Abstract
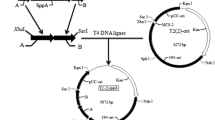
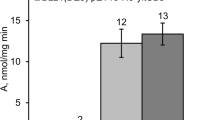
The sipM gene of Bacillus megaterium encoding a type I signal peptidase (SPase) was isolated and structurally characterized. RNA analysis revealed a transcript size in accordance with a bicistronic operon comprising sipM and an adjacent open reading frame. Inactivation of sipM by targeted gene disruption could not be achieved, indicating its essential role for cell viability since there might be no other type I SPase of major importance present in B. megaterium. Plasmid-assisted amplification of the gene resulted in an increase in activity of the heterologous glucanase used as an extracellular reporter, suggesting a potential bottleneck for protein secretion within this species.




Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Black MT (1993) Evidence that the catalytic activity of prokaryote leader peptidase depends upon the operation of a serine-lysine catalytic dyad. J Bacteriol 175:4957–4961
Bolhuis A, Tjalsma H, Smith HE, de Jong A, Meima R, Venema G, Bron S, van Dijl JM (1999) Evaluation of bottlenecks in the late stages of protein secretion in Bacillus subtilis. Appl Environ Microbiol 65:2934–2941
Borriss R, Buettner K, Maentsaelae P (1990) Structure of the beta-1,3-1,4-glucanase gene of Paenibacillus macerans: homologies to other beta-glucanases. Mol Gen Genet 222:278–283
Bron S, Bolhuis A, Tjalsma H, Holsappel S, Venema G, van Dijl JM (1998) Protein secretion and possible roles for multiple signal peptidases for precursor processing in Bacilli. J Biotechnol 64:3–13
Chu HH, Hoang V, Kreutzmann P, Hofemeister B, Melzer M, Hofemeister J (2002) Identification and properties of type I-signal peptidases of Bacillus amyloliquefaciens. Eur J Biochem 269:458–469
Cregg KM (1996) Molecular cloning and expression of the spsB gene encoding an essential type I signal peptidase from Staphylococcus aureus. J Bacteriol 178:5712–5718
Date T (1983) Demonstration by a novel genetic technique that leader peptidase is an essential enzyme of Escherichia coli. J Bacteriol 154:76–83
Debabov VG (1982) The industrial use of Bacilli. In: Dubnau DA (ed) The molecular biology of the Bacilli, vol 1. Academic Press, New York, pp 331–370
Fekkes P, Driessen AJ (1999) Protein targeting to the bacterial cytoplasmic membrane. Microbiol Mol Biol Rev 63:161–173
Ferrari E, Jarnagin AS, Schmidt BF (1993) Commercial production of extracellular enzymes. In: Sonenshein AL, Hoch JA, Losick R (eds) Bacillus subtilis and other Gram-positive bacteria. American Society for Microbiology, Washington, D.C., pp 917–937
Gärtner D, Geissendörfer M, Hillen W (1988) Expression of Bacillus subtilis xyl operon is repressed at the level of transcription and is induced by xylose. J Bacteriol 170:3102–3109
Jarnagin AS, Ferrari E (1992) Extracellular enzymes: gene regulation and structure function relationship studies. In: Doi RH, McGloughlin M (eds) Biology of Bacilli: applications to industry. Butterworth-Heinemann, Stoneham, Mass., pp 191–219
Kunst F, Ogasawara N, Moszer I, Albertini AM, Alloni G, Azevedo V, Bertero MG, Bessieres P, Bolotin A, Borchert S et al (1997) The complete genome sequence of the Gram-positive bacterium Bacillus subtilis. Nature 390:249–256
Lee JS, Wittchen KD, Stahl C, Strey J, Meinhardt F (2001) Cloning, expression, and carbon catabolite repression of the bamM gene encoding beta-amylase of Bacillus megaterium DSM319. Appl Microbiol Biotechnol 56:205–211
Meijer WJ, de Jong A, Bea G, Wisman A, Tjalsma H, Venema G, Bron S, van Dijl JM (1995) The endogenous Bacillus subtilis (natto) plasmids pTA1015 and pTA1040 contain signal peptidase-encoding genes: identification of a new structural module on cryptic plasmids. Mol Microbiol 17:621–631
Meinhardt F, Stahl U, Ebeling W (1989) Highly efficient expression of homologous and heterologous genes in Bacillus megaterium. Appl Microbiol Biotechnol 41:344–351
Miller GL (1959). Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, pp 352–355
Moran CP Jr, Lang N, LeGrice SF, Lee G, Stephens M, Sonenshein AL, Pero J, Losick R (1982) Nucleotide sequences that signal the initiation of transcription and translation in Bacillus subtilis. Mol Gen Genet 186:339–346
Morita M, Tomita K, Ishizawa M, Takagi K, Kawamura J, Takahashi H, Morino T (1999) Cloning of oxetanocin A biosynthetic and resistance genes that reside on a plasmid of Bacillus megaterium strain NK84-0128. Biosci Biotechnol Biochem 63:563–566
Nagao T, Mitamura T, Wang XH, Negoro S, Yomo T, Urabe I, Okada H (1992) Cloning, nucleotide sequences, and enzymatic properties of glucose dehydrogenase isozymes from Bacillus megaterium IAM1030. J Bacteriol 174:5013–5020
Paetzel M, Dalbey RE, Strynadka NC (2002) Crystal structure of a bacterial signal peptidase apoenzyme: implications for signal peptide binding and the Ser-Lys dyad mechanism. J Biol Chem 277:9512–9519
Peng SB, Wang L, Moomaw J, Peery RB, Sun PM, Johnson RB, Lu J, Treadway P, Skatrud PL, Wang QM (2001) Biochemical characterization of signal peptidase I from Gram-positive Streptococcus pneumoniae. J Bacteriol 183:621–627
Priest FG (1989) Products from Bacilli. In: Harwood CF (ed) Handbooks of biotechnology, vol 2. Plenum, New York, pp 293–315
Rygus T, Hillen W (1992) Catabolite repression of the xyl operon in Bacillus megaterium. J Bacteriol 174:3049–3055
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Schmiedel D, Kintrup M, Kuster E, Hillen W (1997) Regulation of expression, genetic organization and substrate specificity of xylose uptake in Bacillus megaterium. Mol Microbiol 23:1053–1062
Shaw GC, Fulco AJ (1992) Barbiturate-mediated regulation of expression of the cytochrome P450BM-3 gene of Bacillus megaterium by Bm3R1 protein. J Biol Chem 267:5515–5526
Shaw GC, Chiou CY, Chou YH, Li JM (2002) mbgA-dependent lactose utilization by Bacillus megaterium. Curr Microbiol 44:102–105
Stahl U, Esser K (1983) Plasmid heterogeneity in various strains of Bacillus megaterium. Eur J Appl Biotechnol 17:248–251
Strey J, Wittchen KD, Meinhardt F (1999) Regulation of beta-galactosidase expression in Bacillus megaterium DSM319 by a XylS/AraC-type transcriptional activator. J Bacteriol 181:3288–3292
Tjalsma H, Bolhuis A, van Roosmalen ML, Wiegert T, Schumann W, Broekhuizen CP, Quax WJ, Venema G, Bron S, van Dijl JM (1998) Functional analysis of the secretory precursor processing machinery of Bacillus subtilis: identification of a eubacterial homolog of archaeal and eukaryotic signal peptidases. Genes Dev 12:2318–2331
Tjalsma H, van den Dolder J, Meijer WJ, Venema G, Bron S, van Dijl JM (1999) The plasmid-encoded signal peptidase SipP can functionally replace the major signal peptidases SipS and SipT of Bacillus subtilis. J Bacteriol 181:2448–2454
Tjalsma H, Bolhuis A, Jongbloed JD, Bron S, van Dijl JM (2000) Signal peptide-dependent protein transport in Bacillus subtilis: a genome-based survey of the secretome. Microbiol Mol Biol Rev 64:515–547
Van Dijl JM, de Jong A, Smith H, Bron S, Venema G (1991) Signal peptidase I overproduction results in increased efficiencies of export and maturation of hybrid secretory proteins in Escherichia coli. Mol Gen Genet 227:40–48
Van Dijl JM, de Jong A, Venema G, Bron S (1995) Identification of the potential active site of the signal peptidase SipS of Bacillus subtilis. Structural and functional similarities with LexA-like proteases. J Biol Chem 270:3611–3618
Vary PS (1992) Development of genetic engineering in Bacillus megaterium: an example of the versatility and potential of industrially important Bacilli. In: Doi RH, McGloughlin M (eds) Biology of Bacilli: applications to industry. Butterworth-Heinemann, Stoneham, Mass., pp 253–310
Vary PS (1993) The genetic map of Bacillus megaterium. In: Ganesan AT, Hoch JA (eds), Genetics and biotechnology of Bacilli, vol 2. Academic Press, New York, pp 475–481
Vary PS (1994) Prime time for Bacillus megaterium. Microbiology 140:1001–1013
Vellanoweth RL, Rabinowitz JC (1992) The influence of ribosome-binding-site elements on translational efficiency in Bacillus subtilis and Escherichia coli in vivo. Mol Microbiol 6:1105–1114
Von Tersch MA, Robbins HL (1990) Efficient cloning in Bacillus megaterium: comparison to Bacillus subtilis and Escherichia coli cloning hosts. FEMS Microbiol Lett 58:305–309
Vorobjeva I, Khemel A, Alföldi I (1980) Transformation of Bacillus megaterium protoplasts by plasmid DNA. FEMS Microbiol Lett 7:261–263
Wittchen KD, Meinhardt F (1995) Inactivation of the major extracellular protease from Bacillus megaterium DSM319 by gene replacement. Appl Microbiol Biotechnol 42:871–877
Wittchen KD, Strey J, Bultmann A (1998) Molecular characterization of the operon comprising the spoIV gene of Bacillus megaterium DSM319 and generation of a deletion mutant. J Gen Appl Microbiol 44:317–326
Yanisch-Perron C, Vieira J, Messing J (1985) Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene 33:103–119
Zukowski MM (1992) Production of commercially valuable products. In: Doi RH, McGloughlin M (eds) Biology of Bacilli: applications to industry. Butterworth-Heinemann, Stoneham, Mass., pp 311–337
Acknowledgements
This work was supported by grants from the Federal Ministry of Education and Research (BMBF, Bonn-Bad Godesberg, Germany), grant no. 0319179E and 0312613. M.A. Rachman thanks the German Academic Exchange Service (DAAD, Bonn-Bad Godesberg, Germany) for financial support. The authors wish to thank K. Müller (Münster, Germany) for cloning the fragment carrying sipM in partially fulfillment of her diploma thesis, and J. Paluszynski (Münster, Germany) for checking the language.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nahrstedt, H., Wittchen, K.D., Rachman, M.A. et al. Identification and functional characterization of a type I signal peptidase gene of Bacillus megaterium DSM319. Appl Microbiol Biotechnol 64, 243–249 (2004). https://doi.org/10.1007/s00253-003-1469-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1469-2