Abstract
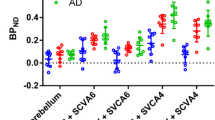
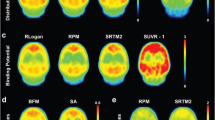
Due to the stochastic nature of radioactive decay, any measurement of radioactivity concentration requires spatial averaging. In pharmacokinetic analysis of time-activity curves (TAC), such averaging over heterogeneous tissues may introduce a systematic error (heterogeneity error) but may also improve the accuracy and precision of parameter estimation. In addition to spatial averaging (inevitable due to limited scanner resolution and intended in ROI analysis), interindividual averaging may theoretically be beneficial, too. The aim of this study was to investigate the effect of such averaging on the binding potential (BP) calculated with Logan’s non-invasive graphical analysis and the “simplified reference tissue method” (SRTM) proposed by Lammertsma and Hume, on the basis of simulated and measured positron emission tomography data {[11C]d-threo-methylphenidate (dMP) and [11C]raclopride (RAC) PET}. dMP was not quantified with SRTM since the low k 2 (washout rate constant from the first tissue compartment) introduced a high noise sensitivity. Even for considerably different shapes of TAC (dMP PET in parkinsonian patients and healthy controls, [11C]raclopride in patients with and without haloperidol medication) and a high variance in the rate constants (e.g. simulated standard deviation of K 1=25%), the BP obtained from average TAC was close to the mean BP (error <5%). However, unfavourably distributed parameters, especially a correlated large variance in two or more parameters, may lead to larger errors. In Monte Carlo simulations, interindividual averaging before quantification reduced the variance from the SRTM (beyond a critical signal to noise ratio) and the bias in Logan’s method. Interindividual averaging may further increase accuracy when there is an error term in the reference tissue assumption E=DV 2−DV′ (DV 2 = distribution volume of the first tissue compartment, DV′ = distribution volume of the reference tissue). This can be explained by the fact that the distribution volume ratio (DVR=DV/DV′) obtained from averaged TAC is an approximation for ΣDV/ΣDV′ rather than for ΣDVR/n. We conclude that Logan’s non-invasive method and SRTM are suitable for heterogeneous tissues and that discussion of group differences in PET studies generally should include qualitative and quantitative assessment of interindividually averaged TAC.





Similar content being viewed by others
References
Herholz K, Patlak CS. The influence of tissue heterogeneity on results of fitting nonlinear model equations to regional tracer uptake curves: with an application to compartmental models used in positron emission tomography. J Cereb Blood Flow Metab 1987; 7:214–229.
Herholz K, Wienhard K, Heiss W-D. Validity of PET studies in brain tumors. Cerebrovasc Brain Metab 1990; 2:240–265.
Blomqvist G, Lammertsma AA, Mazoyer B, Wienhard K. Effect of tissue heterogeneity on quantification in positron emission tomography. Eur J Nucl Med 1995; 22:652–663.
Yokoi T, Kanno I, Iida H, Miura S, Uemura K. A new approach of weighted integration technique based on accumulated images using dynamic PET and15H2O. J Cereb Blood Flow Metab 1991; 11:492–501.
Schmidt K, Mies G, Sokoloff L. Model of kinetic behavior of deoxyglucose in heterogeneous tissues in brain: a reinterpretation of the significance of parameters fitted to homogeneous tissue models. J Cereb Blood Flow Metab 1991; 11:10–24.
Schmidt K, Lucignani RM, Moresco RM, Rizzo G, Gilardi MC, Messa C, Colombo F, Fazio F, Sokoloff L. Errors introduced by tissue heterogeneity in estimation of local cerebral glucose utilization with current kinetic models of the [18F]fluorodeoxyglucose method. J Cereb Blood Flow Metab 1992; 12:823–834.
Rousset OG, Deep P, Kuwabara H, Evans AC, Gjedde AH, Cumming P. Effect of partial volume correction on estimates of the influx and cerebral metabolites of 6-[(18)F]fluoro-l-dopa studied with PET in normal control and Parkinson’s disease subjects. Synapse 2000; 37:81–89.
Logan J, Fowler JS, Volkow ND, Wang G-J, Ding Y-S, Alexoff DL. Distribution volume ratios without blood sampling from graphical analysis of PET data. J Cereb Blood Flow Metab 1996; 16:834–840.
Lammertsma AA, Hume SP. Simplified reference tissue model for PET receptor studies. Neuroimage 1996; 4:153–158.
Laruelle M, Slifstein M, Huang Y. Positron emission tomography: imaging and quantification of neurotransporter availability. Methods 2002; 27:287–299.
Müller-Schauenburg W. A new method for multi-compartment pharmacokinetic analysis: the eigenvector decomposition principle. Eur J Clin Pharmacol 1973; 6:203–206.
Carson RE, Channing MA, Blasberg RG, Dunn BB, Cohen RM, Rice KC, Herscovitch P. Comparison of bolus and infusion methods for receptor quantification: application to [18F]cyclofoxy and positron emission tomography. J Cereb Blood Flow Metab 1993; 13:24–42.
Logan J. Graphical analysis of PET data applied to reversible and irreversible tracers. Nucl Med Biol 2000; 27:661–670.
Logan J, Fowler JS, Volkow ND, Wolf AP, Dewey SL, Schlyer DJ, Macgregor RR, Hitzmann R, Bendriem B, Gatley SJ, Christman DR. Graphical analysis of reversible radioligand binding from time-activity measurements applied to [N-11C-methyl]-(-)-cocaine PET studies in human subjects. J Cereb Blood Flow Metab 1990; 10:740–747.
Lammertsma AA, Bench CJ, Hume SP, Osman S, Gunn K, Brooks DJ, Frackowiak RSJ. Comparison of methods for analysis of clinical [11C]raclopride studies. J Cereb Blood Flow Metab 1996; 16:42–52.
Blomqvist G. On the construction of functional maps in positron emission tomography. J Cereb Blood Flow Metab 1984; 4:629–632.
Evans AC. A double integral form of the three-compartment model, four rate-constant model for faster generation of parameter maps. J Cereb Blood Flow Metab 1987; 7(Suppl 1):S453.
Ichise M, Ballinger JR, Golan H, Vines D, Luong A, Tsai S, Kung HF. Noninvasive quantification of dopamine D2 receptors with iodine-123-IBF SPECT. J Nucl Med. 1996; 37:513–520.
Gjedde A. Calculation of cerebral glucose phosphorylation from brain uptake of glucose analogs in vivo: a re-examination. Brain Res 1982; 4:237–274.
Patlak CS, Blasberg RG, Fenstermacher JD. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. J Cereb Blood Flow Metab 1983; 3:1–7.
Carson RE. PET parameter estimation using linear integration methods: bias and variability consideration. In: Uemura K, Lassen NA, Jones T, Kanno I, eds. Quantification of brain function: tracer kinetics and image analysis in brain PET. Amsterdam: Elsevier Science; 1993:499–507.
Slifstein M, Laruelle M. Effects of statistical noise on graphic analysis of PET neuroreceptor studies. J Nucl Med 2000; 41:2083–2088.
Varga J, Szabo Z. Modified regression model for the Logan plot. J Cereb Blood Flow Metab 2002; 22:240–244.
Ichise M, Toyama H, Innis RB, Carson RE. Strategies to improve neuroreceptor parameter estimation by linear regression analysis. J Cereb Blood Flow Metab 2002; 22:1271–1281.
Ichise M, Liow JS, Lu JQ, Takano A, Model K, Toyama H, Suhara T, Suzuki K, Innis RB, Carson RE. Linearized reference tissue parametric imaging methods: application to [11C]DASB positron emission tomography studies of the serotonin transporter in human brain. J Cereb Blood Flow Metab 2003; 23:1096-1112.
Feng D, Wang ZZ, Huang SC. A study on statistically reliable and computationally efficient algorithms for the generation of local cerebral blood flow parametric images with positron emission tomography. IEEE Trans Med Imag 1993; 12:182–188.
Feng D, Huang SC, Wang ZZ, Ho D. An unbiased parametric imaging algorithm for nonuniformly sampled biomedical system parameter estimation. IEEE Trans Med Imag 1996; 15:512–518.
Logan J, Fowler JS, Volkow ND, Ding YS, Wang GJ, Alexoff DL. A strategy for removing the bias in the graphical analysis methodJ Cereb Blood Flow Metab 2001 21:307–320.
Buchert R, Wilke F, van den Hoff J, Mester J. Improved statistical power of the multilinear reference tissue approach to the quantification of neuroreceptor ligand binding by regularization. J Cereb Blood Flow Metab 2003; 23:612–620.
Ehrin E, Gawell L, Högberg T, de Paulis S, Ström P. Synthesis of [methoxy-3H]- and [methoxy-11C]-labelled raclopride. Specific dopamine-D2 receptor ligands. J Labelled Compd Radiopharm 1987; 24:931–940.
Ding YS, Sugano Y, Fowler JS, Salata C. Synthesis of the racemate and individual enantiomeres of [11C]methylphenidate for studying presynaptic dopaminergic neuron with positron emission tomography. J Labelled Compd Radiopharm 1994; 34:989–997.
Lammertsma AA. In: Maguire RP, Leenders KL, eds. PET pharmacokinetic course manual. Groningen, The Netherlands: University of Groningen, 2002.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A. Symbols
Throughout the paper, primed variables refer to the reference tissue
BP (unitless) | Binding potential, here defined as k 3 /k 4 |
C(t) (kBq/ml) | Decay-corrected radioactivity concentration (target region), optionally subdivided into C=C 2 +C 3 (first and second tissue compartments) Any concentration denoted C̄refers to true steady state |
DV (mlblood mltissue −1) | Total distribution volume (target region), optionally subdivided into DV=DV 2 +DV 3 (first and second tissue compartments) |
DVR (unitless) | Distribution volume ratio DVR=DV/DV′ |
IRF(t) (mlblood mltissue −1 s−1] | Impulse response function |
K 1 (mlblood mltissue −1 min−1), k 2, k 3, k 4 (min−1) | Transfer rate constants within the two-tissue compartment model |
.
Appendix B. “Heterogeneity effect” from variable plasma curve
For different individual plasma curves C Plasma =ΔC Plasma + mean{C Plasma } one obtains according to Eq. 2:
The heterogeneity effect ΔC Tissue (target and reference region each) according to Eq. 9can be calculated from:
with
Thus ΔC Tissue becomes small if ΔC Plasma is not correlated with ΔIRF or if interindividual differences in the IRF or C Plasma are small.
Appendix C. Mean{DV}/mean{DV’} versus mean{DVR}
From DV = DV 2·(1+BP), the mean{DV} can be formulated as:
with:
If ΔDV 2 is not correlated with BP across the patients, the rightmost term in Eq. 12 becomes small and one can approximate:
If an error term E in the reference tissue assumption is assumed:
one obtains:
However, if E is distributed symmetrically (mean{E} ≈0):
and together with Eq. 13:
Rights and permissions
About this article
Cite this article
Reimold, M., Mueller-Schauenburg, W., Becker, G.A. et al. Non-invasive assessment of distribution volume ratios and binding potential: tissue heterogeneity and interindividually averaged time-activity curves. Eur J Nucl Med Mol Imaging 31, 564–577 (2004). https://doi.org/10.1007/s00259-003-1389-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-003-1389-5