Abstract
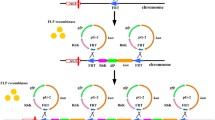
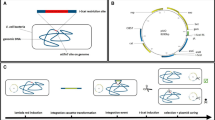
We developed a method to insert multiple desired genes into target loci on the Escherichia coli chromosome. The method was based on Red-mediated recombination, flippase and the flippase recognition target recombination, and P1 transduction. Using this method, six copies of the lacZ gene could be simultaneously inserted into different loci on the E. coli chromosome. The inserted lacZ genes were functionally expressed, and β-galactosidase activity increased in proportion to the number of inserted lacZ genes. This method was also used for metabolic engineering to generate overproducers of aromatic compounds. Important genes of the shikimate pathway (aroF fbr and tyrA fbr or aroF fbr and pheA fbr) were introduced into the chromosome to generate a tyrosine or a phenylalanine overproducer. Moreover, a heterologous decarboxylase gene was introduced into the chromosome of the tyrosine or phenylalanine overproducer to generate a tyramine or a phenethylamine overproducer, respectively. The resultant strains selectively overproduced the target aromatic compounds. Thus, the developed method is a convenient tool for the metabolic engineering of E. coli for the production of valuable compounds.





Similar content being viewed by others
References
Albermann C, Trachtmann N, Sprenger GA (2010) A simple and reliable method to conduct and monitor expression cassette integration into the Escherichia coli chromosome. Biotechnol J 5:32–38
An G (1986) Development of plant promoter expression vectors and their use for analysis of differential activity of nopaline synthase promoter in transformed tobacco cells. Plant Physiol 81:86–91
Baba T, Ara T, Hasegawa M, Takai Y, Okumura Y, Baba M, Datsenko KA, Tomita M, Wanner BL, Mori H (2006) Construction of Escherichia coli K-12 in-frame, single-gene knockout mutants: the Keio collection. Mol Syst Biol 2:2006.0008
Báez-Viveros JL, Osuna J, Hernández-Chávez G, Soberón X, Bolívar F, Gosset G (2004) Metabolic engineering and protein directed evolution increase the yield of l-phenylalanine synthesized from glucose in Escherichia coli. Biotechnol Bioeng 87:516–524
Bailey JE, Da Silva NA, Peretti SW, Seo JH, Srienc F (1986) Studies of host-plasmid interactions in recombinant microorganisms. Ann NY Acad Sci 469:194–211
Bongaerts J, Krämer M, Müller U, Raeven L, Wubbolts M (2001) Metabolic engineering for microbial production of aromatic amino acids and derived compounds. Metab Eng 3:289–300
Cherepanov PP, Wackemagel W (1995) Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158:9–14
Chiang CJ, Chen PT, Chao YP (2008) Replicon-free and markerless methods for genomic insertion of DNAs in phage attachment sites and controlled expression of chromosomal genes in Escherichia coli. Biotechnol Bioeng 101:985–995
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97:6640–6645
de Lorenzo V, Herrero M, Sanchez JM, Timmis KN (1998) Minitransposons in microbial ecology and environmental biotechnology. FEMS Microbiol Ecol 27:211–224
Diaz Ricci JC, Hernandez ME (2000) Plasmid effects on Escherichia coli metabolism. Crit Rev Biotechnol 20:79–108
Díaz E, Ferrández A, Prieto MA, García JL (2001) Biodegradation of aromatic compounds by Escherichia coli. Microbiol Mol Biol Rev 65:523–569
Haldimann A, Wanner BL (2001) Conditional-replication, integration, excision, and retrieval plasmid–host systems for gene structure–function studies of bacteria. J Bacteriol 183:6384–6393
Jones KL, Keasling JD (1998) Construction and characterization of F plasmid-based expression vectors. Biotechnol Bioeng 59:659–665
Jones KL, Kim SW, Keasling JD (2000) Low-copy plasmids can perform as well as or better than high-copy plasmids for metabolic engineering of bacteria. Metab Eng 2:328–338
Lütke-Eversloh T, Stephanopoulos G (2005) Feedback inhibition of chorismate mutase/prephenate dehydrogenase (TyrA) of Escherichia coli: generation and characterization of tyrosine-insensitive mutants. Appl Environ Microbiol 71:7224–7228
Lütke-Eversloh T, Stephanopoulos G (2007) l-Tyrosine production by deregulated strains of Escherichia coli. Appl Microbiol Biotechnol 75:103–110
Martinez-Morales F, Borges AC, Martinez A, Shanmugam KT, Ingram LO (1999) Chromosomal integration of heterologous DNA in Escherichia coli with precise removal of markers and replicons used during construction. J Bacteriol 181:7143–7148
Meynial-Salles I, Cervin MA, Soucaille P (2005) New tool for metabolic pathway engineering in Escherichia coli: One-step method to modulate expression of chromosomal genes. Appl Environ Microbiol 71:2140–2144
Murphy KC, Campellone KG, Poteete AR (2000) PCR-mediated gene replacement in Escherichia coli. Gene 246:21–330
Nelms J, Edwards RM, Warwick J, Fotheringham I (1992) Novel mutations in the pheA gene of Escherichia coli K-12 which result in highly feedback inhibition-resistant variants of chorismate mutase/prephenate dehydratase. Appl Environ Microbiol 58:2592–2598
Olson MM, Templeton LJ, Suh W, Youderian P, Sariaslani FS, Gatenby AA, Van Dyk TK (2007) Production of tyrosine from sucrose or glucose achieved by rapid genetic changes to phenylalanine-producing Escherichia coli strains. Appl Microbiol Biotechnol 74:1031–1040
Parrott S, Jones S, Cooper RA (1987) 2-Phenylethylamine catabolism by Escherichia coli K12. J Gen Microbiol 133:347–351
Peredelchuk MY, Bennett GN (1997) A method for construction of E. coli strains with multiple DNA insertions in the chromosome. Gene 187:231–238
Pittard J, Camakaris H, Yang J (2005) The TyrR regulon. Mol Microbiol 55:16–26
Posfai G, Koob M, Hradecna Z, Hasan N, Filutowicz M, Szybalski W (1994) In vivo excision and amplification of large segments of the Escherichia coli genome. Nucleic Acids Res 22:2392–2398
Qi WW, Vannelli T, Breinig S, Ben-Bassat A, Gatenby AA, Haynie SL, Sariaslani FS (2007) Functional expression of prokaryotic and eukaryotic genes in Escherichia coli for conversion of glucose to p-hydroxystyrene. Metab Eng 9:268–276
Ramos CR, Abreu PA, Nascimento AL, Ho PL (2004) A high-copy T7 Escherichia coli expression vector for the production of recombinant proteins with a minimal N-terminal His-tagged fusion peptide. Braz J Med Biol Res 37:1103–1109
Rong R, Slupska MM, Chiang JH, Miller JH (2004) Engineering large fragment insertions into the chromosome of Escherichia coli. Gene 336:73–80
Sambrook J, Fritsch EF, Maniatis T (1998) Molecular cloning, a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor
Sariaslani FS (2007) Development of a combined biological and chemical process for production of industrial aromatics from renewable resources. Annu Rev Microbiol 61:51–69
Shevchenko Y, Bouffard GG, Butterfield YSN, Blaskesley RW, Hartley JL, Young AC, Marra MA, Jones SJM, Touchman JW, Green ED (2002) Systematic sequencing of cDNA clones using the transposon Tn5. Nucleic Acids Res 30:2469–2477
Sternberg N (1990) Bacteriophage P1 cloning system for the isolation, amplification, and recovery of DNA fragments as large as 100 kilobase pairs. Proc Natl Acad Sci USA 87:103–107
Sternberg N, Hoess R (1983) The molecular genetics of bacteriophage P1. Annu Rev Genet 17:123–154
Tischer BK, von Einem J, Kaufer B, Osterrieder N (2006) Two-step Red-mediated recombination for versatile high-efficiency markerless DNA manipulation in Escherichia coli. Biotechniques 40:191–196
Vannelli T, Qi WW, Sweigard J, Gatenby AA, Sariaslani FS (2007) Production of p-hydroxycinnamic acid from glucose in Saccharomyces cerevisiae and Escherichia coli by expression of heterologous genes from plants and fungi. Metab Eng 9:142–151
Wang Z, Xiang L, Shao J, Wegrzyn A, Wegrzyn G (2006) Effects of the presence of ColE1 plasmid DNA in Escherichia coli on the host cell metabolism. Microb Cell Fact 5:34
Yoon YG, Cho JH, Kim SC (1998) Cre/LoxP-mediated excision and amplification of large segments of the Escherichia coli genome. Genet Anal 14:89–95
Yu D, Ellis HM, Lee EC, Jenkins NA, Copeland NG, Court DL (2000) An efficient recombination system for chromosome engineering in Escherichia coli. Proc Natl Acad Sci USA 97:5978–5983
Yu D, Sawitzke JA, Ellis H, Court DL (2003) Recombineering with overlapping single-stranded DNA oligonucleotides: testing a recombination intermediate. Proc Natl Acad Sci USA 100:7207–7212
Yuan LZ, Rouviere PE, Larossa RA, Suh W (2006) Chromosomal promoter replacement of the isoprenoid pathway for enhancing carotenoid production in E. coli. Metab Eng 8:79–90
Acknowledgments
This work was partially supported by a Grant-in-Aid for Young Scientists (B) (19780082 and 21780105).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Primers used in this study (DOC 113 kb)
Rights and permissions
About this article
Cite this article
Koma, D., Yamanaka, H., Moriyoshi, K. et al. A convenient method for multiple insertions of desired genes into target loci on the Escherichia coli chromosome. Appl Microbiol Biotechnol 93, 815–829 (2012). https://doi.org/10.1007/s00253-011-3735-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3735-z