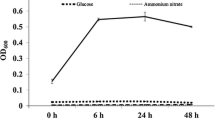
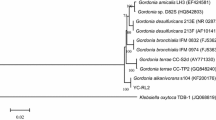
Abstract
Phenol accounts for a large proportion of the contamination in industrial wastewater discharged from chemical plants due to its wide use as a raw chemical. Residual phenol waste in water and soil significantly endangers human health and the natural environment. In this study, an Acinetobacter radioresistens strain, APH1, was isolated and identified for its efficient capability of utilizing phenol as sole carbon source for growth. A draft genome sequence containing 3,290,330 bases with 45 contigs was obtained, and 22 genes were found to be involved in phenol metabolism and 51 putative drug-resistance genes were annotated by genomic analysis. The optimal conditions for cell culture and phenol removal were determined to be 30 °C, pH 6.0, and a phenol concentration of 500 mg/L; the upper limit of phenol tolerance was 950 mg/L. Based on GC-MS analysis, the key metabolites including cis,cis-muconic acid, catechol, and succinic acid were detected. During bioremediation experiment using 450 mg/kg (dry weight) of phenol-contaminated soil, the strain APH1 removed 99% of the phenol within 3 days. According to microbial diversity analysis, the microbial abundance of Chungangia, Bacillus, Nitrospira, Lysinibacillus, and Planomicrobium increased after the addition of phenol. Furthermore, at day 23, the abundance of strain APH1 was greatly reduced, and the microbial diversity and structure of the whole microbial community were gradually recovered, indicating that strain APH1 would not affect this microbial ecosystem. These findings provide insights into the bioremediation of soil contaminated with phenol.



Similar content being viewed by others
References
Baek KH, Yoon BD, Kim BH, Cho DH, Lee IS, Oh HM, Kim HS (2007) Monitoring of microbial diversity and activity during bioremediation of crude oil-contaminated soil with different treatments. J Microbiol Biotechnol 17(1):67–73
Banerjee A, Ghoshal AK (2010) Phenol degradation by Bacillus cereus: pathway and kinetic modeling. Bioresour Technol 101(14):5501–5507. https://doi.org/10.1016/j.biortech.2010.02.018
Bhattacharya A, Gupta A, Kaur A, Malik D (2015) Assessment of phenol-degrading ability of Acinetobacter sp. B9 for its application in bioremediation of phenol-contaminated industrial effluents. Chem Ecol 31(7):607–621. https://doi.org/10.1080/02757540.2015.1075517
Blair JM, Webber MA, Baylay AJ, Ogbolu DO, Piddock LJ (2015) Molecular mechanisms of antibiotic resistance. Nat Rev Microbiol 13(1):42–51. https://doi.org/10.1038/nrmicro3380
Cetinkaya AY, Ozdemir OK (2018) Phenol removal from synthetic solution using low pressure membranes coated with graphene oxide and carbon. Chem Pap 72(2):327–335. https://doi.org/10.1007/s11696-017-0282-9
Chen BY, Chen WM, Wu YP, Hsueh CC, Lin KL, Chang CT (2010) Revealing phenol tolerance of indigenous phenol degraders isolated from Northeast Taiwan. J Taiwan Inst Chem Eng 41(6):636–643. https://doi.org/10.1016/j.jtice.2010.01.010
Clinical and Laboratory Standards Institute (CLSI) (2019) Performance standards for antimicrobial susceptibility testing, 29th ed. CLSI supplement M100. Clinical and Laboratory Standards Institute, Wayne, PA. https://clsi.org/standards/products/microbiology/documents/m100/. Accessed 17 November 2019
Daims H, Nielsen JL, Nielsen PH, Schleifer KH, Wagner M (2001) In situ characterization of Nitrospira-like nitrite-oxidizing bacteria active in wastewater treatment plants. Appl Environ Microbiol 67(11):5273–5284. https://doi.org/10.1128/AEM.67.11.5273-5284.2001
Das R, Tiwary BN (2013) Isolation of a novel strain of Planomicrobium chinense from diesel contaminated soil of tropical environment. J Basic Microbiol 53(9):723–732. https://doi.org/10.1002/jobm.201200131
El Gaidoumi A, Dona-Rodriguez JM, Melian EP, Gonzalez-Diaz OM, El Bali B, Navio JA, Kherbeche A (2019) Mesoporous pyrophyllite-titania nanocomposites: synthesis and activity in phenol photocatalytic degradation. Res Chem Intermed 45(2):333–353. https://doi.org/10.1007/s11164-018-3605-8
Environment and Climate Change Canada (ECCC) (2000) Canadian Environmental Protection Act: priority substances list. http://www.ec.gc.ca/ese-ees/default.asp?lang=En&n=80F69419-1. Accessed 17 November 2019
Gheni SA, Ahmed SMR, Abdulla AN, Mohammed WT (2018) Catalytic wet air oxidation and neural network modeling of high concentration of phenol compounds in wastewater. Environ Process 5(3):593–610. https://doi.org/10.1007/s40710-018-0323-6
Iqbal A, Arshad M, Hashmi I, Karthikeyan R, Gentry TJ, Schwab AP (2018) Biodegradation of phenol and benzene by endophytic bacterial strains isolated from refinery wastewater-fed Cannabis sativa. Environ Technol 39(13):1705–1714. https://doi.org/10.1080/09593330.2017.1337232
Jiang Y, Wen JP, Lan L, Hu ZD (2007) Biodegradation of phenol and 4-chlorophenol by the yeast Candida tropicalis. Biodegradation 18(6):719–729. https://doi.org/10.1007/s10532-007-9100-3
Ke Q, Zhang YG, Wu XL, Su XM, Wang YY, Lin HJ, Mei RW, Zhang Y, Hashmi MZ, Chen CJ, Chen JR (2018) Sustainable biodegradation of phenol by immobilized Bacillus sp. SAS19 with porous carbonaceous gels as carriers. J Environ Manag 222:85–189. https://doi.org/10.1016/j.jenvman.2018.05.061
Keith LH, Telliard WA (1979) Priority pollutants. I. A perspective view. Environ Sci Technol 13:416–423. https://doi.org/10.1021/es60152a601
Lee SY, Kim BN, Han JH, Chang ST, Choi YW, Kim YH, Min J (2010) Treatment of phenol-contaminated soil by Corynebacterium glutamicum and toxicity removal evaluation. J Hazard Mater 182(1–3):937–940. https://doi.org/10.1016/j.jhazmat.2010.06.092
Leonard D, Lindley ND (1999) Growth of Ralstonia eutropha on inhibitory concentrations of phenol: diminished growth can be attributed to hydrophobic perturbation of phenol hydroxylase activity. Enzyme Microb Technol 25(3–5):271–277. https://doi.org/10.1016/S0141-0229(99)00039-3
Liu Z, Xie W, Li D, Peng Y, Li Z, Liu S (2016) Biodegradation of phenol by bacteria strain Acinetobacter Calcoaceticus PA isolated from phenolic wastewater. Int J Environ Res Public Health 13(3):300. https://doi.org/10.3390/ijerph13030300
Nesvera J, Rucka L, Patek M (2015) Catabolism of phenol and its derivatives in bacteria: genes, their regulation, and use in the biodegradation of toxic pollutants. Adv Appl Microbiol 93:107–160. https://doi.org/10.1016/bs.aambs.2015.06.002
Nuhoglu A, Yalcin B (2005) Modelling of phenol removal in a batch reactor. Process Biochem 40(3–4):1233–1239. https://doi.org/10.1016/j.procbio.2004.04.003
Powlowski J, Shingler V (1994) Genetics and biochemistry of phenol degradation by Pseudomonas sp. CF600. Biodegradation 5(3–4):219–236. https://doi.org/10.1007/bf00696461
Qu YY, Ma Q, Liu ZY, Wang WW, Tang HZ, Zhou JT, Xu P (2017) Unveiling the biotransformation mechanism of indole in a Cupriavidus sp. strain. Mol Microbiol 106(6):905–918. https://doi.org/10.1111/mmi.13852
Sepehr S, Shahnavaz B, Asoodeh A, Karrabi M (2019) Biodegradation of phenol by cold-tolerant bacteria isolated from alpine soils of Binaloud Mountains in Iran. J Environ Sci Health A Tox Hazard Subst Environ Eng 54(4):367–379. https://doi.org/10.1080/10934529.2018.1553818
Shourian M, Noghabi KA, Zahiri HS, Bagheri T, Karballaei G, Mollaei M, Rad I, Ahadi S, Raheb J, Abbasi H (2009) Efficient phenol degradation by a newly characterized Pseudomonas sp. SA01 isolated from pharmaceutical wastewaters. Desalination 246(1–3):577–594. https://doi.org/10.1016/j.desal.2008.07.015
Singh U, Arora NK, Sachan P (2018) Simultaneous biodegradation of phenol and cyanide present in coke-oven effluent using immobilized Pseudomonas putida and Pseudomonas stutzeri. Braz J Microbiol 49(1):38–44. https://doi.org/10.1016/j.bjm.2016.12.013
Straube G, Hensel J, Niedan C, Straube E (1990) Kinetic studies of phenol degradation by Rhodococcus sp. P1. I. Batch cultivation. Antonie Van Leeuwenhoek 57(1):29–32. https://doi.org/10.1007/BF00400332
Tu C, Teng Y, Luo YM, Li XH, Sun XH, Li ZG, Liu WX, Christie P (2011) Potential for biodegradation of polychlorinated biphenyls (PCBs) by Sinorhizobium meliloti. J Hazard Mater 186(2–3):1438–1444. https://doi.org/10.1016/j.jhazmat.2010.12.008
Wang YJ, Song J, Zhao W, He XL, Chen J, Xiao M (2011) In situ degradation of phenol and promotion of plant growth in contaminated environments by a single Pseudomonas aeruginosa strain. J Hazard Mater 192(1):354–360. https://doi.org/10.1016/j.jhazmat.2011.05.031
Wu YC, Luo YM, Zou DX, Ni JZ, Liu WX, Teng Y, Li ZG (2008) Bioremediation of polycyclic aromatic hydrocarbons contaminated soil with Monilinia sp.: degradation and microbial community analysis. Biodegradation 19(2):247–257. https://doi.org/10.1007/s10532-007-9131-9
Wynands B, Lenzen C, Otto M, Koch F, Blank LM, Wierckx N (2018) Metabolic engineering of Pseudomonas taiwanensis VLB120 with minimal genomic modifications for high-yield phenol production. Metab Eng 47:121–133. https://doi.org/10.1016/j.ymben.2018.03.011
Zhao B, He YL, Hughes J, Zhang XF (2010) Heterotrophic nitrogen removal by a newly isolated Acinetobacter calcoaceticus HNR. Bioresour Technol 101(14):5194–5200. https://doi.org/10.1016/j.biortech.2010.02.043
Zhou Y, Nemati M (2018) Treatment of waters contaminated by phenol and cresols in circulating packed bed bioreactors-biodegradation and toxicity evaluations. Water Air Soil Pollut 229(9):288–214. https://doi.org/10.1007/s11270-018-3949-0
Funding
This work was supported by grants from the Science and Technology Commission of Shanghai Municipality (17JC1403300), National Key Research and Development Project (SQ2018YFA090024); by the “Shuguang Program” (17SG09) supported by Shanghai Education Development Foundation and Shanghai Municipal Education Commission; by grants from the Chinese National Science Foundation for Excellent Young Scholars (31422004); and by grants from the National Natural Science Foundation of Heilongjiang Province (ZD2018005).
Author information
Authors and Affiliations
Contributions
YF Liu and HZ Tang designed the experiments. YF Liu and WW Wang performed the experiments. YF Liu and HZ Tang wrote the paper. YF Liu, WW Wang, Shah SB, and Giulio Zanaroli analyzed the data. HZ Tang and P Xu contributed the reagents and materials. All of the authors reviewed the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 648 kb)
Rights and permissions
About this article
Cite this article
Liu, Y., Wang, W., Shah, S.B. et al. Phenol biodegradation by Acinetobacter radioresistens APH1 and its application in soil bioremediation. Appl Microbiol Biotechnol 104, 427–437 (2020). https://doi.org/10.1007/s00253-019-10271-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10271-w