Abstract
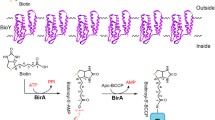
Following the analysis of transposon Tn5432-induced mutants of Corynebacterium glutamicum ATCC 13032, a gene encoding a protein with a biotin-binding motif was cloned. The DNA sequence of this gene revealed an open reading frame encoding 591 amino acids with a calculated mol. mass of 63.4 kDa. The protein is composed of two domains, an N-terminal biotin carboxylase and a C-terminal biotin-carboxyl-carrier protein, that are highly similar to corresponding subunits from prokaryotic and eukaryotic biotin enzymes. Over 70% identity was found to a protein from Mycobacterium leprae proposed to be part of an acyl-CoA carboxylase. Since it was not possible to inactivate the C. glutamicum gene, the gene most likely encodes a subunit of the essential acetyl-CoA carboxylase, which catalyzes the committed step in fatty acid synthesis.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 2 December 1995 / Accepted: 20 May 1996
Rights and permissions
About this article
Cite this article
Jäger, W., Peters-Wendisch, P., Kalinowski, J. et al. A Corynebacterium glutamicum gene encoding a two-domain protein similar to biotin carboxylases and biotin-carboxyl-carrier proteins. Arch Microbiol 166, 76–82 (1996). https://doi.org/10.1007/s002030050359
Issue Date:
DOI: https://doi.org/10.1007/s002030050359