Abstract
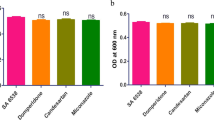
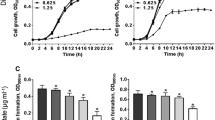
Staphylococcus aureus is an opportunistic pathogen that has the ability to cause a wide range of diseases including superficial infection and severe invasive life threatening infections. The pathogenicity of S. aureus is mediated by a group of virulence factors that mediate the colonization and penetration. The antibiotic resistance of S. aureus has evolved due to the abuse of antibiotics rendering the cure of infection very difficult especially with the shortage in new antibiotic production. To combat this shortage, repurposing of FDA-approved drugs against the virulence factors is a new strategy. The analgesic drug Diclofenac was found to have anti-virulence activity against Pseudomonas aeruginosa and Proteus mirabilis. This study aimed to demonstrate the anti-virulence effect of diclofenac against clinical MRSA isolates phenotypically and genotypically using qRT-PCR. In this study, diclofenac showed significant reduction in biofilm formation when compared to controls, the inhibition ranged between 22.67% and 70%. Also, remarkable inhibition of hemolysin activity was found (5.4–66.34%). Additionally, diclofenac has inhibitory activity against the staphyloxanthin production (8–57.2%). The results were confirmed by qRT-PCR that showed significant down-regulation of tested virulence genes. The down-regulation ranged from 43 to 64.05% for SarA, 36.85–64.75% for AgrA, 50–63.2% for hla, 38.55–60.35% for FnbA, 46.75–61.05% for IcaA, 27.55–64% for SigB and 51.05–72.8% for CrtM. In conclusion, diclofenac can be used in combination with antibiotics as anti-virulence agent against MDR-MRSA which will enhance the ability of immune system to eradicate infection.





Similar content being viewed by others
References
Abbas H (2015a) Inhibition of virulence factors of Pseudomonas aeruginosa by diclofenac sodiumn. Roum Arch Microbiol Immunol 74:79–85
Abbas H (2015b) Inhibtion of virulence factors of Pseudomonas aeruginosa by diclofenac sodiumn. Roum Arch Microbiol Immunol 74:79–85
Abbas HA, Hegazy WAH (2020) Repurposing anti-diabetic drug “Sitagliptin” as a novel virulence attenuating agent in Serratia marcescens. PLoS ONE 15:e0231625
Abbas HAMAH, Elsherbini AM, Shaldam MA (2019) Glyceryl trinitrate blocks staphyloxanthin and biofilm formation in Staphylococcus aureus. Afr Health Sci 19:1376–1384. https://doi.org/10.4314/ahs.v19i1.10
Ahmed EF, Gad GFM, Abdalla AM et al (2014) Prevalence of methicillin resistant Staphylococcus aureus among egyptian patients after surgical interventions. Surg Infect (Larchmt) 15:404–411. https://doi.org/10.1089/sur.2013.212
Alavi M, Antonic V, Stojadinovic A et al (2013) Pseudomonas aeruginosa induces pigment production and enhances virulence in a white phenotypic variant of Staphylococcus aureus. Infect Drug Resist 6:175–186. https://doi.org/10.2147/IDR.S49039
Al-kazaz EJ, Melconian AK, Kandela NJ (2014) Extraction of staphyloxanthin from staphylococcus aureus isolated from clincal sources to determine its antibaterial ativity against other bacteria. Iraqi J Sci 55:1823–1832
Askoura M, Saleh M, Abbas H (2020) An innovative role for tenoxicam as a quorum sensing inhibitor in Pseudomonas aeruginosa. Arch Microbiol 202:555–565. https://doi.org/10.1007/s00203-019-01771-4
Bien J, Sokolova O, Bozko P (2011) Characterization of virulence Factors of Staphylococcus aureus: novel function of known virulence factors that are implicated in activation of airway epithelial proinflammatory response. J Pathog 2011:601905. https://doi.org/10.4061/2011/601905
Burnside K, Lembo A, de los Reyes M et al (2010) Regulation of hemolysin expression and virulence of Staphylococcus aureus by a serine/threonine kinase and phosphatase. PLoS One 5:e11071
Chambers HF, Deleo FR (2009) Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat Rev Microbiol 7:629–641. https://doi.org/10.1038/nrmicro2200
Chatterjee SS, Otto M (2013) Improved understanding of factors driving methicillin-resistant Staphylococcus aureus epidemic waves. Clin Epidemiol 5:205–217. https://doi.org/10.2147/CLEP.S37071
Chen F, Di H, Wang Y et al (2016) Small-molecule targeting of a diapophytoene desaturase inhibits S. aureus virulence. Nat Chem Biol 12:174
Clinical and Laboratory Standards Institute (CLSI) (2016) Performance standards for antimicrobial susceptibility testing; Twenty-sixth informational supplement. CLSI document M100-S26, Wayne, PA, USA
Craft KM, Nguyen JM, Berg LJ, Townsend SD (2019) Methicillin-resistant: Staphylococcus aureus (MRSA): antibiotic-resistance and the biofilm phenotype. Medchemcomm 10:1231–1241. https://doi.org/10.1039/c9md00044e
EI AilaAl Laham NANA, Ayesh BM (2017) Nasal carriage of methicillin resistant Staphylococcus aureus among health care workers at Al Shifa hospital in Gaza Strip. BMC Infect Dis 17:1–7. https://doi.org/10.1186/s12879-016-2139-1
El-Gayar MH, Aboulwafa MM, Aboshanab KM, Hassouna NAH (2014) Virulence characters of some methicillin resistant Staphylococcus aureus isolates. Arch Clin Microbiol 5:1–14. https://doi.org/10.3823/283
El-Mowafy SA, Abd El Galil KH, Habib ESE, Shaaban MI (2017) Quorum sensing inhibitory activity of sub-inhibitory concentrations of β-lactams. African Health Sciences 17(1):199–207
Foster TJ (2004) The Staphylococcus aureus “superbug”. J Clin Invest 114:1693–1696. https://doi.org/10.1172/JCI200422123.7
Gertz S, Engelmann S, Schmid R et al (2000) Characterization of the sigB Regulon in Staphylococcus aureus. J Bacteriol 182:6983–6991
Gordon CP, Williams P, Chan WC (2013) Attenuating Staphylococcus aureus virulence gene regulation: a medicinal chemistry perspective. J Med Chem 56:1389–1404. https://doi.org/10.1021/jm3014635
Gould IM, David MZ, Esposito S et al (2012) New insights into meticillin-resistant Staphylococcus aureus (MRSA) pathogenesis, treatment and resistance. Int J Antimicrob Agents 39:96–104. https://doi.org/10.1016/j.ijantimicag.2011.09.028
Guo Y, Song G, Sun M et al (2020) Prevalence and therapies of antibiotic-resistance in Staphylococcus aureus. Front Cell Infect Microbiol 10:107
Hendrix AS, Spoonmore TJ, Wilde AD et al (2016) Repurposing the nonsteroidal anti-inflammatory drug diflunisal as an osteoprotective, antivirulence therapy for Staphylococcus aureus osteomyelitis. Antimicrob Agents Chemother 60:5322–5330. https://doi.org/10.1128/AAC.00834-16
Ibrahim ES, El-Baghdady KZ, El-All A, Said M, Warda MA, Prince AM, Ibrahim MK (2020) Prevalence of multidrug resistance in the Egyptian methicillin-resistant Staphylococcus aureus isolates. African J Biol Sci 16:43–52. https://doi.org/10.1017/CBO9781107415324.004
Kane TL, Carothers KE, Lee SW (2018) Virulence factor targeting of the bacterial pathogen Staphylococcus aureus for vaccine and therapeutics. Curr Drug Targets 19:111–127. https://doi.org/10.2174/1389450117666161128123536
Khodaverdian V, Pesho M, Truitt B et al (2013) Discovery of antivirulence agents against methicillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother 57:3645–3652. https://doi.org/10.1128/AAC.00269-13
Koneman EW, Allen SD, Janda WM, Scheckenberger PC, Winn WC (2006) Color atlas and textbook of diagnostic microbiology, 6th edn. Lippincott, Philadelphia
Kong C, Chee CF, Richter K et al (2018) Suppression of Staphylococcus aureus biofilm formation and virulence by a benzimidazole derivative, UM-C162. Sci Rep 8:1–16. https://doi.org/10.1038/s41598-018-21141-2
Kot B, Sytykiewicz H, Sprawka I (2018) Expression of the biofilm-associated genes in methicillin-resistant Staphylococcus aureus in biofilm and planktonic conditions. Int J Mol Sci 19:3487–3500. https://doi.org/10.3390/ijms19113487
Latimer J, Forbes S, McBain AJ (2012) Attenuated virulence and biofilm formation in Staphylococcus aureus following sublethal exposure to triclosan. Antimicrob Agents Chemother 56:3092–3100. https://doi.org/10.1128/AAC.05904-11
Lee JH, Cho HS, Kim Y et al (2013) Indole and 7-benzyloxyindole attenuate the virulence of Staphylococcus aureus. Appl Microbiol Biotechnol 97:4543–4552. https://doi.org/10.1007/s00253-012-4674-z
Lee J-H, Kim Y-G, Lee K et al (2016) Streptomyces-derived actinomycin D inhibits biofilm formation by Staphylococcus aureus and its hemolytic activity. Biofouling 32:45–56. https://doi.org/10.1080/08927014.2015.1125888
Leejae S, Hasap L, Voravuthikunchai SP (2013) Inhibition of staphyloxanthin biosynthesis in Staphylococcus aureus by rhodomyrtone, a novel antibiotic candidate. J Med Microbiol 62:421–428. https://doi.org/10.1099/jmm.0.047316-0
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Mimica MJ, Berezin EN, Carvalho RLB et al (2007) Detection of methicillin resistance in Staphylococcus aureus isolated from pediatric patients: is the cefoxitin disk diffusion test accurate enough? Brazilian J Infect Dis 11:415–417. https://doi.org/10.1590/S1413-86702007000400009
Mu YQ, Xie TT, Zeng H et al (2020) Streptomyces-derived actinomycin D inhibits biofilm formation via downregulating ica locus and decreasing production of PIA in Staphylococcus epidermidis. J Appl Microbiol 128:1201–1207. https://doi.org/10.1111/jam.14543
Nalca Y, Jänsch L, Bredenbruch F et al (2006) Quorum-sensing antagonistic activities of azithromycin in Pseudomonas aeruginosa PAO1: a global approach. Antimicrob Agents Chemother 50:1680–1688. https://doi.org/10.1128/AAC.50.5.1680-1688.2006
Otto M (2012) MRSA virulence and spread. Cell Microbiol 14:1513–1521. https://doi.org/10.1111/j.1462-5822.2012.01832.x
Pereira V, Lopes C, Castro A et al (2009) Characterization for enterotoxin production, virulence factors, and antibiotic susceptibility of Staphylococcus aureus isolates from various foods in Portugal. Food Microbiol 26:278–282. https://doi.org/10.1016/j.fm.2008.12.008
Rajaduraipandi K, Mani KR, Panneerselvam K et al (2006) Prevalence and antimicrobial susceptibility pattern of methicillin resistant Staphylococcus aureus: a multicentre study. Indian J Med Microbiol 24:34–38. https://doi.org/10.4103/0255-0857.19892
Sambanthamoorthy K, Smeltzer MS, Elasri MO (2006) Identification and characterization of msa (SA 1233), a gene involved in expression of SarA and several virulence factors in Staphylococcus aureus. Microbiology 152:2559–2572. https://doi.org/10.1099/mic.0.29071-0
Seleem NM, Abd El Latif HK, Shaldam MA, El-Ganiny A (2020) Drugs with new lease of life as quorum sensing inhibitors: for combating MDR Acinetobacter baumannii infections. Eur J Clin Microbiol Infect Dis. https://doi.org/10.1007/s10096-020-03882-z
Selvaraj A, Jayasree T, Valliammai A, Pandian SK (2019) Myrtenol attenuates MRSA biofilm and virulence by suppressing sarA expression dynamism. Front Microbiol 10:1–15. https://doi.org/10.3389/fmicb.2019.02027
Sethupathy S, Vigneshwari L, Valliammai A et al (2017) L-Ascorbyl 2,6-dipalmitate inhibits biofilm formation and virulence in methicillin-resistant: Staphylococcus aureus and prevents triacylglyceride accumulation in Caenorhabditis elegans. RSC Adv 7:23392–23406. https://doi.org/10.1039/c7ra02934a
Song Y, Liu CI, Lin FY et al (2009) Inhibition of staphyloxanthin virulence factor biosynthesis in Staphylococcus aureus: In vitro, in vivo, and crystallographic results. J Med Chem 52:3869–3880. https://doi.org/10.1021/jm9001764
Stepanovic S, Vukovic D, Hola V, Di Bonaventura G, Djukic S, Cirkovic I, Ruzicka F (2007) Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci SRDJAN. Apmis 115:891–899. https://doi.org/10.1111/j.1600-0463.2007.apm
Suzuki N, Ohtaguro N, Yoshida Y et al (2015) A compound inhibits biofilm formation of Staphylococcus aureus from streptomyces. Biol Pharm Bull 38:889–892. https://doi.org/10.1248/bpb.b15-00053
Tong SYC, Davis JS, Eichenberger E et al (2015) Staphylococcus aureus infections: epidemiology, pathophysiology, clinical manifestations, and management. Clin Microbiol Rev 28:603LP–661. https://doi.org/10.1128/CMR.00134-14
Upreti N, Rayamajhee B, Sherchan SP et al (2018) Prevalence of methicillin resistant Staphylococcus aureus, multidrug resistant and extended spectrum β-lactamase producing gram negative bacilli causing wound infections at a tertiary care hospital of Nepal 11 Medical and Health Sciences 1103 Clinical Sci. Antimicrob Resist Infect Control 7:1–10. https://doi.org/10.1186/s13756-018-0408-z
Wael AHH (2016) Diclofenac inhibits virulence of proteus mirabilis isolated from diabetic foot ulcer. African J Microbiol Res 10:733–743. https://doi.org/10.5897/ajmr2016.8043
Xiang H, Qiu JZ, Wang DC et al (2010) Influence of magnolol on the secretion of α-toxin by Staphylococcus aureus. Molecules 15:1679–1689. https://doi.org/10.3390/molecules15031679
Xue L, Chen YY, Yan Z et al (2019) Staphyloxanthin: a potential target for antivirulence therapy. Infect Drug Resist 12:2151–2160. https://doi.org/10.2147/IDR.S193649
Zouhir A, Jridi T, Nefzi A et al (2016) Inhibition of methicillin-resistant Staphylococcus aureus (MRSA) by antimicrobial peptides (AMPs) and plant essential oils. Pharm Biol 54:3136–3150. https://doi.org/10.1080/13880209.2016.1190763
Acknowledgements
The authors would like to acknowledge the staff of Pharmacology and Toxicology Department, Faculty of Pharmacy, Zagazig University for supplying the rabbit RBCs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interests.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Abbas, H.A., Atallah, H., El-Sayed, M.A. et al. Diclofenac mitigates virulence of multidrug-resistant Staphylococcus aureus. Arch Microbiol 202, 2751–2760 (2020). https://doi.org/10.1007/s00203-020-01992-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-01992-y