Abstract
Rationale
Despite the well documented neurochemical actions of 3,4-methylenedioxymethamphetamine (MDMA), acute effects in rats previously exposed to the drug have not been extensively explored.
Objective
To examine motor activity and vigilance effects of MDMA in drug-naive rats and in rats exposed to the drug 3 weeks earlier.
Methods
MDMA (15 mg/kg, IP) was administered to Dark Agouti rats. Motor activity, wakefulness, light slow wave sleep (SWS-1), deep slow wave sleep (SWS-2) and paradoxical sleep (PS), sleep and PS latencies were measured. Acrophases and amplitudes of the 24 h cycles were calculated by cosinor analysis. In parallel groups, local cerebral glucose utilization (lCMRglu) and (3H)-paroxetine binding were measured in motor areas of the brain.
Results
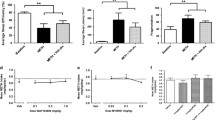
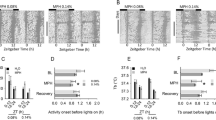
In drug-naive rats MDMA caused marked increases in motor activity and wakefulness for at least 5–6 h. Circadian patterns of motor activity and sleep/vigilance parameters were altered up to 5 days after treatment. Despite most parameters tending to return to normal, there were still significant effects of MDMA on motor activity, wakefulness, and SWS-2 28 days later. Acute MDMA administration caused significant increases in lCMRglu, but after 3 weeks lCMRglu was decreased in the same brain areas. No significant change in [3H]paroxetine binding was observed in motor areas, although significant reductions were seen elsewhere (neocortex −81%). In rats exposed to MDMA 3 weeks earlier, most acute effects induced by MDMA administration were similar to those in drug-naive rats, but shorter duration of the acute effects were found in motor activity and vigilance.
Conclusions
Our findings provide evidence that MDMA use can lead to long-term changes in regulation of circadian rhythms, motor activity and sleep generation.







Similar content being viewed by others
References
Allen RP, McCann UD, Ricaurte GA (1993) Persistent effects of (±)3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”) on human sleep. Sleep 16:560–564
Antle MC, Marchant EG, Niel L, Mistlberger RE (1998) Serotonin antagonists do not attenuate activity-induced phase shifts of circadian rhythms in the Syrian hamster. Brain Res 813:139–149
Bagdy G, Graf M, Anheuer ZE, Modos EA, Kantor S (2001) Anxiety-like effects induced by acute fluoxetine, sertraline or m-CPP treatment are reversed by pretreatment with the 5-HT2C receptor antagonist SB-242084 but not the 5-HT1A receptor antagonist WAY-100635. Int J Neuropsychopharmacol 4:399–408
Biello SM, Dafters RI (2001) MDMA and fenfluramine alter the response of the circadian clock to a serotonin agonist in vitro. Brain Res 920:202–209
Bjorvatn B, Ursin R (1990) Effects of zimeldine, a selective 5-HT reuptake inhibitor, combined with ritanserin, a selective 5-HT2 antagonist, on waking and sleep stages in rats. Behav Brain Res 40:239–246
Borbely AA, Trachsel L, Tobler I (1988) Effect of ritanserin on sleep stages and sleep EEG in the rat. Eur J Pharmacol 156:275–278
Callaway CW, Geyer MA (1992) Tolerance and cross-tolerance to the activating effects of 3,4-methylenedioxymethamphetamine and a 5-hydroxytryptamine1B agonist. J Pharmacol Exp Ther 263:318–326
Callaway CW, Rempel N, Peng RY, Geyer MA (1992) Serotonin 5-HT1-like receptors mediate hyperactivity in rats induced by 3,4-methylenedioxymethamphetamine. Neuropsychopharmacology 7:113–127
Cambras T, Vilaplana J, Campuzano A, Canal-Corretger MM, Carulla M, Díez-Noguera (2000) Entrainment of the rat motor activity rhythm: effects of the light-dark cycle and physical exercise. Physiol Behav 70:227–232
Chapin EM, Andrade R (2001a) A 5-HT(7) receptor-mediated depolarization in the anterodorsal thalamus. I. Pharmacological characterization. J Pharmacol Exp Ther 297:395–402
Chapin EM, Andrade R (2001b) A 5-HT(7) receptor-mediated depolarization in the anterodorsal thalamus. II. Involvement of the hyperpolarization-activated current I(h). J Pharmacol Exp Ther 297:403–409
Colado MI, Esteban B, O’Shea E, Granados R, Green AR (1999) Studies on the neuroprotective effect of pentobarbitone on MDMA-induced neurodegeneration. Psychopharmacology 142:421–425
Colado MI, O’Shea E, Esteban B, Green AR (2001) Studies on the neuroprotective effect of the enantiomers of AR-A008055, a compound structurally related to clomethiazole, on MDMA (“ecstasy”)-induced neurodegeneration in rat brain. Psychopharmacology 157:82–88
Colbron S, Jones M, Biello SM (2002) MDMA alters the response of the circadian clock to a photic and non-photic stimulus. Brain Res 956:45–52
Commins DL, Axt KJ, Vosmer G, Seiden LS (1987a) Endogenously produced 5,6-dihydroxytryptamine may mediate the neurotoxic effects of para-chloroamphetamine. Brain Res 419:253–261
Commins DL, Vosmer G, Virus RM, Woolverton WL, Schuster CR, Seiden LS (1987b) Biochemical and histological evidence that methylenedioxymethylamphetamine (MDMA) is toxic to neurons in the rat brain. J Pharmacol Exp Ther 241:338–345
De Souza EB, Kuyatt BL (1987) Autoradiographic localization of 3H-paroxetine-labeled serotonin uptake sites in rat brain. Synapse 1:488–496
Doyon S (2001) The many faces of ecstasy. Curr Opin Pediatr 13:170–176
Duncan WC Jr (1996) Circadian rhythms and the pharmacology of affective illness. Pharmacol Ther 71:253–312
Esteban B, O’Shea E, Camarero J, Sanchez V, Green AR, Colado MI (2001) 3,4-Methylenedioxymethamphetamine induces monoamine release, but not toxicity, when administered centrally at a concentration occurring following a peripherally injected neurotoxic dose. Psychopharmacology 154:251–260
Filakovszky J, Kantor S, Halasz P, Bagdy G (2001) 8-OH-DPAT and MK-801 affect epileptic activity independently of vigilance. Neurochem Int 38:551–556
Fischer C, Hatzidimitriou G, Wlos J, Katz J, Ricaurte G (1995) Reorganization of ascending 5-HT axon projections in animals previously exposed to the recreational drug (±)3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). J Neurosci 15:5476–5485
Glass JD, Grossman GH, Farnbauch L, DiNardo L (2003) Midbrain raphe modulation of nonphotic circadian clock resetting and 5-HT release in the mammalian suprachiasmatic nucleus. J Neurosci 23:7451–7460
Gottesmann C (1992) Detection of seven sleep-waking stages in the rat. Neurosci Biobehav. Rev. 16:31–38
Green AR, Cross AJ, Goodwin GM (1995) Review of the pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA or “ecstasy”). Psychopharmacology 119:247–260
Green AR, Mechan AO, Elliott JM, O’Shea E, Colado MI (2003) The pharmacology and clinical pharmacology of 3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). Pharmacol Rev 55:463–508
Jacobs BL, Fornal CA (1999) Activity of serotonergic neurons in behaving animals. Neuropsychopharmacology 21:9S–15S
Johnson SW, Mercuri NB, North RA (1992) 5-Hydroxytryptamine1b receptors block the GABAB synaptic potential in rat dopamine neurons. J Neurosci 12:2000–2006
Kantor S, Gerber K, Halasz P, Bagdy G (2000) The role of 5-HT2A, 5-HT2C and 5HT3 serotonin receptors in the regulation of sleep. J. Physiol 526:66P–67P
Kantor S, Jakus R, Bodizs R, Halasz P, Bagdy G (2002) Acute and long-term effects of the 5-HT2 receptor antagonist ritanserin on EEG power spectra, motor activity, and sleep: changes at the light-dark phase shift. Brain Res 943:105–111
Kelly PA, Ritchie IM, Collins FM (1995) Cerebrovascular consequences of repeated exposure to NG-nitro-l-arginine methyl ester. Br J Pharmacol 116:2771–2777
Kupfer DJ, Reynolds CF 3rd, Ulrich RF, Shaw DH, Coble PA (1982) EEG sleep, depression, and aging. Neurobiol Aging 3:351–360
Manfredini R, Manfredini F, Fersini C, Conconi F (1998) Circadian rhythms, athletic performance, and jet lag. Br J Sports Med 32:101–106
Marston HM, Reid ME, Lawrence JA, Olverman HJ, Butcher SP (1999) Behavioural analysis of the acute and chronic effects of MDMA treatment in the rat. Psychopharmacology 144:67–76
McCann UD, Eligulashvili V, Ricaurte GA (2000) (±)3,4-Methylenedioxymethamphetamine (“ecstasy”)-induced serotonin neurotoxicity: clinical studies. Neuropsychobiology 42:11–16
McCreary AC, Bankson MG, Cunningham KA (1999) Pharmacological studies of the acute and chronic effects of (+)-3, 4-methylenedioxymethamphetamine on locomotor activity: role of 5-hydroxytryptamine(1A) and 5-hydroxytryptamine(1B/1D) receptors. J Pharmacol Exp Ther 290:965–973
McKenna DJ, Peroutka SJ (1990) Neurochemistry and neurotoxicity of 3,4-methylenedioxymethamphetamine (MDMA, “ecstasy”). J Neurochem 54:14–22
Mechan AO, O’Shea E, Elliott JM, Colado MI, Green AR (2001) A neurotoxic dose of 3,4-methylenedioxymethamphetamine (MDMA; ecstasy) to rats results in a long-term defect in thermoregulation. Psychopharmacology 155:413–418
Meijer JH, Rietveld WJ (1989) Neurophysiology of the suprachiasmatic circadian pacemaker in rodents. Physiol Rev 69:671–707
Meyer-Bernstein EL, Morin LP (1996) Differential serotonergic innervation of the suprachiasmatic nucleus and the intergeniculate leaflet and its role in circadian rhythm modulation. J Neurosci 16:2097–111
Meyer-Bernstein EL, Blanchard JH, Morin LP (1997) The serotonergic projection from the median raphe nucleus to the suprachiasmatic nucleus modulates activity phase onset, but not other circadian rhythm parameters. Brain Res 755:112–120
Mintz EM, Gillespie CF, Marvel CL, Huhman KL, Albers HE (1997) Serotonergic regulation of circadian rhythms in Syrian hamsters. Neuroscience 79:563–569
Mistlberger RE (1994) Circadian food-anticipatory activity: formal models and physiological mechanisms. Neurosci Biobehav Rev 18:171–195
Monckton J, McCormick DA (2003) Comparative physiological and serotoninergic properties of pulvinar neurons in the monkey, cat and ferret. Thalamus Related Systems (in press)
Monti JM, Jantos H (1992) Dose-dependent effects of the 5-HT1A receptor agonist 8-OH-DPAT on sleep and wakefulness in the rat. J Sleep Res 1:169–175
Monti JM, Monti D, Jantos H, Ponzoni A (1995) Effects of selective activation of the 5-HT1B receptor with CP-94,253 on sleep and wakefulness in the rat. Neuropharmacology 34:1647–1651
Monti JM, Ponzoni A, Jantos H, Lagos P, Silveira R, Banchero P (1999) Effects of accumbens m-chlorophenylbiguanide microinjections on sleep and waking in intact and 6-hydroxydopamine-treated rats. Eur J Pharmacol 364:89–98
Morgan MJ (2000) Ecstasy (MDMA): a review of its possible persistent psychological effects. Psychopharmacology 152:230–248
Morin LP (1999) Serotonin and the regulation of mammalian circadian rhythmicity. Ann Med 31:12–33
Mrosovsky N (1996) Locomotor activity and non-photic influences on circadian clocks. Biol Rev Camb Philos Soc 71:343–372
Nelson W, Tong YL, Lee J, Halberg F (1979) Methods for cosinor-rhythmometry. Chronobiologia 6:305–323
O’Hearn E, Battaglia G, De Souza EB, Kuhar MJ, Molliver ME (1988) Methylenedioxyamphetamine (MDA) and methylenedioxymethamphetamine (MDMA) cause selective ablation of serotonergic axon terminals in forebrain: immunocytochemical evidence for neurotoxicity. J Neurosci 8:2788–2803
Parrott AC (2000) Human research on MDMA (3,4-methylene- dioxymethamphetamine) neurotoxicity: cognitive and behavioural indices of change. Neuropsychobiology 42:17–24
Parrott AC (2001) Human psychopharmacology of ecstasy (MDMA): a review of 15 years of empirical research. Hum Psychopharmacol 16:557–577
Parrott AC, Lasky J (1998) Ecstasy (MDMA) effects upon mood and cognition: before, during and after a Saturday night dance. Psychopharmacology 139:261–268
Parrott AC, Milani RM, Parmar R, Turner JD (2001) Recreational ecstasy/MDMA and other drug users from the UK and Italy: psychiatric symptoms and psychobiological problems. Psychopharmacology 159:77–82
Portas CM, Bjorvatn B, Ursin R (2000) Serotonin and the sleep/wake cycle: special emphasis on microdialysis studies. Prog Neurobiol 60:13–35
Prosser RA, Dean RR, Edgar DM, Heller HC, Miller JD (1993) Serotonin and the mammalian circadian system: I. in vitro phase shifts by serotonergic agonists and antagonists. J Biol Rhythms 8:1–16
Rea MA, Pickard GE (2000) Serotonergic modulation of photic entrainment in the Syrian hamster. Biol Rhythm Res 31:284–314
Rempel NL, Callaway CW, Geyer MA (1993) Serotonin1B receptor activation mimics behavioral effects of presynaptic serotonin release. Neuropsychopharmacology 8:201–211
Reneman L, Booij J, Schmand B, van den Brink W, Gunning B (2000) Memory disturbances in “ecstasy” users are correlated with an altered brain serotonin neurotransmission. Psychopharmacology 148:322–324
Reynolds CF 3rd, Taska LS, Jarrett DB, Coble PA, Kupfer DJ (1983) REM latency in depression: is there one best definition? Biol Psychiatry 18:849–863
Ricaurte GA, DeLanney LE, Irwin I, Langston JW (1988a) Toxic effects of MDMA on central serotonergic neurons in the primate: importance of route and frequency of drug administration. Brain Res 446:165–168
Ricaurte GA, Forno LS, Wilson MA, DeLanney LE, Irwin I, Molliver ME, Langston JW (1988b) (±)3,4-Methylenedioxymethamphetamine selectively damages central serotonergic neurons in nonhuman primates. JAMA 260:51–55
Ricaurte GA, Martello AL, Katz JL, Martello MB (1992) Lasting effects of (±)-3,4-methylenedioxymethamphetamine (MDMA) on central serotonergic neurons in nonhuman primates: neurochemical observations. J Pharmacol Exp Ther 261:616–622
Ricaurte GA, Yuan J, McCann UD (2000) (±)3,4-Methylenedioxymethamphetamine (“ecstasy”)-induced serotonin neurotoxicity: studies in animals. Neuropsychobiology 42:5–10
Richardson GS, Malin HV (1996) Circadian rhythm sleep disorders: pathophysiology and treatment. J Clin Neurophysiol 13:17–31
Rusak B, Zucker I (1979) Neural regulation of circadian rhythms. Physiol Rev 59:449–526
Sanchez V, Camarero J, Esteban B, Peter MJ, Green AR, Colado MI (2001) The mechanisms involved in the long-lasting neuroprotective effect of fluoxetine against MDMA (“ecstasy”)-induced degeneration of 5-HT nerve endings in rat brain. Br J Pharmacol 134:46–57
Saper CB, Chou TC, Scammell TE (2001) The sleep switch: hypothalamic control of sleep and wakefulness. Trends Neurosci 24:726–731
Scanzello CR, Hatzidimitriou G, Martello AL, Katz JL, Ricaurte GA (1993) Serotonergic recovery after (±)3,4-(methylenedioxy) methamphetamine injury: observations in rats. J Pharmacol Exp Ther 264:1484–1491
Scearce K, Kassir S, Lucas J, Castanon N, Segu L, Arango V, Hen R (1997) Dopaminergic compensations in knockout mice lacking the serotonin 1B receptor. Society of Neuroscience, 27th Annual Meeting. Society for Neuroscience, New Orleans, La., p. 202.2
Schifano F (2000) Potential human neurotoxicity of MDMA (“ecstasy”): subjective self-reports, evidence from an Italian drug addiction centre and clinical case studies. Neuropsychobiology 42:25–33
Schifano F, Di Furia L, Forza G, Minicuci N, Bricolo R (1998) MDMA (“ecstasy”) consumption in the context of polydrug abuse: a report on 150 patients. Drug Alcohol Depend 52:85–90
Schmidt CJ (1987) Neurotoxicity of the psychedelic amphetamine, methylenedioxymethamphetamine. J Pharmacol Exp Ther 240:1–7
Schmidt CJ, Wu L, Lovenberg W (1986) Methylenedioxymethamphetamine: a potentially neurotoxic amphetamine analogue. Eur J Pharmacol 124:175–178
Schuster P, Lieb R, Lamertz C, Wittchen HU (1998) Is the use of ecstasy and hallucinogens increasing? Results from a community study. Eur Addict Res 4:75–82
Shankaran M, Yamamoto BK, Gudelsky GA (2001) Ascorbic acid prevents 3,4-methylenedioxymethamphetamine (MDMA)-induced hydroxyl radical formation and the behavioral and neurochemical consequences of the depletion of brain 5-HT. Synapse 40:55–64
Sharkey J, McBean DE, Kelly PA (1991) Alterations in hippocampal function following repeated exposure to the amphetamine derivative methylenedioxymethamphetamine (“ecstasy”). Psychopharmacology 105:113–118
Sokoloff L, Reivich M, Kennedy C, Des Rosiers MH, Patlak CS, Pettigrew KD, Sakurada O, Shinohara M (1977) The [14C]deoxyglucose method for the measurement of local cerebral glucose utilization: theory, procedure, and normal values in the conscious and anesthetized albino rat. J Neurochem 28:897–916
Sprague JE, Everman SL, Nichols DE (1998) An integrated hypothesis for the serotonergic axonal loss induced by 3,4-methylenedioxymethamphetamine. Neurotoxicology 19:427–441
Steele TD, McCann UD, Ricaurte GA (1994) 3,4-Methylenedioxymethamphetamine (MDMA, “ecstasy”): pharmacology and toxicology in animals and humans. Addiction 89:539–551
Stone DM, Stahl DC, Hanson GR, Gibb JW (1986) The effects of 3,4-methylenedioxymethamphetamine (MDMA) and 3,4-methylenedioxyamphetamine (MDA) on monoaminergic systems in the rat brain. Eur J Pharmacol 128:41–48
Svensson K, Alfoldi P, Hajos M, Rubicsek G, Johansson AM, Carlsson A, Obal F Jr (1987) Dopamine autoreceptor antagonists: effects on sleep-wake activity in the rat. Pharmacol Biochem Behav 26:123–129
Wallace TL, Gudelsky GA, Vorhees CV (2001) Alterations in diurnal and nocturnal locomotor activity in rats treated with a monoamine-depleting regimen of methamphetamine or 3,4-methylenedioxymethamphetamine. Psychopharmacology 153:321–326
Webb E, Ashton CH, Kelly P, Kamali F (1996) Alcohol and drug use in UK university students. Lancet 348:922–925
Yamamoto BK, Nash JF, Gudelsky GA (1995) Modulation of methylenedioxymethamphetamine-induced striatal dopamine release by the interaction between serotonin and gamma-aminobutyric acid in the substantia nigra. J Pharmacol Exp Ther 273:1063–1070
Acknowledgements
This study was supported by the Fifth Framework Programme of the European Community, QLG3-CT-2002-00809, the Hungarian Research Fund Grant T020500 and M27976, the Ministry of Welfare Research Grant 058/2003 and the Fund Management of Ministry of Education OMFB 01926/2002. The experiments complied with the Hungarian Governmental Regulation about animal studies (presented December 31, 1998) and the United Kingdom Animals Act (1986). We thank Professor Franz Halberg and Dr. Germaine Cornelissen (Chronobiology Laboratories, University of Minnesota, Minneapolis, Minn., USA) for their expert advice regarding parameters of circadian activity.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balogh, B., Molnar, E., Jakus, R. et al. Effects of a single dose of 3,4-methylenedioxymethamphetamine on circadian patterns, motor activity and sleep in drug-naive rats and rats previously exposed to MDMA. Psychopharmacology 173, 296–309 (2004). https://doi.org/10.1007/s00213-004-1787-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-1787-9