome. AJNR Am J Neuroradiol. 2008;29(5):e34.
Mizuguchi M, Tanaka S, Fujii I, Tanizawa H, Suzuki Y, Igarashi T, et al. Neuronal and vascular pathology produced by verocytotoxin 2 in the rabbit central nervous system. Acta neuropathologica. 1996;91(3):254–62.
Conflict of Interest
The authors declare that there are no actual or potential conflicts of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Löbel, U., Eckert, B., Simova, O. et al. Cerebral Magnetic Resonance Imaging Findings in Adults with Haemolytic Uraemic Syndrome Following an Infection with Escherichia coli, Subtype O104:H4. Clin Neuroradiol 24, 111–119 (2014). https://doi.org/10.1007/s00062-013-0231-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-013-0231-0