Abstract
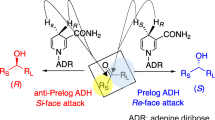
We describe an efficient method for producing both enantiomers of chiral alcohols by asymmetric hydrogen-transfer bioreduction of ketones in a 2-propanol (IPA)–water medium with E. coli biocatalysts expressing phenylacetaldehyde reductase (PAR: wild-type and mutant enzymes) from Rhodococcus sp. ST-10 and alcohol dehydrogenase from Leifsonia sp. S749 (LSADH). We also describe the detailed properties of mutant PARs, Sar268, and HAR1, which were engineered to have high activity and productivity in media composed of polar organic solvent and water, and the construction of three-dimensional structure of PAR by homology modeling. The K m and V max values for some substrates and the substrate specificity of mutant PARs were quite different from those of wild-type PAR. The results well explained the increased productivity of engineered PARs in IPA–water medium.




Similar content being viewed by others
References
Breuer M, Ditrich K, Habicher T, Hauer B, Keβeker M, Stürmer R, Zelinski T (2004) Industrial methods for the production of optically active intermediates. Angew Chem Int Eng 43:788–824
Chen K, Arnold FH (1993) Tuning the activity of an enzyme for unusual environments: sequential random mutagenesis of subtilisin E for catalysis in dimethylformamide. Proc Natl Acad Sci 90:5618–5622
Ema T, Ide S, Okita N, Sakai T (2008) Highly efficient chemoenzymatic synthesis of methyl (R)-o-chloromandelate, a key intermediate for clopidogrel, via asymmetric reduction with recombinant Escherichia coli. Adv Synth Catal 13:2039–2044
Hummel W (1997) New alcohol dehydrogenases for the synthesis of chiral compounds. Adv Biochem Eng Biotechnol 58:145–184
Hummel W, Kula MR (1989) Dehydrogenases for the synthesis of chiral compounds. Eur J Biochem 184:1–13
Inoue K, Makino Y, Itoh N (2005a) Purification and characterization of a novel alcohol dehydrogenase from Leifsonia sp. strain S749: a promising biocatalyst for an asymmetric hydrogen transfer bioreduction. Appl Environ Microbiol 71:3633–3641
Inoue K, Makino Y, Itoh N (2005b) Production of (R)-chiral alcohols by a hydrogen-transfer bioreduction with NADH-dependent Leifsonia alcohol dehydrogenase (LSADH). Tetrahedron-Asymmetry 16:2539–2549
Inoue K, Makino Y, Dairi T, Itoh N (2006) Gene cloning and expression of Leifsonia alcohol dehydrogenase (LSADH) involved in asymmetric hydrogen-transfer bioreduction to produce (R)-form chiral alcohols. Biosci Biotechnol Biochem 70:418–426
Itoh N, Yoshida K, Okada K (1996) Isolation and identification of styrene-degrading Corynebacterium strains, and their styrene metabolism. Biosci Biotechnol Biochem 60:1826–1830
Itoh N, Morihama R, Wang J-C, Okada K, Mizuguchi N (1997) Purification and characterization of phenylacetaldehyde reductase from a styrene-assimilating Corynebacterium strain ST-10. Appl Environ Microbiol 63:3783–3788
Itoh N, Mizuguchi N, Mabuchi M (1999) Production of chiral alcohols by enantioselective reduction with NADH-dependent phenylacetaldehyde reducatse from Corynebacterium strain, ST-10. J Mol Cat B Enzymatic 6:41–50
Itoh N, Matsuda M, Mabuchi M, Dairi T, Wang J-C (2002) Chiral alcohol production by NADH-dependent phenylacetaldehyde reductase coupled with in situ regeneration of NADH. Eur J Biochem 269:2394–2402
Itoh N, Nakamura M, Inoue K, Makino Y (2007) Continuous production of chiral 1,3-butanediol using immobilized biocatalysts in a packed bed reactor: promising biocatalysis method with an asymmetric hydrogen-transfer bioreduction. Appl Microbiol Biotechnol 75:1249–1256
Karabec M, Lyskowski A, Tauber KC, Steinkellner G, Kroutil W, Grogan G, Gruber K (2010) Structural insights into substrate specificity and solvent tolerance in alcohol dehydrogenase ADH-‘A’ from Rhodococcus ruber DSM 44541. Chem Comm 46:6314–6316
Kosjek B, Stampfer W, Pogorevc M, Goessler W, Faber K, Kroutil W (2004) Purification and characterization of a chemotolerant alcohol dehydrogenase applicable to coupled redox reactions. Biotechnol Bioeng 86:55–62
Kujo C, Ohshima T (1998) Enzymological characteristics of the hyperthermostable NAD-dependent glutamate dehydrogenase from the archaeon Pyrobaculum islandicum and effects of denaturants and organic solvents. Appl Environ Microbiol 64:2152–2157
Lanne C, Boeren S, Vos K, Veeger C (1987) Rules for optimization of biocatalysis in organic solvents. Biotechnol Bioeng 30:81–87
Makino Y, Inoue K, Dairi T, Itoh N (2005) Engineering of phenylacetaldehyde reductase for efficient substrate conversion in concentrated 2-propanol. Appl Environ Microbiol 71:4713–4720
Makino Y, Dairi T, Itoh N (2007) Engineering the phenylacetaldehyde reductase mutant for improved substrate conversion in the presence of concentrated 2-propanol. Appl Microbiol Biotechnol 77:833–843
Matsuyama A, Yamamoto H, Kobayashi Y (2002) Practical application of recombinant whole-cell biocatalysts for the manufacturing of pharamaceutical such as chiral alcohols. Org Process Res Dev 6:558–561
Nakamura K, Yamanaka R, Matsuda T, Harada T (2003) Recent development in asymmetric reduction of ketones with biocatalysts. Tetrahedron-Asymmetry 14:2659–2681
Nakasako M (2004) Water-protein interactions from high-resolution protein crystallography. Phil Trans Roy Soc London B 359:1191–1206
Noyori Y (1994) Asymmetric catalysis in organic synthesis. Wiley, New York
Noyori Y, Ohkuma T (2001) Asymmetric catalysis by architectural and functional molecular engineering: practical chemo- and stereoselctive hydrogenation of ketones. Angew Chem Int Eng 40:40–73
Prelog V (1964) Specification of the stereospecificity of some oxido-reductases by diamond lattice sections. Pure Appl Chem 9:119–130
Saito T, Yokozawa T, Ishizaki T, Moroi T, Sayo N, Miura T, Kumobayashi H (2001) New chiral diphosphine ligands designed to have a narrow dihedral angle in the binary backbone. Adv Synth Catal 343:264–267
Shimizu S, Kataoka M, Kita K (1998) Chiral alcohol synthesis with microbial carbonyl reductases in a water-organic solvent two-phase system. Ann N Y Acad Sci 864:87–95
Tirado-Rives J, Jorgensen WL (2006) Contribution of conformer focusing to the uncertainty in predicting free energies for protein-ligand binding. J Med Chem 49:5880–5884
Wong C-H, Drueckhammer DG, Sweers HM (1985) Enzymatic vs. fermentative synthesis: thermostable glucose dehydrogenase catalyzed regeneration of NAD(P)H for use in enzymatic synthesis. J Am Chem Soc 107:4028–4031
Wu G, Robertson DH, Brooks CL III, Vieth M (2003) Detailed analysis of grid-based molecular docking: a case study of CDOCKER-A CHARMm-based MD docking algorithm. J Comput Chem 24:1549–1562
Yamaguchi T, Takamuku T, Soper AK (2005) Neutron diffraction study on microinhomogeneities in ethanol-water mixtures. J Neutron Res 13:129–131
Yamamoto H, Mitsuhashi K, Kimoto N, Kobayashi Y, Esaki N (2005) Robust NADH-regenerator: improved alpha-haloketone-resistant formate dehydrogenase. Appl Microbiol Biotechnol 67:33–39
Zaks A, Klibanov AM (1984) Enzymatic catalysis in organic media at 100 degrees C. Science 224:1249–1251
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research (B) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan, and by a grant from the Japan Foundation for Applied Enzymology.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00253-012-3910-x.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 70 kb)
Rights and permissions
About this article
Cite this article
Itoh, N., Isotani, K., Nakamura, M. et al. Efficient synthesis of optically pure alcohols by asymmetric hydrogen-transfer biocatalysis: application of engineered enzymes in a 2-propanol–water medium. Appl Microbiol Biotechnol 93, 1075–1085 (2012). https://doi.org/10.1007/s00253-011-3447-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3447-4