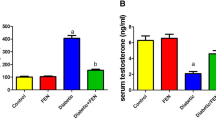
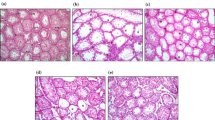
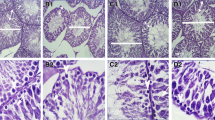
Abstract
Diabetes mellitus, a state of persistent hyperglycemia, is a major cause of micro- and macrovascular diseases. It affects nearly every system in the body including the reproductive system. Abnormalities in spermatogenesis and sexual function have been documented in animal models for both types of diabetes. The purpose of the present study is to determine the possible protective effects of enalapril against the germ cell toxicity in diabetic rat. Sprague–Dawley rats were divided into four groups: (1) control, (2) control + enalapril, (3) diabetic, and (4) diabetic + enalapril. Enalapril was administered per orally for 4 and 8 weeks continuously. After the treatment, animals were sacrificed and blood glucose level, sperm count, sperm DNA damage, apoptotic cell death, immunohistochemistry of 8-oxo- 7,8-dihydro- 2′-deoxyguanosine, and the cellular toxicity were performed. Furthermore, western blotting was performed to evaluate the expression of NFκB and COX-2 in testes. The results of the present study indicate that intervention of enalapril ameliorates the sperm DNA damage, reduces the oxidative stress, and down-regulates the expression of NFκB and COX-2 expression in streptozotocin-induced diabetic rat.








Similar content being viewed by others
References
Agarwal A, Said TM (2005) Oxidative stress, DNA damage and apoptosis in male infertility: a clinical approach. BJU Int 95(4):503–507
Agarwal A, Gupta S, Sikka S (2006) The role of free radicals and antioxidants in reproduction. Curr Opin Obstet Gynecol 18(3):325–332
Agbaje IM, Rogers DA, McVicar CM, McClure N, Atkinson AB, Mallidis C, Lewis SE (2007) Insulin dependant diabetes mellitus: implications for male reproductive function. Hum Reprod 22(7):1871–1877
Aitken RJ, Gordon E, Harkiss D, Twigg JP, Milne P, Jennings Z, Irvine DS (1998) Relative impact of oxidative stress on the functional competence and genomic integrity of human spermatozoa. Biol Reprod 59(5):1037–1046
Al-Aubaidy HA, Jelinek HF (2010) 8-Hydroxy-2-deoxy-guanosine identifies oxidative DNA damage in a rural prediabetes cohort. Redox Rep 15(4):155–160
Arun KH, Kaul CL, Ramarao P (2005) AT1 receptors and L-type calcium channels: functional coupling in supersensitivity to angiotensin II in diabetic rats. Cardiovasc Res 65(2):374–386
Bain SC, Le Guen CA, Lunec J, Barnett AH (1991) Comparison of the free radical scavenging activity of captopril versus enalapril: a three month in vivo study in hypertensive diabetic patients. J Hum Hypertens 5(6):511–515
Baluchnejadmojarad T, Roghani M, Imani A (2004) Protective effect of enalapril on vascular reactivity of the rat aorta. Vascul Pharmacol 40(6):301–307
Blankfield RP (2002) Angiotensin-receptor blockers, type 2 diabetes, and renoprotection. N Engl J Med 346(9):705–707
Brown CD, Forman CL, McEuen SF, Miller MG (1994) Metabolism and testicular toxicity of 1,3-dinitrobenzene in rats of different ages. Fundam Appl Toxicol 23(3):439–446
Burruel VR, Yanagimachi R, Whitten WK (1996) Normal mice develop from oocytes injected with spermatozoa with grossly misshapen heads. Biol Reprod 55(3):709–714
Burul-Bozkurt N, Pekiner C, Kelicen P (2010) Diabetes alters aromatase enzyme levels in gonadal tissues of rats. Naunyn Schmiedebergs Arch Pharmacol 382(1):33–41
Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S (2000) Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention by endothelin antagonism. Urol Res 28(5):342–347
Caneguim BH, Cerri PS, Spolidorio LC, Miraglia SM, Sasso-Cerri E (2009) Structural alterations in the seminiferous tubules of rats treated with immunosuppressor tacrolimus. Reprod Biol Endocrinol 7:19
Ceriello A, Motz E (2002) Angiotensin-receptor blockers, type 2 diabetes, and renoprotection. N Engl J Med 346(9):705–707
Chen SX, Song T, Zhou SH, Liu YH, Wu SJ, Liu LY (2008) Protective effects of ACE inhibitors on vascular endothelial dysfunction induced by exogenous advanced oxidation protein products in rats. Eur J Pharmacol 584(2–3):368–375
Costa JC, Costa RS, Silva CG, Coimbra TM (2006) Enalapril reduces the expression of nuclear factor-kappaB and c-Jun N-terminal kinase in the renal cortices of five-sixths-nephrectomized rats. Am J Nephrol 26(3):281–286
Cozzoli A, Nico B, Sblendorio VT, Capogrosso RF, Dinardo MM, Longo V, Gagliardi S, Montagnani M, De Luca A (2011) Enalapril treatment discloses an early role of angiotensin II in inflammation- and oxidative stress-related muscle damage in dystrophic mdx mice. Pharmacol Res 64(5):482–492
de Cavanagh EM, Inserra F, Ferder L, Romano L, Ercole L, Fraga CG (1995) Superoxide dismutase and glutathione peroxidase activities are increased by enalapril and captopril in mouse liver. FEBS Lett 361(1):22–24
de Cavanagh EM, Inserra F, Ferder L, Fraga CG (2000) Enalapril and captopril enhance glutathione-dependent antioxidant defenses in mouse tissues. Am J Physiol Regul Integr Comp Physiol 278(3):R572–R577
de Cavanagh EM, Inserra F, Toblli J, Stella I, Fraga CG, Ferder L (2001) Enalapril attenuates oxidative stress in diabetic rats. Hypertension 38(5):1130–1136
de Cavanagh EM, Piotrkowski B, Basso N, Stella I, Inserra F, Ferder L, Fraga CG (2003) Enalapril and losartan attenuate mitochondrial dysfunction in aged rats. FASEB J 17(9):1096–1098
de Cavanagh EM, Flores I, Ferder M, Inserra F, Ferder L (2008) Renin–angiotensin system inhibitors protect against age-related changes in rat liver mitochondrial DNA content and gene expression. Exp Gerontol 43(10):919–928
Duarte J, Martinez A, Bermejo A, Vera B, Gamez MJ, Cabo P, Zarzuelo A (1999) Cardiovascular effects of captopril and enalapril in obese Zucker rats. Eur J Pharmacol 365(2–3):225–232
Eidem I, Stene LC, Henriksen T, Hanssen KF, Vangen S, Vollset SE, Joner G (2010) Congenital anomalies in newborns of women with type 1 diabetes: nationwide population-based study in Norway, 1999–2004. Acta Obstet Gynecol Scand 89(11):1403–1411
Evans MD, Cooke MS (2004) Factors contributing to the outcome of oxidative damage to nucleic acids. Bioessays 26(5):533–542
Ghorbanihaghjo A, Veisi P, Argani H, Aghaeishahsavari M, Noroozianavval M, Rashtchizadeh N, Mesgari M, Safa J, Babaei H (2008) Prevention of DNA damage in renal transplantation by losartan and enalapril: the role of renin–angiotensin system polymorphisms. Clin Exp Nephrol 12(1):65–73
Griveau JF, Dumont E, Renard P, Callegari JP, Le Lannou D (1995) Reactive oxygen species, lipid peroxidation and enzymatic defence systems in human spermatozoa. J Reprod Fertil 103(1):17–26
Haines GA, Hendry JH, Daniel CP, Morris ID (2002) Germ cell and dose-dependent DNA damage measured by the comet assay in murine spermatozoaa after testicular X-irradiation. Biol Reprod 67(3):854–861
Ishikawa T, Fujioka H, Ishimura T, Takenaka A, Fujisawa M (2007) Increased testicular 8-hydroxy-2′-deoxyguanosine in patients with varicocele. BJU Int 100(4):863–866
Jackson G (2004) Sexual dysfunction and diabetes. Int J Clin Pract 58(4):358–362
Kakimoto M, Inoguchi T, Sonta T, Yu HY, Imamura M, Etoh T, Hashimoto T, Nawata H (2002) Accumulation of 8-hydroxy-2′-deoxyguanosine and mitochondrial DNA deletion in kidney of diabetic rats. Diabetes 51(5):1588–1595
Kishikawa H, Tateno H, Yanagimachi R (1999) Chromosome analysis of BALB/c mouse spermatozoa with normal and abnormal head morphology. Biol Reprod 61(3):809–812
Kodama H, Yamaguchi R, Fukuda J, Kasai H, Tanaka T (1997) Increased oxidative deoxyribonucleic acid damage in the spermatozoa of infertile male patients. Fertil Steril 68(3):519–524
Koh PO (2007a) Streptozotocin-induced diabetes increases apoptosis through JNK phosphorylation and Bax activation in rat testes. J Vet Med Sci 69(9):969–971
Koh PO (2007b) Streptozotocin-induced diabetes increases the interaction of Bad/Bcl-XL and decreases the binding of pBad/14-3-3 in rat testis. Life Sci 81(13):1079–1084
Kushwaha S, Vikram A, Jena GB (2011) Protective effects of enalapril in streptozotocin-induced diabetic rat: studies of DNA damage, apoptosis and expression of CCN2 in the heart, kidney and liver. J Appl Toxicol
Mughal MZ, Eelloo J, Roberts SA, Maresh M, Ward KA, Ashby R, Sibley CP, Adams JE (2010) Body composition and bone status of children born to mothers with type 1 diabetes mellitus. Arch Dis Child 95(4):281–285
Munzel T, Keaney JF Jr (2001) Are ACE inhibitors a "magic bullet" against oxidative stress? Circulation 104(13):1571–1574
Murray FT, Cameron DF, Orth JM (1983) Gonadal dysfunction in the spontaneously diabetic BB rat. Metabolism 32(7 Suppl 1):141–147
National High Blood Pressure Education Program (NHBPEP) Working Group (1994) National High Blood Pressure Education Program Working Group report on hypertension in diabetes. Hypertension 23(2):145–158
Neeraja S, Sreenath AS, Reddy PR, Reddanna P (2003) Expression of cyclooxygenase-2 in rat testis. Reprod Biomed Online 6(3):302–309
Ricci G, Catizone A, Esposito R, Pisanti FA, Vietri MT, Galdieri M (2009) Diabetic rat testes: morphological and functional alterations. Andrologia 41(6):361–368
Rossi A, Kapahi P, Natoli G, Takahashi T, Chen Y, Karin M, Santoro MG (2000) Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IkappaB kinase. Nature 403(6765):103–108
Sakai Y, Aminaka M, Takata A, Kudou Y, Yamauchi H, Aizawa Y, Sakagami H (2010) Oxidative stress in mature rat testis and its developmental changes. Dev Growth Differ 52(7):657–663
Savoia C, Schiffrin EL (2007) Vascular inflammation in hypertension and diabetes: molecular mechanisms and therapeutic interventions. Clin Sci (Lond) 112(7):375–384
Scarano WR, Messias AG, Oliva SU, Klinefelter GR, Kempinas WG (2006) Sexual behaviour, sperm quantity and quality after short-term streptozotocin-induced hyperglycaemia in rats. Int J Androl 29(4):482–488
Schmid U, Stopper H, Schweda F, Queisser N, Schupp N (2008) Angiotensin II induces DNA damage in the kidney. Cancer Res 68(22):9239–9246
Shanmugam N, Gaw Gonzalo IT, Natarajan R (2004) Molecular mechanisms of high glucose-induced cyclooxygenase-2 expression in monocytes. Diabetes 53(3):795–802
Sikka SC (2001) Relative impact of oxidative stress on male reproductive function. Curr Med Chem 8(7):851–862
Sun JG, Jurisicova A, Casper RF (1997) Detection of deoxyribonucleic acid fragmentation in human sperm: correlation with fertilization in vitro. Biol Reprod 56(3):602–607
Tattersall R (1982) Sexual problems of diabetic men. Br Med J (Clin Res Ed) 285(6346):911–912
Tokunaga I, Gotohda T, Ishigami A, Kitamura O, Kubo S (2003) Toluene inhalation induced 8-hydroxy-2′-deoxyguanosine formation as the peroxidative degeneration in rat organs. Leg Med (Tokyo) 5(1):34–41
Tripathi DN, Jena GB (2009) Intervention of astaxanthin against cyclophosphamide-induced oxidative stress and DNA damage: a study in mice. Chem Biol Interact 180(3):398–406
Varin R, Mulder P, Tamion F, Richard V, Henry JP, Lallemand F, Lerebours G, Thuillez C (2000) Improvement of endothelial function by chronic angiotensin-converting enzyme inhibition in heart failure: role of nitric oxide, prostanoids, oxidant stress, and bradykinin. Circulation 102(3):351–356
Vikram A, Tripathi DN, Ramarao P, Jena GB (2008) Intervention of d-glucose ameliorates the toxicity of streptozotocin in accessory sex organs of rat. Toxicol Appl Pharmacol 226(1):84–93
Walser M (2002) Angiotensin-receptor blockers, type 2 diabetes, and renoprotection. N Engl J Med 346(9):705–707
Yamazaki T, Tanimoto M, Gohda T, Ohara I, Hagiwara S, Murakoshi M, Matsumoto M, Kaneko S, Aoki T, Toyoda H, Ishikawa Y, Funabiki K, Horikoshi S, Tomino Y (2009) Combination effects of enalapril and losartan on lipid peroxidation in the kidneys of KK-Ay/Ta mice. Nephron Exp Nephrol 113(2):e66–e76
Yao LS, Wang YT, Chen Y, Dai YT (2009) Expressions of NOS isoforms in the cavernous tissues of diabetic rat models. Zhonghua Nan Ke Xue 15(10):915–919
Zemel P (1988) Sexual dysfunction in the diabetic patient with hypertension. Am J Cardiol 61(16):27H–33H
Zhang BQ, Huang YS, Zhang JP, Zhang DX, Dang YM, Wang G, Hu JY, Lei ZY, Xiao R (2007) Protective effects of administration of enalapril maleate on rat myocardial damage in early stage of burns. Zhonghua Shao Shang Za Zhi 23(5):335–338
Zhu JH, Jia RP, Xu LW, Wu JP, Wang ZZ, Wang SK, Bo CJ (2010) Reduced expression of SK3 and IK1 channel proteins in the cavernous tissue of diabetic rats. Asian J Androl 12(4):599–604
Acknowledgments
We wish to acknowledge the financial assistance received from the Department of Science and Technology (DST), India (vide letter no. SR/SO/HS-83/2008) for the present work. Furthermore, the financial assistance received from the National Institute of Pharmaceutical Education and Research (NIPER), S.A.S. Nagar, Mohali, Punjab, India is duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kushwaha, S., Jena, G.B. Enalapril reduces germ cell toxicity in streptozotocin-induced diabetic rat: investigation on possible mechanisms. Naunyn-Schmiedeberg's Arch Pharmacol 385, 111–124 (2012). https://doi.org/10.1007/s00210-011-0707-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-011-0707-x