Abstract
Assessment of the diversity of algal assemblages in Antarctica has until now largely relied on traditional microbiological culture approaches. Here we used DNA metabarcoding through high-throughput sequencing (HTS) to assess the uncultured algal diversity at two sites on Deception Island, Antarctica. The first was a relatively undisturbed site within an Antarctic Specially Protected Area (ASPA 140), and the second was a site heavily impacted by human visitation, the Whalers Bay historic site. We detected 65 distinct algal taxa, 50 from within ASPA 140 and 61 from Whalers Bay. Of these taxa, 46 were common to both sites, and 19 only occurred at one site. Algal richness was about six times greater than reported in previous studies using culture methods. A high proportion of DNA reads obtained was assigned to the highly invasive species Caulerpa webbiana at Whalers Bay, and the potentially pathogenic genus Desmodesmus was found at both sites. Our data demonstrate that important differences exist between these two protected and human-impacted sites on Deception Island in terms of algal diversity, richness, and abundance. The South Shetland Islands have experienced considerable effects of climate change in recent decades, while warming through geothermal activity on Deception Island itself makes this island one of the most vulnerable to colonization by non-native species. The detection of DNA of non-native taxa highlights concerns about how human impacts, which take place primarily through tourism and national research operations, may influence future biological colonization processes in Antarctica.


Similar content being viewed by others
Data Availability
All soil samples analyzed in this paper are stored in the Laboratory of Microbiology at Universidade Federal de Minas Gerais.
References
Environment Protocol (2005) Management Plan for Antarctic Specially Protected Area No. 140. http://www.ats.aq/documents/recatt/Att291_e.pdf. Accessed 18 Feb 2020
Smellie JL (2001) Lithostratigraphy and volcanic evolution of Deception Island, South Shetland Islands. Antarct Sci 13:188–209. https://doi.org/10.1017/S0954102001000281
Smith RIL (2005) The thermophilic bryoflora of Deception Island: unique plant communities as a criterion for designating an Antarctic Specially Protected Area. Antarct Sci 17(1):17–27. https://doi.org/10.1017/S0954102005002385
Roura R (2012) Being there: examining the behaviour of Antarctic tourists through their blogs. Polar Res 31:1–23. https://doi.org/10.3402/polar.v31i0.10905
Ochyra R, Smith RIL, Bednarek-Ochyra H (2008) The illustrated moss flora of Antarctica. Cambridge University Press, Cambridge, p 685
Bednarek-Ochyra H, Vána J, Ochyra R, Smith RIL (2000) The liverwort flora of Antarctica. Polish Academy of Sciences, Institute of Botany, Cracow 236p
Convey P, Smith RIL (2006) Geothermal bryophyte habitats in the South Sandwich Islands, maritime Antarctic. J Veg Sci 17:529–538. https://doi.org/10.1111/j.1654-1103.2006.tb02474.x
Convey P, Smith RIL, Hodgson DA, Peat HJ (2000) The flora of the South Sandwich Islands, with particular reference to the influence of geothermal heating. J Biogeogr 27:1279–1295. https://doi.org/10.1046/j.1365-2699.2000.00512.x
Management Plan for Antarctic Specially Protected Area N° 140 - Parts of Deception Island, South Shetland Islands (2005). Available at: https://www.ats.aq/devph/en/apa-database/45. Accessed 20 May 2020
Downie RH, Convey P, McInnes SJ, Pugh PJA (2000) The non-marine invertebrate fauna of Deception Island (Maritime Antarctic): a baseline for a comprehensive biodiversity database. Polar Record 36(199):297–304. https://doi.org/10.1017/S0032247400016788
Greenslade P, Patopov M, Russell D, Convey P (2012) Global Collembola on Deception Island. J Insect Sci 12(1):111–116. https://doi.org/10.1673/031.012.11101
Pugh PJA, Convey P (2000) Scotia Arc Acari: antiquity and origin. Zool J Linnean Soc 130:309–328. https://doi.org/10.1111/j.1096-3642.2000.tb01633.x
Smith RIL, Richardson M (2011) Fuegian plants in Antarctica: natural or anthropogenically assisted immigrants? Biol Invasions 13:1–5. https://doi.org/10.1007/s10530-010-9784-x
Adams BJ, Bardgett RD, Ayres E, Wall DH, Aislabie J, Bamforth S, Bargagli R, Cary C, Cavacini P, Connell L, Convey P, Fell JW, Frati F, Hogg ID, Newsham KK, O'Donnell A, Russell N, Seppelt RD, Stevens MI (2005) Diversity and distribution of Victoria land biota. Soil Biol Biochem 38:3003–3018. https://doi.org/10.1016/j.soilbio.2006.04.030
Broady PA (1996) Diversity, distribution and dispersal of Antarctic terrestrial algae. Biodivers Conserv 5:1307–1335. https://doi.org/10.1007/BF00051981.pdf. Accessed 18 Feb 2020
Cavacini P (2001) Soil algae from northern Victoria Land (Antarctica). Polar Biosci 14:45- 60. https://www.researchgate.net/publication/252601941_Cavacini_P_Soil_algae_from_northern_Victoria_Land_Antarctica_Polar_Biosci_14. Accessed 18 Feb 2020
Fermani P, Mataloni G, Vijver BV (2007) Soil microalgal communities on an Antarctic active volcano (Deception Island, South Shetlands). Polar Biol 30:1381–1393. https://doi.org/10.1007/s00300-007-0299-6
Garraza GG, Mataloni G, Fermani P, Vinocur A (2011) Ecology of algal communities of different soil types from Cierva Point, Antarctic Peninsula. Polar Biol 34:339–351. https://doi.org/10.1007/s00300-010-0887-8
Mataloni G, Tell G, Wynn-Williams D (2000) Structure and diversity of soil algal communities from Cierva Point (Antarctic Peninsula). Polar Biol 23(3):205–211. https://doi.org/10.1007/s003000050028
Zidarova R (2007) Diversity and distribution of algae on Livingston Island, Antarctica. Comptes rendus de l'Académie bulgare des sciences: sciences mathématiques et naturelles 60(4):435–442. https://www.researchgate.net/publication/228490897_Algae_from_Livingston_Island_S_Shetland_Islands_a_checklist. Accessed 01 Jan 2020
Huss V, Frank C, Hartmann EC, Hirmer M (1999) Biochemical taxonomy and molecular phylogeny of the genus Chlorella sensu lato (Chlorophyta). J Phycol 35(3):587–598. https://doi.org/10.1046/j.1529-8817.1999.3530587.x
Rondon MR, August PR, Bettermann AD, Brady SF, Grossman TH, Liles MR, Loiacono KA, Lynch BA, MacNeil IA, Minor C (2000) Cloning the soil metagenome: a strategy for accessing the genetic and functional diversity of uncultured microorganisms. Appl Environ Microbiol 66(6):2541–2547. https://doi.org/10.1128/aem.66.6.2541-2547.2000
Ruppert K, Kline RJ, Rahman MS (2019) Past, present, and future perspectives of environmental DNA (eDNA) metabarcoding: a systematic review in methods, monitoring, and applications of global eDNA. Global Ecol Conserv 17:1–29. https://doi.org/10.1016/j.gecco.2019.e00547
Rippin M, Borchhardt N, Williams L, Colesie C, Jung P, Büdel B, Karsten U, Becker B (2018) Genus richness of microalgae and cyanobacteria in biological soil crusts from Svalbard and Livingston Island: morphological versus molecular approaches. Polar Biol 41:909–923. https://doi.org/10.1007/s00300-018-2252-2
Medinger R, Nolte V, Pandey RV, Jost S, Ottenwalder B, Schlotterer C, Boenigk J (2010) Diversity in a hidden world: potential and limitation of next-generation sequencing for surveys of molecular diversity of eukaryotic microorganisms. Mol Ecol 19(1):32–40. https://doi.org/10.1111/j.1365-294X.2009.04478.x
Fraser CI, Connell L, Lee CK, Cary SC (2018) Evidence of plant and animal communities at exposed and subglacial (cave) geothermal sites in Antarctica. Polar Biol 41:417–421. https://doi.org/10.1007/s00300-017-2198-9
Garrido-Benavent I, Pérez-Ortega S, Durán J, Ascaso C, Pointing SB, Rodríguez-Cielos R, Navarro F, de los Ríos A (2020) Differential colonization and succession of microbial communities in rock and soil substrates on a maritime Antarctic glacier forefield. Front Microbiol 11. https://doi.org/10.3389/fmicb.2020.00126
Leliaert F, Smith DR, Moreau H, Herron MD, Verbruggen H, Delwiche CF, De Clerck O (2012) Phylogeny and molecular evolution of the green algae. Crit Rev Plant Sci 31:1–46. https://doi.org/10.1080/07352689.2011.615705
Guiry MD, Guiry GM (2020) AlgaeBase. World-wide electronic publication, National University of Ireland, Galway. http://www.algaebase.org. Accessed 01 Jan 2020
Archer SDJ, Lee KC, Caruso T, Maki T, Lee CK, Cary SC, Cowan DA, Maestre FT, Pointing SB (2019) Airborne microbial transport limitation to isolated Antarctic soil habitats. Nat Microbiol 4:925–932. https://doi.org/10.1038/s41564-019-0370-4
Borruso L, Sannino C, Selbmann L, Battistel D, Zucconi L, Azzaro M, Turchetti B, Buzzini P, Guglielmin M (2018) A thin ice layer segregates two distinct fungal communities in Antarctic brines from Tarn Flat (Northern Victoria Land). Sci Rep 8:6582. https://doi.org/10.1038/s41598-018-25079-3
Cerqueira AES, Silva TH, Nunes ACS, Nunes DD, Lobato LC, Veloso TGR, De Paulo SO, Kasuya MCM, Silva CC (2018) Amazon basin pasture soils reveal susceptibility to phytopathogens and lower fungal community dissimilarity than forest. Appl Soil Ecol 131:1–11. https://doi.org/10.1016/j.apsoil.2018.07.004
Chen S, Yao H, Han J, Liu C, Song J, Shi L, Zhu Y, Ma X, Gao T, Pang X, Luo K, Li Y, Li X, Jia X, Lin Y, Leon C (2010) Validation of the ITS2 region as a novel DNA barcode for identifying medicinal plant species. PLoS One 5(1):e8613. https://doi.org/10.1371/journal.pone.0008613
Richardson RT, Lin C, Sponsler DB, Quijia JO, Goodell K, Johnson RM (2015) Application of ITS2 metabarcoding to determine the provenance of pollen collected by honey bees in an agroecosystem. Appl Plant Sci 3(1):1400066. https://doi.org/10.3732/apps.1400066
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Joshi NA, Fass JN (2011) Sickle: a sliding-window, adaptive, quality-based trimming tool for FastQ files (version 1.33) [software]. https://github.com/najoshi/sickle. Accessed 20 May 2020
Ankenbrand MJ, Keller A, Wolf M, Schultz J, Förster F (2015) ITS2 database V: twice as much. Mol Biol Evol 32:3030–3032. https://doi.org/10.1093/molbev/msv174
Huson DH, Beier S, Flade I, Górska A, El-Hadidi M, Mitra S et al (2016) MEGAN community edition - interactive exploration and analysis of large-scale microbiome sequencing data. PLoS Comput Biol 12:e1004957. https://doi.org/10.1371/journal.pcbi.1004957
Hammer, Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):1–9. http://palaeo-electronica.org/2001_1/past/issue1_01.htm. Accessed 01 Jan 2020
Baselga A, Orme CDL (2012) “betapart”: an R package for the study of beta diversity. Methods Ecol. Evol. 3, 808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Broady PA (1983) Taxonomic and ecological investigations of algae on steam-warmed soil on Mt Erebus, Ross Island, Antarctica. Phycologia 23(3):257–271. https://doi.org/10.2216/i0031-8884-23-3-257.1
Broady PA (1989) Survey of algae and other terrestrial biota at Edward VII Peninsula, Marie Byrd Land. Antarct Sci 1(3):215–224. https://doi.org/10.1017/S0954102089000337
Bardou P, Mariette J, Escudié F, Djemiel C, Klopp C (2014) jvenn: an interactive Venn diagram viewer. BMC Bioinforma 15:293. https://doi.org/10.1186/1471-2105-15-293
Chen W, Zhang CK, Cheng Y, Zhang S, Zhao H (2013) A comparison of methods for clustering 16S rRNA sequences into OTUs. PLoS One 8(8):e70837. https://doi.org/10.1371/journal.pone.0070837
Koeppel AF, Wu M (2013) Surprisingly extensive mixed phylogenetic and ecological signals among bacterial operational taxonomic units. Nucleic Acids Res 41:5175–5188. https://doi.org/10.1093/nar/gkt241
Darling JA, Mahon AR (2011) From molecules to management: adopting DNA-based methods for monitoring biological invasions in aquatic environments. Environ Res 111:978–988. https://doi.org/10.1016/j.envres.2011.02.001
Comtet T, Sanionigi A, Viard F, Casiraghi M (2015) DNA (meta)barcoding of biological invasions: a powerful tool to elucidate invasion processes and help managing aliens. Biol Invasions 17:905–922. https://doi.org/10.1007/s10530-015-0854-y
Weber AA, Pawlowski J (2013) Can abundance of protists be inferred from sequence data: a case study of Foraminifera. PLoS One 8:e56739. https://doi.org/10.1371/journal.pone.0056739
Giner CR, Forn I, Romac S, Logares RC, Massana R (2016) Environmental sequencing provides reasonable estimates of the relative abundance of specific picoeukaryotes. Appl Environ Microbiol 82(15):4757–4766. https://doi.org/10.1128/AEM.00560-16
Deiner K, Bik HM, Mächler E, Seymour M, Lacoursière-Roussel A, Altermatt F, Creer S, Bista I, Lodge DM, de Vere N, Pfrender ME, Bernatchez L (2017) Environmental DNA metabarcoding: transforming how we survey animal and plant communities. Mol Ecol 26:5872e5895–5872e5895. https://doi.org/10.1111/mec.14350
Hering D, Borja A, Jones JI, Pont D, Boets P, Bouchez A, Bruce K, Drakare S, Hanfling B, Kahlert M, Leese F, Meissner K, Mergen P, Reyjol Y, Segurado P, Vogler A, Kelly M (2018) Implementation options for DNA-based identification into ecological status assessment under the European Water Framework Directive. Water Res 138:192–205. https://doi.org/10.1016/j.watres.2018.03.003
Cooley DR, Mullins RF, Bradley PM, Wilce RT (2011) Culture of the upper littoral zone marine alga Pseudendoclonium submarinum induces pathogenic interaction with the fungus Cladosporium cladosporioides. Phycologia 50(5):541–547. https://doi.org/10.2216/10-84.1
Pellizzari F, Silva MC, Silva EM, Medeiros A, Oliveira MC, Yokoya NS, Pupo D, Rosa LH, Colepicolo P (2017) Diversity and spatial distribution of seaweeds in the South Shetland Islands, Antarctica: an updated database for environmental monitoring under climate change scenarios. Polar Biol 40(8):1671–1685. https://doi.org/10.1007/s00300-017-2092-5
Amat JN, Cardigos F, Santos RS (2008) The recent northern introduction of the seaweed Caulerpa webbiana (Caulerpales, Chlorophyta) in Faial, Azores Islands (north-eastern Atlantic). Aquat Invasions 3:417–422. https://doi.org/10.3391/ai.2008.3.4.7
Cardigos F, Monteiro M, Fontes J, Serrão R (2015) Fighting invasions in the marine realm, a case study with Caulerpa webbiana proliferation in the Azores. In: Canning-Clode J (ed) Biological invasions in changing ecosystems vectors, ecological impacts. Management and Predictions. De Gruyter Open Ltd, Warsaw/Berlin, pp 279–300
Westblade LF, Ranganath S, Dunne WM, Burnham CAD, Fader R, Ford BA (2015) Infection with a chlorophyllic eukaryote after a traumatic freshwater injury. N Engl J Med 372(10):982–984. https://doi.org/10.1056/NEJMc1401816
Hodač L, Hallmann C, Spitzer K, Elster J, Faßhauer F, Brinkmann N, Lepka D, Diwan V, Friedl T (2016) Widespread green algae Chlorella and Stichococcus exhibit polar-temperate and tropical-temperate biogeography. FEMS Microbiol Ecol 92(8):fiw122. https://doi.org/10.1093/femsec/fiw122
Acknowledgments
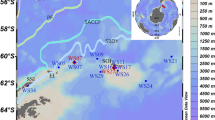
The authors thank the National Council for Scientific and Technological Development (CNPq), the Brazilian Antarctic Program (PROANTAR), Science and Technology National Institute of Cryosphere (INCT Criosfera II), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo a Pesquisa do Estado de Minas Gerais (FAPEMIG), National Fund for Scientific and Technological Development (FNDCT), Brazilian Navy, and Brazilian Congresswoman Jô Moraes and Instituto de Ciências Biológicas at University of Brasilia for their support for this research. PC is supported by NERC core funding to the BAS “Biodiversity, Evolution and Adaptation” Team. We thank Laura Gerrish, BAS Mapping and Geographic Information Centre, for preparing Fig. 1. This study also contributes to the SCAR “State of the Antarctic Ecosystem” international research program. Michael Stech for providing useful insights into the manuscript.
Funding
The research was funded by the PROANTAR, University of Brasilia Funds, and Brazilian Congresswoman Jô Moraes parliament fund.
Author information
Authors and Affiliations
Contributions
PEASC collected soil samples, optimized protocols for DNA extraction, and wrote the first version of manuscript. MCS contributed significantly in the later versions of manuscript. OHBP filtered the data and performed the metagenomic analysis once DNA information was available. ETA performed the ecological analyses. DKH helped optimizing laboratory protocols and revised all the manuscript versions. THS worked with protocol optimization and gave inputs to methodology. FP provided significant contributions to the manuscript discussion and revised all its versions. PC contributed to the result interpretations, gave important feedback to discussion and revised all manuscript versions. LHR collected the soil samples, provided the necessary infrastructure for DNA extraction, helped writing the first version of manuscript, and revised all versions.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
The collections and studies performed in ASPA 140 were authorized by the Secretariat of the Antarctic Treaty and by PROANTAR.
Code Availability
Not applicable.
Rights and permissions
About this article
Cite this article
Câmara, P.E.A.S., Carvalho-Silva, M., Pinto, O.H.B. et al. Diversity and Ecology of Chlorophyta (Viridiplantae) Assemblages in Protected and Non-protected Sites in Deception Island (Antarctica, South Shetland Islands) Assessed Using an NGS Approach. Microb Ecol 81, 323–334 (2021). https://doi.org/10.1007/s00248-020-01584-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00248-020-01584-9