Abstract
Purpose
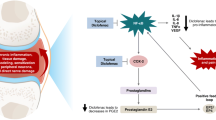
The present study was designed to evaluate the penetration of diclofenac sodium 4 % spray gel in synovial tissue, synovial fluid and blood plasma after topical application in subjects with joint effusions and planned total knee arthroplasty (TKA) due to osteoarthritis.
Methods
A total of 39 patients were randomised to two- or three-times daily application of diclofenac sodium 4 % spray gel to knees requiring surgery over a treatment period of 3 days. Within 8 h after the last application, TKA was conducted, and the diclofenac concentrations in synovial tissue, synovial fluid and blood plasma were measured by liquid chromatography.
Results
The median diclofenac concentration was approximately 10–20-fold higher in synovial tissue (36.2 and 42.8 ng/g) than in synovial fluid (2.6 and 2.8 ng/mL) or plasma (3.9 and 4.1 ng/mL) in both treatment groups. Dose proportionality for any compartment or treatment groups could not be detected. Treatment-related adverse events were noted in two cases and limited to skin reactions.
Conclusion
Diclofenac sodium 4 % spray gel was found to penetrate the skin locally in substantial amounts and thus reach the desired target tissue. Concentrations were not dose-dependent, and application was well tolerated by 97.4 % of patients. Topical application of diclofenac should be considered a valuable alternative to systemic NSAID therapy in the initial treatment of osteoarthritis.

Similar content being viewed by others
References
Arcangeli P, Andreotti L, Palazzini E (1996) Effective treatment of osteoarthritis with a 150 mg prolonged-release of diclofenac sodium. Riv Eur Sci Med Farmacol 18(5–6):217–223
Barron MC, Rubin BR (2007) Managing osteoarthritic knee pain. J Am Osteopath Assoc 107(10 Suppl 6):21–27
Benson MD, Aldo-Benson M, Brandt KD (1985) Synovial fluid concentrations of diclofenac in patients with rheumatoid arthritis or osteoarthritis. Semin Arthritis Rheum 15(2 Suppl 1):65–67
Brune K (2007) Persistence of NSAIDs at effect sites and rapid disappearance from side effect compartments contributes to tolerability. Curr Med Res Opin 23(12):2985–2995
Brunner M, Dehghanyar P, Seigfried B, Martin W, Menke G, Muller M (2005) Favourable dermal penetration of diclofenac after administration to the skin using a novel spray gel formulation. Br J Clin Pharmacol 60(5):573–577
Courtney P, Doherty M (2002) Key questions concerning paracetamol and NSAIDs for osteoarthritis. Ann Rheum Dis 61(9):767–773
Davies NM, Anderson KE (1997) Clinical pharmacokinetics of diclofenac. Therapeutic insights and pitfalls. Clin Pharmacokinet 33(3):184–213
Elmquist WF, Chan KK, Sawchuk RJ (1994) Transsynovial drug distribution: synovial mean transit time of diclofenac and other nonsteroidal antiinflammatory drugs. Pharm Res 11(12):1689–1697
Escribano E, Calpena AC, Queralt J, Obach R, Domenech J (2003) Assessment of diclofenac permeation with different formulations: anti-inflammatory study of a selected formula. Eur J Pharm Sci 19(4):203–210
Felson DT, Neogi T (2004) Osteoarthritis: is it a disease of cartilage or of bone? Arthritis Rheum 50(2):341–344
Fowler PD, Shadforth MF, Crook PR, John VA (1983) Plasma and synovial fluid concentrations of diclofenac sodium and its major hydroxylated metabolites during long-term treatment of rheumatoid arthritis. Eur J Clin Pharmacol 25(3):389–394
Hamad M, Metwally S, El-Shafey A, Geneidi A (1994) Comparative percutaneous absorption of diclofenac emulgel penetrations in normal volunteers. J Drug Res 21:133–141
Henry D, Lim LL, Rodriguez LAG, Gutthann SP, Carson JL, Griffin M, Savage R, Logan R, Moride Y, Hawkey C, Hill S, Fries JT (1996) Variability in risk of gastrointestinal complications with individual non-steroidal anti-inflammatory drugs: results of a collaborative meta-analysis. BMJ 312(7046):1563–1566
Jordan KM, Arden NK, Doherty M, Bannwarth B, Bijlsma JW, Dieppe P, Gunther K, Hauselmann H, Herrero-Beaumont G, Kaklamanis P, Lohmander S, Leeb B, Lequesne M, Mazieres B, Martin-Mola E, Pavelka K, Pendleton A, Punzi L, Serni U, Swoboda B, Verbruggen G, Zimmerman-Gorska I, Dougados M (2003) EULAR Recommendations 2003: an evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann Rheum Dis 62(12):1145–1155
Liauw HL, Ku E, Brandt KD, Benson MD, Aldo-Benson MA, Waiter SL, Lee W, Chan K, Vyas K (1985) Effects of Voltaren on arachidonic acid metabolism in arthritis patients. Agents Actions Suppl 17:195–199
Mason L, Moore RA, Edwards JE, Derry S, McQuay HJ (2004) Topical NSAIDs for acute pain: a meta-analysis. BMC Fam Pract 5:10–18
Mason L, Moore RA, Edwards JE, Derry S, McQuay HJ (2004) Topical NSAIDs for chronic musculoskeletal pain: systematic review and meta-analysis. BMC Musculoskelet Disord 5:28–35
Miyatake S, Ichiyama H, Kondo E, Yasuda K (2008) Randomized clinical comparisons of diclofenac concentration in the soft tissues and blood plasma between topical and oral applications. BJCP 67(1):125–129
Moore RA (2004) Topical nonsteroidal antiinflammatory drugs are effective in osteoarthritis of the knee. J Rheumatol 31(10):1893–1895
Moore RA, Tramer MR, Carroll D, Wiffen PJ, McQuay HJ (1998) Quantitative systematic review of topically applied non-steroidal anti-inflammatory drugs. BMJ 316(7128):333–338
Nishihata T, Kotera K, Nakano Y, Yamazaki M (1987) Rat percutaneous transport of diclofenac and influence of hydrogenated soya phospholipids. Chem Pharm Bull (Tokyo) 35(9):3807–3812
Radermacher J, Jentsch D, Scholl MA, Lustinetz T, Frolich JC (1991) Diclofenac concentrations in synovial fluid and plasma after cutaneous application in inflammatory and degenerative joint disease. Br J Clin Pharmacol 31(5):537–541
Riess W, Schmid K, Botta L, Kobayashi K, Moppert J, Schneider W, Sioufi A, Strusberg A, Tomasi M (1986) The percutaneous absorption of diclofenac. Arzneimittelforschung 36(7):1092–1096
Sherertz EF, Sloan KB, McTiernan RG (1987) Use of theoretical partition coefficients determined from solubility parameters to predict permeability coefficients for 5-fluorouracil. J Invest Dermatol 89(2):147–151
Taylor RS, Fotopoulos G, Maibach H (2011) Safety profile of topical diclofenac: a meta analysis of blinded, randomized, controlled trials in musculoskeletal conditions. Curr Med Res Opin 27(3):605–622
Zhang W, Doherty M, Leeb BF, Alekseeva L, Arden NK, Bijlsma JW, Dincer F, Dziedzic K, Hauselmann HJ, Herrero-Beaumont G, Kaklamanis P, Lohmander S, Maheu E, Martin-Mola E, Pavelka K, Punzi L, Reiter S, Sautner J, Smolen J, Verbruggen G, Zimmermann-Gorska I (2007) EULAR evidence based recommendations for the management of hand osteoarthritis: report of a Task Force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 66(3):377–388
Zhang W, Moskowitz RW, Nuki G, Abramson S, Altman RD, Arden N, Bierma-Zeinstra S, Brandt KD, Croft P, Doherty M, Dougados M, Hochberg M, Hunter DJ, Kwoh K, Lohmander LS, Tugwell P (2007) OARSI recommendations for the management of hip and knee osteoarthritis, part I: critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthr Cartil 15(9):981–1000
Acknowledgments
The authors thank Kate McFarlane for her support in preparing the manuscript and proofreading.
Conflict of interest
T.E. and M.D.S. are consultants to Smith and Nephew, Endoscopy. The study was supported by MIKA Pharma.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Efe, T., Sagnak, E., Roessler, P.P. et al. Penetration of topical diclofenac sodium 4 % spray gel into the synovial tissue and synovial fluid of the knee: a randomised clinical trial. Knee Surg Sports Traumatol Arthrosc 22, 345–350 (2014). https://doi.org/10.1007/s00167-013-2408-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-013-2408-0