Abstract
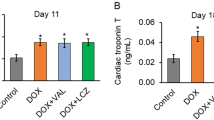
Doxorubicin (Dox)-induced cardiotoxicity constitutes the major adverse effect that limited its use. We investigated the possible protective effects of liraglutide on Dox-induced cardiotoxicity in rats. Rats were divided into the following groups: control group rats received normal saline [1 ml/kg, intraperitoneal (i.p.)]; doxorubicin group rats received doxorubicin (1.25 mg/kg, i.p.), four times per week for 4 weeks; and liraglutide group rats received doxorubicin (1.25 mg/kg, i.p.) four times per week for 4 weeks then received liraglutide (100 μg/kg, i.p) daily for 4 weeks. At the end of the study, animals were sacrificed and serum creatine kinase-MB (CK-MB) and troponin I levels were determined. Malondialdehyde (MDA), superoxide dismutase (SOD), tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and caspase-3 levels of the heart were determined. Cardiac AMPK, phosphorylated-Akt, tissue growth factor-β1 (TGF-β1), and GSK3-β levels of the heart were determined. Hematoxylin and eosin (H&E) stained sections form the heart were examined as well as immunohistochemical sections for detection of Bcl-2 expression. Dox treatment increased serum level of troponin I and CK-MB while decreased SOD activity, decreased AMPK, and p-Akt cardiac levels with increased in MDA, IL-6, TNF-α,GSK-3b, TGFB1, and caspase-3 levels in the heart with inflammation and necrosis in cardiac histopathology with decreased Bcl-2. Treatment with liraglutide decreased troponin I and CK-MB while increased SOD activity, AMPK, p-Akt with decrements in MDA, IL-6, TNF-α, GSK-3β, TGF-β1, and caspase-3 levels with attenuation of inflammation and necrosis while increased Bcl-2 expression. Liraglutide may thus represent a new clinical tool for the treatment of Dox-induced cardiotoxicity.



Similar content being viewed by others
References
Langlois A, Dal S, Viovt K, Mura C, Seyfritz E, Bietiger W, Dollinger C et al (2016) Improvement of islet graft function using liraglutide is correlated with its anti-inflammatory properties. British J of Pharmac 173:3443–3453
Arumugam TV, Shiels IA, Woodruff TM, Granger DN, Taylor SM (2004) The role of the complement system in ischemic-reperfusion injury. Shock 21(5):401–409
Bonadonna G, Monfardini S, DeLena M, Fossati-Bellani F, Beretta G (1970) Phase I and preliminary phase II evaluation of adriamycin (NSC/23/27). Cancer Res 30(10):2572–2582
Bullock BP, Helelr RS, Habener JF (1996) Tissue distribution of messenger ribonucleic acid encoding the rat glucagon-like peptide-1 receptor. Endocrinology 137:2968–2978
Buteau J et al (2004) Glucagon-like peptide-1 prevents beta cell glucolipotoxicity. Diabetologia 47:806–815
Chatterjee K, Zhang J, Honbo N, Karliner JS (2010) Doxorubicin cardiomyopathy. Cardiology 115(2):155–162
Degn KB, Juhl CB, Sturis J, Jakobsen G, Brocj B, Chandramouli V, Rungby J, Lanbdau BR, Schmitz O (2004) One week’s treatment with the long-acting glucagon-like peptide-1 derivative Liraglutide (NN211) markedly improves 24-h glycaemia and alpha- and beta-cell function and reduces endogenous glucose in patient with type 2 diabetes. Diabetes 53:1187–1194
Dong W, Miao Y, Chen A, Cheng M, Ye X, Song F, Zheng G (2017). Delayed administration of the GLP-1 receptor agonist liraglutide improves metabolic and functional recovery after cerebral ischemia in rats. Neurosci Lett. 22; 641:1-7
Dragojevi-Simi V, Dobri S, Bokonji D, Vucinic Z, Sinovec S, Jacevic V et al (2004) Amifostine protection against doxorubicin cardiotoxicity in rats. Anti-Cancer Drugs 15(2):169–178
During MJ, Cao L, Zuzga DS, Francis JS, Fitzsimon HL, Jiao X, Bland RJ, Klugmann M, Banks WA, Drucker DJ, Haile CN (2003) Glucagon-like peptide-1 receptor is involved in learning and neuroprotection. Nat Med 9:1173–1179
Fan GC, Zhou X, Wang X, Song G, Qian J, Nicolaou P, Chen G, Ren X, Kranias EG (2008) Heat shock protein 20 interacting with phosphorylated Akt reduces doxorubicin-triggered oxidative stress and cardiotoxicity. Cir Res 103(11):1270–1279
Fernandes JL, de Oliveira RT, Mamoni RL, Coelho OR, Nicolaau JC, Blotta MH et al (2008) Pentoxifylline reduced pro-inflammatory and increase anti-inflammatory activity in patients with coronary artery disease-randomized placebo-controlled study. Atherosclerosis 196(1):434–442
Gao H, Hu L, Li D, et al. (2013): Effects of glycagon like peptide-1 on liver oxidative stress, TNFα and TGF-β1 in rats with non-alcoholic fatty liver disease. Nan Fang Yi Ke DaXue Xue Bao; 33: 1661–1664 (In Chinese)
Gratia S, Kay L, Potenza L, Seffouh A, Novel-Chate V, Schnebelen C, Sestili P, Schalttner U, Tokarska-Schlattner M (2012) Inhibition of AMPK signaling by doxorubicin: at the crossroads of the cardiac responses to energetic, oxidative and genotoxic stress. Cardiovasc Res 95(3):290–299
Hausenloy DJ, Yellon DM (2004) New directions for protecting the heart against ischemic-reperfusion injury: targeting the reperfusion injury: targeting the reperfusion injury salvage kinase (RISK)-pathway. Cardiovasc Res 61:448–460
Holz GGT, Kuhtreiber WM, Habener JF (1993) Pancreatic beta cells are rendered glucose-competent by the insulinotropic hormone glucagon-like peptide-1 (7-37). Nature 361:362–365
Hortobagyi GN (1997) Anthracycline in the treatment of cancer. An over view. Drugs 54(Suppl. 4):1–7
Konishi M, Haraguchi OH, Ishihara T, Saito K, Nakano Y, Isobe M (2011) Adiponectin protects against doxorubicin-induced cardiomyopathy by anti-apoptotic effects through AMPK up regulation. Cardiovasc Res 89:309–319
Lai RS, Wang JS, Hsu HK, Chang HC, Lin CH, Lin MH (2002) Prognostic evaluation of the expression of p53 and Bcl-2 oncoproteins in patients with surgically resected non-small cell lung cancer. Jpn J Clin Oncol 32(10):393–397
Li XN, Song J, Zhang L, LeMaire SA, Hou X, Zhang C et al (2009) Activation of the AMPK-FOXO3 pathway reduces fatty acid-induced increase in intracellular reactive oxygen species by upregulating thioredoxin. Diabetes 58:2246–2257
Li Y, Hansotia T, Yusta B, Ris F, Halban PA, Drucker DJ (2003) Glycagon-like peptide-1 receptor signaling modulates beta cell apoptosis. J Biol Chem 278:471–478
Liu Y, Tanabe K, Baronnier D, Patel S, Woodgett J, et al. (2010): Conditional ablation of GSK-3β in islet beta cells results in expanded mass and resistance to fall feeding-induced diabetes in mice. Diabetologia; 53: 2600–2610
Ma YX, Li WH, Xie Q (2013) Rosuvastatin inhibits TGF-beta1 expression and alleviates myocardial fibrosis in diabetic rats. Pharmazie 68:355–358
Mann DL (2015) Innate immunity and the failing heart: the cytokine hypothesis revisited. Cir Res 116(7):1254–1268
Marre M, Shaw J, Brandle M, Bebakar WM, Kamauddin NA, Strand J et al (2009) LEAD-1 SU study group. Liraglutide a once daily human GLP-1 analogue added to sulphonylurea over 26 weeks produces greater improvements in glycemic and weight control compared with adding rosiglitazone or placebo in subjects with type 2 diabetes (LEAD-1 SU). Diabet Med 26:268–278
Maslov MY, Chacko VP, Hirsch GA, Akki A, Leppo MK, Steenbergen C et al (2010) Reduced in vivo high-energy phosphates precede adriamycin-induced cardiac dysfunction. Am J Physiol Heart Circ Physiol 299:H332–H337
Matsui T, Tao K, del Monte F et al (2001) Akt activation preserve cardiac function and prevents injury after transient cardiac ischemia in vivo. Circulation 104:330–335
Milic-Tores V, Srdjenovic B, Jacevic V, Dragojevic-Simic V, Djordjeric A, Luisa Simplicio A (2010) Fullerenol C60(OH)24 prevents doxorubicin-induced acute cardiotoxicity in rats. Pharmacol Rep 62(4):707–718
Nitobe J, Yamaguchi S, Okuyama M, Nozaki N, Sata M, Miyamoto T et al (2003) Reactive oxygen species regulate FLICE inhibitory protein (FLIP) and susceptibility to Fas-mediated apoptosis in cardiac myocytes. Cardiovasc Res 57:119–128
Noyan-Ashraf HM, Abdul Momen M, Ban K, Sadi A, Zhou Y, Riazi AM, Baggio LL, Henkelman RM, Husain M, Drucker DJ (2009) GLP-IR agonist Liraglutide activates cytoprotection pathways and improves outcomes after experimental myocardial infarction in mice. Diabetes 58(4):975–983
Okada K, Kotani K, Yagyu H, Ando A, Osuga JI, Ishibashi S (2014) Effect of treatment with liraglutide on oxidative stress and cardiac natriuretic peptide levels in patients with type 2 diabetes mellitus. Endocrine 47:962–964
Olsen RD, Mushlin PS (1990) Doxorubicin cardiotoxicity: analysis of prevailing hypotheses. FASEB 4:3076–3086
Parlakpinar H, Tasdemir S, Polat A, Bay-Karabulut A, Vardi N, Ucar M (2005) Protective role of caffeic acid phenethylester (cape) on gentamicin-induced acute renal toxicity in rats. Toxicology 24:33–37
Popkie AP, Zeidner LC, Albrecht AM, D’Ippolite A, Eckardt S, Newsom DE, Groden J, Doble BW, Aronow B, McLaughlin KJ, White P, Phiel CJ (2010) Phosphatidylinositol3-kinase (PI3K) signaling via glycogen synthase kinase-3 (GSK-3) regulates DNA methylation of imprinted loci. J Biol Chem 285:41337–41347
Quintanilla RA, Dolan PJ, Jin YN, Hohnson GV (2012) Truncated tau and Abeta cooperatively impair mitochondrial in primary neurons. Neurobial aging 33:619e25–619e35
Rahimi Balaei M1, Momeny M, Babaeikelishomi R, Ejtemaei Mehr S, Tavangar SM, Dehpour AR. (2010) The modulatory effect of lithium on doxorubicin-induced cardiotoxicity in rat. Eur J Pharmacol 641(2–3):193–198
Reeve JL, Szegezdi E, Logue SE et al (2007) Distinct mechanisms of cardiomyocytes apoptosis induced doxorubicin and hypoxia converge on mitochondria and are inhibited by BCl-xl. J Cell Mol Med 11:509–520
Rubin J, Matsushita K, Ballantyne CM, Hoogeveen R, Coresh J, Selvin E (2012) Chronic hyperglycemia and subclinical myocardial injury. Journal of the Amwerican College of Cardiology 59(5):484–489
Ruderman NB, Carling D, Prentki M, Cacicedo JM (2013) AMPK, insulin resistance, and the metabolic syndrome. J Clin Invest 123:2764–2772
Scharstuhl A, Glansbeek HL, Van Beuningen HM, Vitters EL, Vander Krann PM, Vanden Berg WB (2002) Inhibition of endogenous TGF-beta during experimental osteoarthritis prevents osteophyte formation and impairs cartilage repair. J Immunol 169:507–514
Sheu MT, Jhan HJ, Hsieh CM et al (2015) Efficacy of antioxidants as a complementary and alternative medicine (CAM) in combination with chemotherapeutic agents doxorubicin. Integr Cancer Ther 14:184–195
Simunek T, Sterba M, Popelova O, Adamcova M, Hrdina R, Gersi V (2009) Anthracycline-induced cardiotoxicity: overview of studies examining the roles of oxidative stress and free cellular iron. Pharmacol Rep 61(1):154–171
Tomoaki I, Toyoshi I, Sonoda N, Hari H, Hiroaki M et al (2015) GLP1 analog liraglutide protects against cardiac steatosis, oxidative stress and apoptosis in streptozotocin-induced diabetic rats. Atherosclerosis 240:250–259
Ueno M, Kakinuma Y, Yuhki K, Murakoshi N, Iemitsu M, Miyauchi T, Yamaguchi O (2006) Doxorubicin induces apoptosis by activation of caspase-3 in cultured cardiomyocytes in vitro and rat cardiac ventricles in vivo. J Pharmacol Sci 101(2):151–158
Wang B, Hao J, Jones SC, Yee MS, Roth JC, Dixon IMC (2002) Decreased Smad 7 expression contributes to cardiac fibrosis in the infracted rat heart. Am J Physiol 282(5):H1685–H1696
Wang S, Song P, Zhou MH (2012) Inhibition of AMP-activated protein kinase alpha (AMPK alpha) by doxorubicin accentuates genotoxic stress and cell death in mouse embryonic fibroblasts and cardiomyocytes: role of p53 and SITR1. J Biol Chem 287:8001–8012
Wang Y, Yang T (2015) Liraglutide reduces oxidized DL induced oxidative stress and fatty degeneration in raw 264.7 cells involving the AMPK/SREBP1 pathway. J of Geriatric Cardiology 12:410–416
Yu W, Wu J, Cai F, Xiang J, Zha W et al (2012) Curcumin alleviates diabetic cardiomyopathy in experimental diabetic rats PLOS one 7:e52013
Zhu H, Zhang Y, Shi Z, Lu D, Li T, Ding Y, Ruan Y, Xu (2016): The neuroprotection of liraglutide against ischemia-induced apoptosis through the activation of the PI3k/Akt and MAP pathways. Scientific Reports 6, Article number: 26859 doi: 10.10381sreo26859
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental protocols were approved by the ethics committee of Zagazig University.
The study was approved by the local Animal Ethical Committee of Zagazig University, Egypt. All experimental procedures were carried out in accordance with the guidelines set forth by the National Institutes of Health (USA).
Rights and permissions
About this article
Cite this article
Abbas, N.A.T., Kabil, S.L. Liraglutide ameliorates cardiotoxicity induced by doxorubicin in rats through the Akt/GSK-3β signaling pathway. Naunyn-Schmiedeberg's Arch Pharmacol 390, 1145–1153 (2017). https://doi.org/10.1007/s00210-017-1414-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-017-1414-z