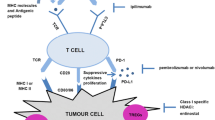
Abstract
Immunotherapy confers durable clinical benefit to melanoma, lung, and kidney cancer patients. Challengingly, most other solid tumors, including ovarian carcinoma, are not particularly responsive to immunotherapy, so combination with a complementary therapy may be beneficial. Recent findings suggest that epigenetic modifying drugs can prime antitumor immunity by increasing expression of tumor-associated antigens, chemokines, and activating ligands by cancer cells as well as cytokines by immune cells. This review, drawing from both preclinical and clinical data, describes some of the mechanisms of action that enable DNA methyltransferase inhibitors to facilitate the establishment of antitumor immunity.


Similar content being viewed by others
Abbreviations
- AML:
-
Acute myeloid leukemia
- AZA:
-
Azacitidine
- CLL:
-
Chronic lymphocytic leukemia
- CTA:
-
Cancer testis antigen
- CTL:
-
Cytotoxic T lymphocyte
- DAC:
-
Decitabine
- DNMT:
-
DNA methyltransferases
- DNMTi:
-
DNA methyltransferase inhibitor
- HDAC:
-
Histone deacetylase
- MAGE:
-
Melanoma-associated antigen
- MDS:
-
Myelodysplastic syndrome
- MDSC:
-
Myeloid-derived suppressor cell
- MHC I:
-
Major histocompatibility complex class I
- NK cells:
-
Natural killer cells
- NKG2D:
-
Natural-killer group 2, member D
- NSCLC:
-
Non-small cell lung cancer
- KIR:
-
Killer immunoglobulin-like receptor
- Tregs:
-
Regulatory T cells
References
Hanahan D, Weinberg R (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. doi:10.1016/j.cell.2011.02.013
Baylin S, Jones P (2011) A decade of exploring the cancer epigenome: biological and translational implications. Nat Rev Cancer 11:726–734. doi:10.1038/nrc3130
Esteller M (2008) Epigenetics in cancer. N Engl J Med 358:1148–1159. doi:10.1056/nejmra072067
Rodríguez-Paredes M, Esteller M (2011) Cancer epigenetics reaches mainstream oncology. Nat Med. doi:10.1038/nm.2305
Dawson M, Kouzarides T (2012) Cancer epigenetics: from mechanism to therapy. Cell 150:12–27. doi:10.1016/j.cell.2012.06.013
Minucci S, Pelicci P (2006) Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat Rev Cancer 6:38–51. doi:10.1038/nrc1779
West A, Johnstone R (2014) New and emerging HDAC inhibitors for cancer treatment. J Clin Invest 124:30–39. doi:10.1172/jci69738
Xu W, Parmigiani R, Marks P (2007) Histone deacetylase inhibitors: molecular mechanisms of action. Oncogene 26:5541–5552. doi:10.1038/sj.onc.1210620
Choudhary C, Kumar C, Gnad F, Nielsen M, Rehman M, Walther T, Olsen J, Mann M (2009) Lysine acetylation targets protein complexes and co-regulates major cellular functions. Science 325:834–840. doi:10.1126/science.1175371
Stresemann C, Lyko F (2008) Modes of action of the DNA methyltransferase inhibitors azacytidine and decitabine. Int J Cancer 123:8–13. doi:10.1002/ijc.23607
Derissen E, Beijnen J, Schellens J (2013) Concise drug review: azacitidine and decitabine. Oncologist 18:619–624. doi:10.1634/theoncologist.2012-0465
Bubeník J (2004) MHC class I down-regulation: tumour escape from immune surveillance? (Review). Int J Oncol 25:487–491. doi:10.3892/ijo.25.2.487
Tomasi T, Magner W, Khan A (2006) Epigenetic regulation of immune escape genes in cancer. Cancer Immunol Immun 55:1159–1184. doi:10.1007/s00262-006-0164-4
Maio M, Coral S, Fratta E, Altomonte M, Sigalotti L (2003) Epigenetic targets for immune intervention in human malignancies. Oncogene 22:6484–6488. doi:10.1038/sj.onc.1206956
Fratta E, Coral S, Covre A, Parisi G, Colizzi F, Danielli R, Marie Nicolay H, Sigalotti L, Maio M (2011) The biology of cancer testis antigens: putative function, regulation and therapeutic potential. Mol Oncol 5:164–182. doi:10.1016/j.molonc.2011.02.001
Dubovsky J, McNeel D, Powers J, Gordon J, Sotomayor E, Pinilla-Ibarz J (2009) Treatment of chronic lymphocytic leukemia with a hypomethylating agent induces expression of NXF2, an immunogenic cancer testis antigen. Clin Cancer Res 15:3406–3415. doi:10.1158/1078-0432.ccr-08-2099
Atanackovic D, Luetkens T, Kloth B, Fuchs G, Cao Y, Hildebrandt Y et al (2011) Cancer-testis antigen expression and its epigenetic modulation in acute myeloid leukemia. Am J Hematol 86:918–922. doi:10.1002/ajh.22141
Cruz C, Gerdemann U, Leen A, Shafer J, Ku S, Tzou B, Horton T et al (2011) Improving T-cell therapy for relapsed EBV-negative Hodgkin lymphoma by targeting upregulated MAGE-A4. Clin Cancer Res 17:7058–7066. doi:10.1158/1078-0432.ccr-11-1873
Fonsatti E, Nicolay H, Sigalotti L, Calabro L, Pezzani L, Colizzi F et al (2007) Functional up-regulation of human leukocyte antigen class I antigens expression by 5-aza-2′-deoxycytidine in cutaneous melanoma: immunotherapeutic implications. Clin Cancer Res 13:3333–3338. doi:10.1158/1078-0432.ccr-06-3091
Wang L, Amoozgar Z, Huang J, Saleh M, Xing D, Orsulic S, Goldberg M (2015) Decitabine enhances lymphocyte migration and function and synergizes with CTLA-4 blockade in a murine ovarian cancer model. Cancer Immunol Res 3:1030–1041. doi:10.1158/2326-6066.cir-15-0073
Adair S, Hogan K (2008) Treatment of ovarian cancer cell lines with 5-aza-2′-deoxycytidine upregulates the expression of cancer-testis antigens and class I major histocompatibility complex-encoded molecules. Cancer Immunol Immun 58:589–601. doi:10.1007/s00262-008-0582-6
Coral S, Sigalotti L, Colizzi F, Spessotto A, Nardi G, Cortini E et al (2006) Phenotypic and functional changes of human melanoma xenografts induced by DNA hypomethylation: immunotherapeutic implications. J Cell Physiol 207:58–66. doi:10.1002/jcp.20540
Sigalotti L, Fratta E, Coral S, Tanzarella S, Danielli R, Colizzi F et al (2004) Intratumor heterogeneity of cancer/testis antigens expression in human cutaneous melanoma is methylation-regulated and functionally reverted by 5-Aza-2′-deoxycytidine. Cancer Res 64:9167–9171. doi:10.1158/0008-5472.can-04-1442
Odunsi K, Matsuzaki J, James S, Mhawech-Fauceglia P, Tsuji T, Miller A et al (2014) Epigenetic potentiation of NY-ESO-1 vaccine therapy in human ovarian cancer. Cancer Immunol Res 2:37–49. doi:10.1158/2326-6066.cir-13-0126
Gunda V, Frederick D, Bernasconi M, Wargo J, Parangi S (2014) A potential role for immunotherapy in thyroid cancer by enhancing NY-ESO-1 cancer antigen expression. Thyroid 24:1241–1250. doi:10.1089/thy.2013.0680
Wachowska M, Gabrysiak M, Muchowicz A, Bednarek W, Barankiewicz J, Rygiel T et al (2014) 5-Aza-2′-deoxycytidine potentiates antitumour immune response induced by photodynamic therapy. Eur J Cancer 50:1370–1381. doi:10.1016/j.ejca.2014.01.017
Šímová J, Polláková V, Indrová M, Mikyšková R, Bieblová J, Štěpánek I et al (2011) Immunotherapy augments the effect of 5-azacytidine on HPV16-associated tumours with different MHC class I-expression status. Br J Cancer 105:1533–1541. doi:10.1038/bjc.2011.428
Krishnadas D, Bao L, Bai F, Chencheri S, Lucas K (2014) Decitabine facilitates immune recognition of sarcoma cells by upregulating CT antigens, MHC molecules, and ICAM-1. Tumor Biol 35:5753–5762. doi:10.1007/s13277-014-1764-9
Natsume A, Wakabayashi T, Tsujimura K, Shimato S, Ito M, Kuzushima K et al (2008) The DNA demethylating agent 5-aza-2′-deoxycytidine activates NY-ESO-1 antigenicity in orthotopic human glioma. Int J Cancer 122:2542–2553. doi:10.1002/ijc.23407
Coral S, Sigalotti L, Altomonte M, Engelsberg A, Cattarossi I, Maraskovsky E et al (2002) 5-aza-2′-Deoxycytidine-induced expression of functional cancer testis antigens in human renal cell carcinoma immunotherapeutic implications. Clin Cancer Res 8:2690
Guo Z (2006) De novo induction of a cancer/testis antigen by 5-aza-2′-deoxycytidine augments adoptive immunotherapy in a murine tumor model. Cancer Res 66:1105–1113. doi:10.1158/0008-5472.can-05-3020
Gunda V, Cogdill A, Bernasconi M, Wargo J, Parangi S (2013) Potential role of 5-Aza-2′-deoxycytidine induced MAGE-A4 expression in immunotherapy for anaplastic thyroid cancer. Surgery 154:1456–1462. doi:10.1016/j.surg.2013.07.009
Ayyoub M, Taub R, Keohan M, Hesdorffer M, Metthez G, Memeo L et al (2004) The frequent expression of cancer/testis antigens provides opportunities for immunotherapeutic targeting of sarcoma. Cancer Immun 4:7
Bao L, Dunham K, Lucas K (2011) MAGE-A1, MAGE-A3, and NY-ESO-1 can be upregulated on neuroblastoma cells to facilitate cytotoxic T lymphocyte-mediated tumor cell killing. Cancer Immunol Immun 60:1299–1307. doi:10.1007/s00262-011-1037-z
Sigalotti L, Coral S, Altomonte M, Natali L, Gaudino G, Cacciotti P et al (2002) Cancer testis antigens expression in mesothelioma: role of DNA methylation and bioimmunotherapeutic implications. Br J Cancer 86:979–982. doi:10.1038/sj.bjc.6600174
Schrump D, Fischette M, Nguyen D, Zhao M, Li X, Kunst T et al (2006) Phase I study of decitabine-mediated gene expression in patients with cancers involving the lungs, esophagus, or pleura. Clin Cancer Res 12:5777–5785. doi:10.1158/1078-0432.ccr-06-0669
Wu X, Tao Y, Hou J, Meng X, Shi J (2012) Valproic acid upregulates NKG2D ligand expression through an ERK-dependent mechanism and potentially enhances NK cell-mediated lysis of myeloma. Neoplasia 14:1178–1189
Marbach D, Costello J, Küffner R, Vega N, Prill R, Camacho D et al (2012) Wisdom of crowds for robust gene network inference. Nat Methods 9:796–804. doi:10.1038/nmeth.2016
Tang K, He C, Zeng G, Wu J, Song G, Shi Y et al (2008) Induction of MHC class I-related chain B (MICB) by 5-aza-2′-deoxycytidine. Biochem Biophys Res Commun 370:578–583. doi:10.1016/j.bbrc.2008.03.131
Chan H, Kurago Z, Stewart C, Wilson M, Martin M, Mace B et al (2003) DNA methylation maintains allele-specific KIR gene expression in human natural killer cells. J Exp Med 197:245–255. doi:10.1084/jem.20021127
Schmiedel B, Arélin V, Gruenebach F, Krusch M, Schmidt S, Salih H (2010) Azacytidine impairs NK cell reactivity while decitabine augments NK cell responsiveness toward stimulation. Int J Cancer 128:2911–2922. doi:10.1002/ijc.25635
Bruniquel D, Schwartz R (2003) Selective, stable demethylation of the interleukin-2 gene enhances transcription by an active process. Nat Immunol 4:235–240. doi:10.1038/ni887
Northrop J, Thomas R, Wells A, Shen H (2006) Epigenetic remodeling of the IL-2 and IFN-loci in memory CD8 T cells is influenced by CD4 T cells. J Immunol 177:1062–1069. doi:10.4049/jimmunol.177.2.1062
Berkley A, Hendricks D, Simmons K, Fink P (2013) Recent thymic emigrants and mature naive T cells exhibit differential DNA methylation at key cytokine loci. J Immunol 190:6180–6186. doi:10.4049/jimmunol.1300181
Fitzpatrick D, Shirley K, McDonald L, Bielefeldt-Ohmann H, Kay G, Kelso A (1998) Distinct methylation of the interferon (IFN-) and interleukin 3 (IL-3) genes in newly activated primary CD8+ T lymphocytes: regional IFN- promoter demethylation and mRNA expression are heritable in CD44highCD8+ T cells. J Exp Med 188:103–117. doi:10.1084/jem.188.1.103
Kersh E, Fitzpatrick D, Murali-Krishna K, Shires J, Speck S, Boss J, Ahmed R (2006) Rapid demethylation of the IFN-gamma gene occurs in memory but not naive CD8 T cells. J Immunol 176:4083–4093. doi:10.4049/jimmunol.176.7.4083
Janson P, Linton L, Ahlen Bergman E, Marits P, Eberhardson M, Piehl F et al (2010) Profiling of CD4+ T cells with epigenetic immune lineage analysis. J Immunol 186:92–102. doi:10.4049/jimmunol.1000960
Costantini B, Kordasti S, Kulasekararaj A, Jiang J, Seidl T, Abellan P et al (2012) The effects of 5-azacytidine on the function and number of regulatory T cells and T-effectors in myelodysplastic syndrome. Haematologica 98:1196–1205. doi:10.3324/haematol.2012.074823
Frikeche J, Clavert A, Delaunay J, Brissot E, Grégoire M, Gaugler B, Mohty M (2011) Impact of the hypomethylating agent 5-azacytidine on dendritic cells function. Exp Hematol 39:1056–1063. doi:10.1016/j.exphem.2011.08.004
Dubovsky J, Powers J, Gao Y, Mariusso L, Sotomayor E, Pinilla-Ibarz J (2011) Epigenetic repolarization of T lymphocytes from chronic lymphocytic leukemia patients using 5-aza-2′-deoxycytidine. Leukemia Res 35:1193–1199. doi:10.1016/j.leukres.2011.02.007
Sinha P, Clements V, Bunt S, Albelda S, Ostrand-Rosenberg S (2007) Cross-talk between myeloid-derived suppressor cells and macrophages subverts tumor immunity toward a Type 2 response. J Immunol 179:977–983. doi:10.4049/jimmunol.179.2.977
Guislain A, Gadiot J, Kaiser A, Jordanova E, Broeks A, Sanders J et al (2015) Sunitinib pretreatment improves tumor-infiltrating lymphocyte expansion by reduction in intratumoral content of myeloid-derived suppressor cells in human renal cell carcinoma. Cancer Immunol Immun 64:1241–1250. doi:10.1007/s00262-015-1735-z
Kim K, Skora A, Li Z, Liu Q, Tam A, Blosser R et al (2014) Eradication of metastatic mouse cancers resistant to immune checkpoint blockade by suppression of myeloid-derived cells. Proc Natl Acad Sci USA 111:11774–11779. doi:10.1073/pnas.1410626111
Triozzi P, Aldrich W, Achberger S, Ponnazhagan S, Alcazar O, Saunthararajah Y (2012) Differential effects of low-dose decitabine on immune effector and suppressor responses in melanoma-bearing mice. Cancer Immunol Immun 61:1441–1450. doi:10.1007/s00262-012-1204-x
Mikysková R, Indrova M, Vlkova V, Bieblova J, Šimova J, Paračková Z et al (2014) DNA demethylating agent 5-azacytidine inhibits myeloid-derived suppressor cells induced by tumor growth and cyclophosphamide treatment. J Leukoc Biol 95:743–753. doi:10.1189/jlb.0813435
Daurkin I, Eruslanov E, Vieweg J, Kusmartsev S (2009) Generation of antigen-presenting cells from tumor-infiltrated CD11b myeloid cells with DNA demethylating agent 5-aza-2′-deoxycytidine. Cancer Immunol Immun 59:697–706. doi:10.1007/s00262-009-0786-4
Tsai H, Li H, Van Neste L, Cai Y, Robert C, Rassool F et al (2012) Transient low doses of DNA-demethylating agents exert durable antitumor effects on hematological and epithelial tumor cells. Cancer Cell 21:430–446. doi:10.1016/j.ccr.2011.12.029
Wrangle J, Wang W, Koch A, Easwaran H, Mohammad H, Vendetti F et al (2013) Alterations of immune response of non-small cell lung cancer with azacytidine. Oncotarget 4:2067–2079
Li H, Chiappinelli K, Guzzetta A, Easwaran H, Yen R, Vatapalli R et al (2014) Immune regulation by low doses of the DNA methyltransferase inhibitor 5-azacitidine in common human epithelial cancers. Oncotarget 5:587–598
Yang H, Bueso-Ramos C, DiNardo C, Estecio M, Davanlou M, Geng Q et al (2013) Expression of PD-L1, PD-L2, PD-1 and CTLA4 in myelodysplastic syndromes is enhanced by treatment with hypomethylating agents. Leukemia 28:1280–1288. doi:10.1038/leu.2013.355
Pico de Coaña Y, Choudhury A, Kiessling R (2015) Checkpoint blockade for cancer therapy: revitalizing a suppressed immune system. Trends Mol Med 21:482–491. doi:10.1016/j.molmed.2015.05.005
Paradis T, Floyd E, Burkwit J, Cole S, Brunson B, Elliott E, Gilman S, Gladue R (2001) The anti-tumor activity of anti-CTLA-4 is mediated through its induction of IFN gamma. Cancer Immunol Immunother 50:125–133
Peng W, Liu C, Xu C, Lou Y, Chen J, Yang Y et al (2012) PD-1 blockade enhances T-cell migration to tumors by elevating IFN- inducible chemokines. Cancer Res 72:5209–5218. doi:10.1158/0008-5472.can-12-1187
Fan H, Lu X, Wang X, Liu Y, Guo B, Zhang Y et al (2014) Low-dose decitabine-based chemoimmunotherapy for patients with refractory advanced solid tumors: a phase I/II report. J Immunol Res 2014:371087. doi:10.1155/2014/371087
Krishnadas D, Shusterman S, Bai F, Diller L, Sullivan J, Cheerva A et al (2015) A phase I trial combining decitabine/dendritic cell vaccine targeting MAGE-A1, MAGE-A3 and NY-ESO-1 for children with relapsed or therapy-refractory neuroblastoma and sarcoma. Cancer Immunol Immun 64:1251–1260. doi:10.1007/s00262-015-1731-3
Acknowledgments
We thank the Ovarian Cancer Research Fund (Liz Tilberis Scholar award) and the Susan F. Smith Center for Women’s Cancer for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
This paper is a Focussed Research Review based on a presentation given at the Fourth International Conference on Cancer Immunotherapy and Immunomonitoring (CITIM 2015), held in Ljubljana, Slovenia, 27th–30th April 2015. It is part of a series of Focussed Research Reviews and meeting report in Cancer Immunology, Immunotherapy.
Rights and permissions
About this article
Cite this article
Saleh, M.H., Wang, L. & Goldberg, M.S. Improving cancer immunotherapy with DNA methyltransferase inhibitors. Cancer Immunol Immunother 65, 787–796 (2016). https://doi.org/10.1007/s00262-015-1776-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-015-1776-3