Abstract
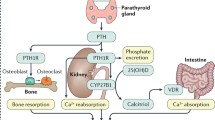
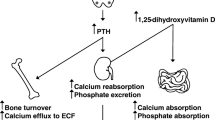
Hypoparathyroidism is characterized by hypocalcemia with inappropriately low parathyroid hormone (PTH) levels. Bone turnover is abnormally low and bone mineral density (BMD) is typically increased. Plasma calcium levels can be normalized by treatment with calcium supplements and vitamin D analogs, but bone turnover remains low and patients complain of a reduced quality of life (QoL). During recent years, a number of studies have shown that PTH replacement therapy (PTH-RT) may maintain calcium levels within the normal range, while the need for calcium and vitamin D supplements is reduced. In the initial response to subcutaneous PTH injections once or twice daily, bone turnover is overstimulated. BMD increases in cancellous bone, but decreases in cortical bone due to an increased porosity. Microcomputed tomography scans and histomorphometric studies on bone biopsies have shown changes similar to the well-known bone anabolic effects of PTH treatment in osteoporosis rather than a normalization of bone remodeling balancing the anabolic and catabolic effects of PTH. Most recently, continuous PTH delivery by pump was shown to increase the levels of bone markers into the normal range (without overstimulation of bone turnover) and with a normalization of renal calcium excretion. As PTH has a short plasma half-life, these findings indicate that exposure to PTH once or twice daily is not sufficient to reestablish a calcium homeostasis and bone metabolism that resembles normal physiology. Further studies should assess the effects of continuous PTH exposure by pump delivery (or multiple daily injections) on BMD and bone histology, as well as the effects of PTH-RT on indices of QoL.
Similar content being viewed by others
References
Rosato L, Avenia N, Bernante P, Palma M, Gulino G, Nasi PG, Pelizzo MR, Pezzullo L (2004) Complications of thyroid surgery: analysis of a multicentric study on 14,934 patients operated on in Italy over 5 years. World J Surg 28:271–276
Youngwirth L, Benavidez J, Sippel R, Chen H (2010) Parathyroid hormone deficiency after total thyroidectomy: incidence and time. J Surg Res 163:69–71
Bergenfelz A, Jansson S, Mårtensson H, Reihnér E, Wallin G, Kristoffersson A, Lausen I (2007) Scandinavian quality register for thyroid and parathyroid surgery: audit of surgery for primary hyperparathyroidism. Langenbecks Arch Surg 392:445–451
Mittendorf EA, Merlino JI, McHenry CR (2004) Post-parathyroidectomy hypocalcemia: incidence, risk factors, and management. Am Surg 70:114–119
Bilezikian JP, Khan A, Potts JT, Brandi ML, Clarke BL, Shoback D, Juppner H, D’Amour P, D’Amour P, D’Amour P, Fox J, Rejnmark L, Rejnmark L, Mosekilde L, Rubin MR, Dempster D, Dempster D, Gafni R, Collins MT, Collins MT, Sliney J, Sanders J (2011) Hypoparathyroidism in the adult: epidemiology, diagnosis, pathophysiology, target organ involvement, treatment, and challenges for future research. J Bone Miner Res 26:2317–2337
Pollak MR, Brown EM, Estep HL, McLaine PN, Kifor O, Park J, Hebert SC, Seidman CE, Seidman JG (1994) Autosomal dominant hypocalcaemia caused by a Ca(2+)-sensing receptor gene mutation. Nat Genet 8:303–307
Baron J, Winer KK, Yanovski JA, Cunningham AW, Laue L, Zimmerman D, Cutler GB Jr (1996) Mutations in the Ca(2+)-sensing receptor gene cause autosomal dominant and sporadic hypoparathyroidism. Hum Mol Genet 5:601–606
Christensen SE, Nissen PH, Vestergaard P, Mosekilde L (2011) Familial hypocalciuric hypercalcaemia: a review. Curr Opin Endocrinol Diabetes Obes 18:359–370
Watanabe S, Fukumoto S, Chang H, Takeuchi Y, Hasegawa Y, Okazaki R, Chikatsu N, Fujita T (2002) Association between activating mutations of calcium-sensing receptor and Bartter’s syndrome. Lancet 360:692–694
Mundy GR, Guise TA (1999) Hormonal control of calcium homeostasis. Clin Chem 45:1347–1352
Shoback D (2008) Hypoparathyroidism. N Engl J Med 359:391–403
Santos F, Smith MJV, Chan JCM (1986) Hypercalciuria associated with long-term administration of calcitriol (1,25-dihydroxyvitamin D3) action of hydrochlorothiazide. Am J Dis Child 140:139–142
Sato K, Hasegawa Y, Nakae J, Nanao K, Takahashi I, Tajima T, Shinohara N, Fujieda K (2002) Hydrochlorothiazide effectively reduces urinary calcium excretion in two Japanese patients with gain-of-function mutations of the calcium-sensing receptor gene. J Clin Endocrinol Metab 87:3068–3073
Uncini A, Tartaro A, Di SE, Gambi D (1985) Parkinsonism, basal ganglia calcification and epilepsy as late complications of postoperative hypoparathyroidism. J Neurol 232:109–111
Forman MB, Sandler MP, Danziger A, Kalk WJ (1980) Basal ganglia calcification in postoperative hypoparathyroidism. Clin Endocrinol (Oxf) 12:385–390
Langdahl BL, Mortensen L, Vesterby A, Eriksen EF, Charles P (1996) Bone histomorphometry in hypoparathyroid patients treated with vitamin D. Bone 18:103–108
Rubin MR, Dempster DW, Kohler T, Stauber M, Zhou H, Shane E, Nickolas T, Stein E, Sliney J, Silverberg SJ, Bilezikian JP, Müller R (2010) Three dimensional cancellous bone structure in hypoparathyroidism. Bone 46:190–195
Compston J (2011) Pathophysiology of atypical femoral fractures and osteonecrosis of the jaw. Osteoporos Int 22:2951–2961
Subramanian G, Fritton J, Quek S (2012) Osteonecrosis and atypical fractures—common origins? Osteoporos Int. doi:10.1007/s00198‐012‐1953‐6
Wang L, Manson JE, Song Y, Sesso HD (2010) Systematic review: vitamin D and calcium supplementation in prevention of cardiovascular events. Ann Intern Med 152:315–323
Zittermann A (2006) Vitamin D, and disease prevention with special reference to cardiovascular disease. Prog Biophys Mol Biol 92:39–48
Shoji T, Shinohara K, Kimoto E, Emoto M, Tahara H, Koyama H, Inaba M, Fukumoto S, Ishimura E, Miki T, Tabata T, Nishizawa Y (2004) Lower risk for cardiovascular mortality in oral 1{alpha}-hydroxy vitamin D3 users in a haemodialysis population. Nephrol Dial Transplant 19:179–184
Murray TM, Rao LG, Divieti P, Bringhurst FR (2005) Parathyroid hormone secretion and action: evidence for discrete receptors for the carboxyl-terminal region and related biological actions of carboxyl-terminal ligands. Endocr Rev 26:78–113
Gardella TJ, Jüppner H (2001) Molecular properties of the PTH/PTHrP receptor. Trends Endocrinol Metab 12:210–217
Velasco PJ, Manshadi M, Breen K, Lippmann S (1999) Psychiatric aspects of parathyroid disease. Psychosomatics 40:486–490
Arlt W, Fremerey C, Callies F, Reincke M, Schneider P, Timmermann W, Allolio B (2002) Well-being, mood and calcium homeostasis in patients with hypoparathyroidism receiving standard treatment with calcium and vitamin D. Eur J Endocrinol 146:215–222
Winer KK, Yanovski JA, Cutler GB Jr (1996) Synthetic human parathyroid hormone 1–34 vs calcitriol and calcium in the treatment of hypoparathyroidism. JAMA 276:631–636
Winer KK, Yanovski JA, Sarani B, Cutler GB Jr (1998) A randomized, cross-over trial of once-daily versus twice-daily parathyroid hormone 1–34 in treatment of hypoparathyroidism. J Clin Endocrinol Metab 83:3480–3486
Winer KK, Ko CW, Reynolds JC, Dowdy K, Keil M, Peterson D, Gerber LH, McGarvey C, Cutler GB Jr (2003) Long-term treatment of hypoparathyroidism: a randomized controlled study comparing parathyroid hormone-(1–34) versus calcitriol and calcium. J Clin Endocrinol Metab 88:4214–4220
Winer KK, Sinaii N, Peterson D, Sainz B Jr, Cutler GB Jr (2008) Effects of once versus twice-daily parathyroid hormone1–34 therapy in children with hypoparathyroidism. J Clin Endocrinol Metab 93:3389–3395
Winer KK, Sinaii N, Reynolds J, Peterson D, Dowdy K, Cutler GB Jr (2010) Long-term treatment of 12 children with chronic hypoparathyroidism: a randomized trial comparing synthetic human parathyroid hormone 1–34 versus calcitriol and calcium. J Clin Endocrinol Metab 95:2680–2688
Winer KK, Zhang B, Shrader JA, Peterson D, Smith M, Albert PS, Cutler GB (2012) Synthetic human parathyroid hormone 1–34 replacement therapy: a randomized crossover trial comparing pump versus injections in the treatment of chronic hypoparathyroidism. J Clin Endocrinol Metab 97:391–399
Gafni RI, Brahim JS, Andreopoulou P, Bhattacharyya N, Kelly MH, Brillante BA, Reynolds JC, Zhou H, Dempster DW, Collins MT (2012) Daily parathyroid hormone 1–34 replacement therapy for hypoparathyroidism induces marked changes in bone turnover and structure. J Bone Miner Res 27:1811–1820
Rubin MR, Dempster DW, Sliney J, Zhou H, Nickolas TL, Stein EM, Dworakowski E, Dellabadia M, Ives R, McMahon DJ, Zhang C, Silverberg SJ, Shane E, Cremers S, Bilezikian JP (2011) PTH(1–84) administration reverses abnormal bone-remodeling dynamics and structure in hypoparathyroidism. J Bone Miner Res 26:2727–2736
Sikjaer T, Rejnmark L, Rolighed L, Heickendorff L, Mosekilde L, The Hypoparathyroid Study Group (2011) The effect of adding PTH (1–84) to conventional treatment of hypoparathyroidism—a randomized, placebo controlled study. J Bone Miner Res 26:2358–2370
Sikjaer T, Rejnmark L, Thomsen JS, Tietze A, Brüel A, Andersen G, Mosekilde L (2012) Changes in 3-dimensional bone structure indices in hypoparathyroid patients treated with PTH(1–84): a randomized controlled study. J Bone Miner Res 27:781–788
Rubin M, Sliney J, McMahon D, Silverberg S, Bilezikian J (2010) Therapy of hypoparathyroidism with intact parathyroid hormone. Osteoporos Int 21:1927–1934
Jiang Y, Zhao JJ, Mitlak BH, Wang O, Genant HK, Eriksen EF (2003) Recombinant human parathyroid hormone (1–34) [teriparatide] improves both cortical and cancellous bone structure. J Bone Miner Res 18:1932–1941
Jobke B, Muche B, Burghardt A, Semler J, Link T, Majumdar S (2011) Teriparatide in bisphosphonate-resistant osteoporosis: microarchitectural changes and clinical results after 6 and 18 months. Calcif Tissue Int 89:130–139
Jobke B, Pfeifer M, Minne HW (2009) Teriparatide following bisphosphonates: initial and long-term effects on microarchitecture and bone remodeling at the human iliac crest. Connect Tissue Res 50:46–54
Recker RR, Bare SP, Smith SY, Varela A, Miller MA, Morris SA, Fox J (2009) Cancellous and cortical bone architecture and turnover at the iliac crest of postmenopausal osteoporotic women treated with parathyroid hormone 1–84. Bone 44:113–119
Cosman F, Schnitzer MB, McCann PD, Parisien MV, Dempster DW, Lindsay R (1992) Relationships between quantitative histological measurements and noninvasive assessments of bone mass. Bone 13:237–242
Nottestad SY, Baumel JJ, Kimmel DB, Recker RR, Heaney RP (1987) The proportion of trabecular bone in human vertebrae. J Bone Miner Res 2:221–229
Watanabe A, Yoneyama S, Nakajima M, Sato N, Takao-Kawabata R, Isogai Y, Sakurai-Tanikawa A, Higuchi K, Shimoi A, Yamatoya H, Yoshida K, Kohira T (2012) Osteosarcoma in Sprague–Dawley rats after long-term treatment with teriparatide (human parathyroid hormone (1–34)). J Toxicol Sci 37:617–629
Jolette J, Wilker CE, Smith SY, Doyle N, Hardisty JF, Metcalfe AJ, Marriott TB, Fox J, Wells DS (2006) Defining a noncarcinogenic dose of recombinant human parathyroid hormone 1–84 in a 2-year study in Fischer 344 rats. Toxicol Pathol 34:929–940
Karsdal MA, Qvist P, Christiansen C, Tankó LB (2006) Optimising antiresorptive therapies in postmenopausal women: why do we need to give due consideration to the degree of suppression? Drugs 66:1909–1918
Sikjaer T, Rejnmark L, Mosekilde L (2011) PTH treatment in hypoparathyroidism. Curr Drug Saf 6:89–99
Conflicts of interest
Leif Mosekilde is a principal investigator on the REPLACE study initiated by NPS Pharmaceuticals. All other authors state that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rejnmark, L., Sikjaer, T., Underbjerg, L. et al. PTH replacement therapy of hypoparathyroidism. Osteoporos Int 24, 1529–1536 (2013). https://doi.org/10.1007/s00198-012-2230-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-2230-4