Abstract
Purpose
The purpose of this study was to monitor the metabolic effects of temozolomide (TMZ) chemotherapy in malignant gliomas by means of repeated positron emission tomography (PET) with [11C]methionine (MET).
Methods
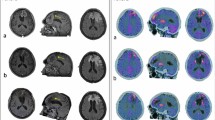
Fifteen patients with histologically proven malignant glioma were treated by TMZ chemotherapy. MET-PET studies were performed before and after the third cycle of TMZ chemotherapy in all patients, and in 12 patients also after the sixth cycle. Gadolinium-enhanced MRI studies were performed in 12 patients before the first and after the sixth cycle. Clinical status was assessed by the modified Rankin scale. Long-term outcome was assessed by calculating the time to progression (TTP) in months.
Results
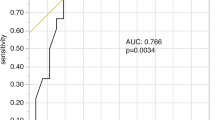
Decline in MET uptake during therapy corresponded to a stable clinical status. The median TTP was significantly longer in patients with decline in MET uptake than in those with increasing MET uptake (23 vs 3.5 months; p=0.01, log rank test). There was no significant correlation between change in MET uptake and change in contrast enhancement during treatment for all patients.
Conclusion
The present data demonstrate that clinical stability, which is often achieved under TMZ chemotherapy of malignant glioma, corresponds to a decline in or stability of tumour amino acid metabolism. Tumour responses can already be demonstrated with MET-PET after three cycles of chemotherapy, and absence of progression at that time indicates a high probability of further stability during the next three cycles. A reduction in MET uptake during TMZ treatment predicts a favourable clinical outcome. Molecular imaging of amino acid uptake by MET-PET offers a new method of measurement of the biological activity of recurrent glioma.





Similar content being viewed by others
References
Stupp R, Gander M, Leyvraz S, Newlands E. Current and future developments in the use of temozolomide for the treatment of brain tumours. Lancet Oncol 2001;2(9):552–60
DeAngelis LM. Benefits of adjuvant chemotherapy in high-grade gliomas. Semin Oncol 2003;30 (6 Suppl 19):15–18
Ataman F, Poortmans P, Stupp R, Fisher B, Mirimanoff RO. Quality assurance of the EORTC 26981/22981; NCIC CE3 intergroup trial on radiotherapy with or without temozolomide for newly-diagnosed glioblastoma multiforme: the individual case review. Eur J Cancer 2004;40(11):1724–30
Burton EC, Prados MD. Malignant gliomas. Curr Treat Options Oncol 2000;1(5):459–68
Grant R, Liang BC, Slattery J, Greenberg HS, Junck L. Chemotherapy response criteria in malignant glioma. Neurology 1997;48(5):1336–40
Levivier M, Becerra A, De Witte O, Brotchi J, Goldman S. Radiation necrosis or recurrence. J Neurosurg 1996;84(1):148–9
Cairncross JG, Macdonald DR, Pexman JH, Ives FJ. Steroid-induced CT changes in patients with recurrent malignant glioma. Neurology 1988;38(5):724–6
Ostergaard L, Hochberg FH, Rabinov JD, Sorensen AG, Lev M, Kim L, et al. Early changes measured by magnetic resonance imaging in cerebral blood flow, blood volume, and blood-brain barrier permeability following dexamethasone treatment in patients with brain tumors. J Neurosurg 1999;90(2):300–5
Alavi JB, Alavi A, Chawluk J, Kushner M, Powe J, Hickey W, et al. Positron emission tomography in patients with glioma. A predictor of prognosis. Cancer 1988;62(6):1074–8
Brock CS, Young H, O’Reilly SM, Matthews J, Osman S, Evans H, et al. Early evaluation of tumour metabolic response using [18F]fluorodeoxyglucose and positron emission tomography: a pilot study following the phase II chemotherapy schedule for temozolomide in recurrent high-grade gliomas. Br J Cancer 2000;82(3):608–15
Kubota R, Yamada S, Kubota K, Ishiwata K, Tamahashi N, Ido T. Intratumoral distribution of fluorine-11-fluorodeoxyglucose in vivo: high accumulation in macrophages and granulation tissues studied by microautoradiography. J Nucl Med 1992;33(11):1972–80
Kaschten B, Stevenaert A, Sadzot B, Deprez M, Degueldre C, Del Fiore G, et al. Preoperative evaluation of 54 gliomas by PET with fluorine-11-fluorodeoxyglucose and/or carbon-11-methionine. J Nucl Med 1998;39(5):778–85
Nuutinen J, Sonninen P, Lehikoinen P, Sutinen E, Valavaara R, Eronen E, et al. Radiotherapy treatment planning and long-term follow-up with [11C]methionine PET in patients with low-grade astrocytoma. Int J Radiat Oncol Biol Phys 2000;48(1):43–52
Würker M, Herholz K, Voges J, Pietrzyk U, Treuer H, Bauer B, et al. Glucose consumption and methionine uptake in low-grade gliomas after iodine-125 brachytherapy. Eur J Nucl Med 1996;23(5):583–6
Voges J, Herholz K, Holzer T, Würker M, Bauer B, Pietrzyk U, et al. 11C-methionine and 18F-2-fluorodeoxyglucose positron emission tomography: a tool for diagnosis of cerebral glioma and monitoring after brachytherapy with 125I seeds. Stereotact Funct Neurosurg 1997;69(1–4 Pt 2):129–35
Shintani S, Tsuruoka S, Shiigai T. Serial positron emission tomography (PET) in gliomatosis cerebri treated with radiotherapy: a case report. J Neurol Sci 2000;173(1):25–31
Herholz K, Kracht LW, Heiss WD. Monitoring the effect of chemotherapy in a mixed glioma by C-11-methionine PET. J Neuroimaging 2003;13(3):269–71
Herholz K, Holzer T, Bauer B, Schroder R, Voges J, Ernestus RI, et al. 11C-methionine PET for differential diagnosis of low-grade gliomas. Neurology 1998;50(5):1316–22
Tovi M, Lilja A, Bergstrom M, Ericsson A, Bergstrom K, Hartman M. Delineation of gliomas with magnetic resonance imaging using Gd-DTPA in comparison with computed tomography and positron emission tomography. Acta Radiol 1990;31(5):417–29
Kracht LW, Miletic H, Busch S, Jacobs AH, Voges J, Hoevels M, et al. Delineation of brain tumor extent with [11C]L-methionine positron emission tomography: local comparison with stereotactic histopathology. Clin Cancer Res 2004;10(21):7163–70
Pauleit D, Floeth F, Hamacher K, Riemenschneider MJ, Reifenberger G, Muller HW, et al. O-(2-[18F]fluoroethyl)-L-tyrosine PET combined with MRI improves the diagnostic assessment of cerebral gliomas. Brain 2005;128(Pt 3):678–87
Reinhardt MJ, Kubota K, Yamada S, Iwata R, Yaegashi H. Assessment of cancer recurrence in residual tumors after fractionated radiotherapy: a comparison of fluorodeoxyglucose, L-methionine and thymidine. J Nucl Med 1997;38(2):280–7
Sonoda Y, Kumabe T, Takahashi T, Shirane R, Yoshimoto T. Clinical usefulness of 11C-MET PET and 201T1 SPECT for differentiation of recurrent glioma from radiation necrosis. Neurol Med Chir (Tokyo) 1998;38(6):342–7; discussion 347–8
Kim do K, Kanai Y, Choi HW, Tangtrongsup S, Chairoungdua A, Babu E, Tachampa K, Anzai N, Iribe Y, Endou H. Characterization of the system L amino acid transporter in T24 human bladder carcinoma cells. Biochim Biophys Acta 2002;1565 (1):112–121
Jager PL, Vaalburg W, Pruim J, de Vries EG, Langen KJ, Piers DA. Radiolabeled amino acids: basic aspects and clinical applications in oncology. J Nucl Med 2001;42 (3):432–445
Bustany P, Chatel M, Derlon JM, Darcel F, Sgouropoulos P, Soussaline F, Syrota A. Brain tumor protein synthesis and histological grades: a study by positron emission tomography (PET) with C11–L–Methionine. J Neurooncol 1986;3 (4):397–404
Berger G, Maziere M, Knipper R, Prenant C, Comar D. Automated synthesis of 11C-labelled radiopharmaceuticals: imipramine, chlorpromazine, nicotine and methionine. Int J Appl Radiat Isot 1979;30 (7):393–399
Cizek J, Herholz K, Vollmar S, Schrader R, Klein J, Heiss WD. Fast and robust registration of PET and MR images of human brain. Neuroimage 2004;22 (1):434–442
Galldiks N, Burghaus L, Vollmar S, Cizek J, Impekoven P, Thomas A, Jacobs AH, Herholz K. Novel Neuroimaging Findings in a Patient with Cerebral Whipple’s Disease: A Magnetic Resonance Imaging and Positron Emission Tomography Study. J Neuroimaging 2004;14 (4):372–376
Rankin J. Cerebral vascular accidents in patients over the age of 60. II. Prognosis. Scott Med J 1957;2 (5):200–215
Bonita R, Beaglehole R. Recovery of motor function after stroke. Stroke 1988;19 (12):1497–1500
Macdonald DR, Cascino TL, Schold SC Jr, Cairncross JG. Response criteria for phase II studies of supratentorial malignant glioma. J Clin Oncol 1990;8 (7):1277–1280
Chang SM, Theodosopoulos P, Lamborn K, Malec M, Rabbitt J, Page M, Prados MD. Temozolomide in the treatment of recurrent malignant glioma. Cancer 2004;100 (3):605–611
Yung WK, Prados MD, Yaya–Tur R, Rosenfeld SS, Brada M, Friedman HS, Albright R, Olson J, Chang SM, O’Neill AM et al. Multicenter phase II trial of temozolomide in patients with anaplastic astrocytoma or anaplastic oligoastrocytoma at first relapse. Temodal Brain Tumor Group. J Clin Oncol 1999;17 (9):2762–2771
Albert FK, Forsting M, Sartor K, Adams HP, Kunze S. Early postoperative magnetic resonance imaging after resection of malignant glioma: objective evaluation of residual tumor and its influence on regrowth and prognosis. Neurosurgery 1994;34 (1):45–60
Roelcke U, Radu E, Ametamey S, Pellikka R, Steinbrich W, Leenders KL. Association of rubidium and C–methionine uptake in brain tumors measured by positron emission tomography. J Neurooncol 1996;27 (2):163–171
Tang BN, Sadeghi N, Branle F, De Witte O, Wikler D, Goldman S. Semi-quantification of methionine uptake and flair signal for the evaluation of chemotherapy in low-grade oligodendroglioma. J Neurooncol 2005;71 (2):161–168
Ribom D, Schoenmaekers M, Engler H, Smits A. Evaluation of 11C–methionine PET as a surrogate endpoint after treatment of grade 2 gliomas. J Neurooncol 2005;71 (3):325–332
Author information
Authors and Affiliations
Corresponding author
Additional information
N. Galldiks and L.W. Kracht contributed equally to this paper
Rights and permissions
About this article
Cite this article
Galldiks, N., Kracht, L.W., Burghaus, L. et al. Use of 11C-methionine PET to monitor the effects of temozolomide chemotherapy in malignant gliomas. Eur J Nucl Med Mol Imaging 33, 516–524 (2006). https://doi.org/10.1007/s00259-005-0002-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-005-0002-5