Abstract
Introduction
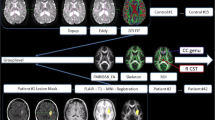
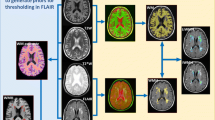
Longitudinal brain volume changes have been investigated in a number of cerebral disorders as a surrogate marker of clinical outcome. In stroke, unique methodological challenges are posed by dynamic structural changes occurring after onset, particularly those relating to the infarct lesion. We aimed to evaluate agreement between different analysis methods for the measurement of post-stroke brain volume change, and to explore technical challenges inherent to these methods.
Methods
Fifteen patients with anterior circulation stroke underwent magnetic resonance imaging within 1 week of onset and at 1 and 3 months. Whole-brain as well as grey- and white-matter volume were estimated separately using both an intensity-based and a surface watershed-based algorithm. In the case of the intensity-based algorithm, the analysis was also performed with and without exclusion of the infarct lesion. Due to the effects of peri-infarct edema at the baseline scan, longitudinal volume change was measured as percentage change between the 1 and 3-month scans. Intra-class and concordance correlation coefficients were used to assess agreement between the different analysis methods. Reduced major axis regression was used to inspect the nature of bias between measurements.
Results
Overall agreement between methods was modest with strong disagreement between some techniques. Measurements were variably impacted by procedures performed to account for infarct lesions.
Conclusions
Improvements in volumetric methods and consensus between methodologies employed in different studies are necessary in order to increase the validity of conclusions derived from post-stroke cerebral volumetric studies. Readers should be aware of the potential impact of different methods on study conclusions.








Similar content being viewed by others
References
Popescu V, Agosta F, Hulst HE et al (2013) Brain atrophy and lesion load predict long term disability in multiple sclerosis. J Neurol Neurosurg Psychiatry 84(10):1082–1091
Li Q, Pardoe H, Lichter R et al (2014) Cortical thickness estimation in longitudinal stroke studies: a comparison of 3 measurement methods. NeuroImage: Clinical. doi:10.1016/j.nicl.2014.08.017
Dang C, Liu G, Xing S et al (2013) Longitudinal cortical volume changes correlate with motor recovery in patients after acute local subcortical infarction. Stroke 44(10):2795–2801
Fox NC, Schott JM (2004) Imaging cerebral atrophy: normal ageing to Alzheimer’s disease. Lancet 363(9406):392–394
Seghier ML, Ramsden S, Lim L, Leff AP, Price CJ (2014) Gradual lesion expansion and brain shrinkage years after stroke. Stroke 45(3):877–879
Aoi MC, Hu K, Lo MT, Selim M, Olufsen MS, Novak V (2012) Impaired cerebral autoregulation is associated with brain atrophy and worse functional status in chronic ischemic stroke. PLoS One 7(10):e46794
Brodtmann A, Pardoe H, Li Q, Lichter R, Ostergaard L, Cumming T (2012) Changes in regional brain volume three months after stroke. J Neurol Sci 322:122–128
Ding G, Jiang Q, Li L et al (2010) Cerebral tissue repair and atrophy after embolic stroke in rat: a magnetic resonance imaging study of erythropoietin therapy. J Neurosci Res 88(14):3206–3214
Shen F, Walker EJ, Jiang L et al (2011) Coexpression of angiopoietin-1 with VEGF increases the structural integrity of the blood-brain barrier and reduces atrophy volume. J Cereb Blood Flow Metab 31(12):2343–2351
Klauschen F, Goldman A, Barra V, Meyer-Lindenberg A, Lundervold A (2009) Evaluation of automated brain MR image segmentation and volumetry methods. Hum Brain Mapp 30(4):1310–1327
Lansberg MG, O’Brien MW, Tong DC, Moseley ME, Albers GW (2001) Evolution of cerebral infarct volume assessed by diffusion-weighted magnetic resonance imaging. Arch Neurol 58(4):613
Wang X, Valdes Hernandez MC, Doubal F, Chappell FM, Wardlaw JM (2012) How much do focal infarcts distort white matter lesions and global cerebral atrophy measures? Cerebrovasc Dis 34(5–6):336–342
Kuceyeski A, Kamel H, Navi BB, Raj A, Iadecola C (2014) Predicting future brain tissue loss from white matter connectivity disruption in ischemic stroke. Stroke 45(3):717–722
Jenkinson M, Smith S (2001) A global optimisation method for robust affine registration of brain images. Med Image Anal 5(2):143–156
Jenkinson M, Bannister P, Brady M, Smith S (2002) Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 17(2):825–841
Wolfson L, Wakefield DB, Moscufo N et al (2013) Rapid buildup of brain white matter hyperintensities over 4 years linked to ambulatory blood pressure, mobility, cognition, and depression in old persons. J Gerontol A: Biol Med Sci 68(11):1387–1394
Smith SM, Jenkinson M, Woolrich MW et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23:S208–S219
Smith SM (2002) Fast robust automated brain extraction. Hum Brain Mapp 17(3):143–155
Zhang Y, Brady M, Smith S (2001) Segmentation of brain MR images through a hidden Markov random field model and the expectation-maximization algorithm. IEEE Trans Med Imaging 20(1):45–57
Battaglini M, Jenkinson M, De Stefano N (2012) Evaluating and reducing the impact of white matter lesions on brain volume measurements. Hum Brain Mapp 33(9):2062–2071
Reuter M, Rosas HD, Fischl B (2010) Highly accurate inverse consistent registration: a robust approach. Neuroimage 53(4):1181–1196
Segonne F, Dale AM, Busa E et al (2004) A hybrid approach to the skull stripping problem in MRI. Neuroimage 22(3):1060–1075
Fischl B, Salat DH, Busa E et al (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33(3):341–355
Fischl B, Salat DH, van der Kouwe AJ et al (2004) Sequence-independent segmentation of magnetic resonance images. Neuroimage 23(Suppl 1):S69–S84
Sled JG, Zijdenbos AP, Evans AC (1998) A nonparametric method for automatic correction of intensity nonuniformity in MRI data. IEEE Trans Med Imaging 17(1):87–97
Fischl B, Liu A, Dale AM (2001) Automated manifold surgery: constructing geometrically accurate and topologically correct models of the human cerebral cortex. IEEE Trans Med Imaging 20(1):70–80
Segonne F, Pacheco J, Fischl B (2007) Geometrically accurate topology-correction of cortical surfaces using nonseparating loops. IEEE Trans Med Imaging 26(4):518–529
Dale AM, Fischl B, Sereno MI (1999) Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage 9(2):179–194
Dale AM, Sereno MI (1993) Improved localization of cortical activity by combining EEG and MEG with MRI cortical surface reconstruction: a linear approach. J Cogn Neurosci 5:162–176
Fischl B, Dale AM (2000) Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc Natl Acad Sci U S A 97(20):11050–11055
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Smith RJ (2009) Use and misuse of the reduced major axis for line‐fitting. Am J Phys Anthropol 140(3):476–486
Winkler AM, Kochunov P, Blangero J et al (2010) Cortical thickness or grey matter volume? The importance of selecting the phenotype for imaging genetics studies. Neuroimage 53(3):1135–1146
De Stefano N, Airas L, Grigoriadis N et al (2014) Clinical relevance of brain volume measures in multiple sclerosis. CNS Drugs 28(2):147–156
Smith SM, Zhang Y, Jenkinson M et al (2002) Accurate, robust, and automated longitudinal and cross-sectional brain change analysis. Neuroimage 17(1):479–489
Jasperse B, Valsasina P, Neacsu V et al (2007) Intercenter agreement of brain atrophy measurement in multiple sclerosis patients using manually‐edited SIENA and SIENAX. J Magn Reson Imaging 26(4):881–885
Fischl B (2012) FreeSurfer. Neuroimage 62(2):774–781
Ashburner J, Friston KJ (2000) Voxel-based morphometry—the methods. Neuroimage 11(6 Pt 1):805–821
Clarkson MJ, Cardoso MJ, Ridgway GR et al (2011) A comparison of voxel and surface based cortical thickness estimation methods. Neuroimage 57(3):856–865
Ethical Standards and Patient Consent
We declare that all human and animal studies have been approved by the Melbourne Health Research Ethics Committee and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. We declare that all patients gave informed consent prior to inclusion in this study.
Conflict of Interest
We declare that we have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 3409 kb)
Rights and permissions
About this article
Cite this article
Yassi, N., Campbell, B.C.V., Moffat, B.A. et al. Know your tools—concordance of different methods for measuring brain volume change after ischemic stroke. Neuroradiology 57, 685–695 (2015). https://doi.org/10.1007/s00234-015-1522-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00234-015-1522-8