Abstract
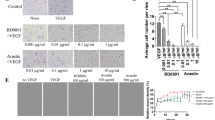
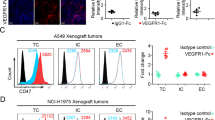
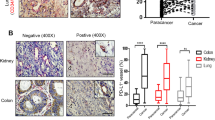
The effectual clinical benefits of immune checkpoint inhibitor (ICI) are hampered by a high rate of innate resistance, and VEGFA may contribute to ICI treatment resistance. In this study, we endeavored to assess the tumor microenvironment (TME) in VEGFA-overexpressed human tumors and mouse tumor models, and to explore whether anti-angiogenesis therapy can overcome the innate resistance to ICI in hyperangiogenesis mouse tumor models and the underlying mechanism. Effect of VEGFA on clinical prognosis and TME was analyzed using TCGA data. The VEGFA-overexpressed mouse breast and colon subcutaneous models were established. PD-1 mAb or apatinib alone and combination therapy were used. Immunohistochemistry and immunofluorescence were used to assess angiogenesis and hypoxia. Flow cytometry, RNA sequencing and MCP-counter were applied to detect tumor immunomicroenvironment. High level of VEGFA mRNA in human tumors is related to poor prognosis and hypoxic, angiogenic and immunosuppressive TME. Upregulation of VEGFA increased the degree of malignancy of tumor cells in vitro and in vivo. VEGFA-overexpressed models were characterized by hypoxic, hyperangiogenic and immunosuppressive TME and indicated innate resistance to ICI. In tumor-bearing mice without VEGFA overexpression, the combination therapy had no synergistic anti-tumor effect compared to monotherapy. However, apatinib alleviated hyperangiogenesis and hypoxia in TME and converted the immunosuppressive TME into an immunostimulatory one in VEGFA-overexpressed tumors. Thus, anti-angiogenesis therapy could improve the efficiency of ICI in VEGFA-overexpressed tumors. Revealing whether there is hypervascularization in tumor tissues may help to clarify the adoption of anti-angiogenesis and ICI combination therapy or ICI monotherapy in cancer treatment.







Similar content being viewed by others
Abbreviations
- AA:
-
Anti-angiogenesis
- GSEA:
-
Gene set enrichment analysis
- ICI:
-
Immune checkpoint inhibitor
- ICER:
-
Incremental cost-effectiveness ratio
- IPRES:
-
Innate anti-PD-1 resistance
- KI:
-
Tyrosine kinase inhibitor
- LAG-3:
-
Lymphocyte activation gene-3
- MDSCs:
-
Myeloid-derived suppressor cells
- MVD:
-
Microvascular density
- ORR:
-
Objective response rates
- OS:
-
Overall survival
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death ligand 1
- QALY:
-
Quality-adjusted life-year
- RCC:
-
Renal cell carcinoma
- TCGA:
-
The Cancer Genome Atlas database
- TIGIT:
-
T cell immunoreceptor with Ig and ITIM domain
- TILs:
-
Tumor-infiltrating lymphocytes
- TIM-3:
-
T cell immunoglobulin and mucin-domain containing-3
- TME:
-
Tumor microenvironment
- TOX:
-
Thymocyte selection-associated HMG box protein
- Tregs:
-
T regulatory cells
- TRAE:
-
Treatment-related adverse effect
- UCSC:
-
University of California Santa Cruz database
- VEGFA:
-
Vascular endothelial growth factor A
References
Zou W, Wolchok JD, Chen L (2016) PD-L1 (B7–H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Science translational medicine 8(328):328rv324. https://doi.org/10.1126/scitranslmed.aad7118
Abril-Rodriguez G, Ribas A (2017) SnapShot: immune checkpoint inhibitors. Cancer Cell 31(6):848–848.e841. https://doi.org/10.1016/j.ccell.2017.05.010
Sharma P, Allison JP (2015) The future of immune checkpoint therapy. Science 348(6230):56–61. https://doi.org/10.1126/science.aaa8172
Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB et al (2010) Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med 363(8):711–723. https://doi.org/10.1056/NEJMoa1003466
Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF et al (2012) Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 366(26):2443–2454. https://doi.org/10.1056/NEJMoa1200690
Hamid O, Robert C, Daud A, Hodi FS, Hwu WJ, Kefford R et al (2013) Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med 369(2):134–144. https://doi.org/10.1056/NEJMoa1305133
Wang Q, Wu X (2017) Primary and acquired resistance to PD-1/PD-L1 blockade in cancer treatment. Int Immunopharmacol 46:210–219. https://doi.org/10.1016/j.intimp.2017.03.015
Hugo W, Zaretsky JM, Sun L, Song C, Moreno BH, Hu-Lieskovan S et al (2016) Genomic and transcriptomic features of response to Anti-PD-1 therapy in metastatic melanoma. Cell 165(1):35–44. https://doi.org/10.1016/j.cell.2016.02.065
Huang Y, Goel S, Duda DG, Fukumura D, Jain RK (2013) Vascular normalization as an emerging strategy to enhance cancer immunotherapy. Cancer Res 73(10):2943–2948. https://doi.org/10.1158/0008-5472.can-12-4354
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65(1–2):271–284
Viallard C, Larrivee B (2017) Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis 20(4):409–426. https://doi.org/10.1007/s10456-017-9562-9
Jain RK (2014) Antiangiogenesis strategies revisited: from starving tumors to alleviating hypoxia. Cancer Cell 26(5):605–622. https://doi.org/10.1016/j.ccell.2014.10.006
Colegio OR, Chu NQ, Szabo AL, Chu T, Rhebergen AM, Jairam V et al (2014) Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513(7519):559–563. https://doi.org/10.1038/nature13490
Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P et al (2004) Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med 10(9):942–949. https://doi.org/10.1038/nm1093
Noy R, Pollard JW (2014) Tumor-associated macrophages: from mechanisms to therapy. Immunity 41(1):49–61. https://doi.org/10.1016/j.immuni.2014.06.010
Facciabene A, Peng X, Hagemann IS, Balint K, Barchetti A, Wang LP et al (2011) Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and T(reg) cells. Nature 475(7355):226–230. https://doi.org/10.1038/nature10169
Gabrilovich D, Ishida T, Oyama T, Ran S, Kravtsov V, Nadaf S et al (1998) Vascular endothelial growth factor inhibits the development of dendritic cells and dramatically affects the differentiation of multiple hematopoietic lineages in vivo. Blood 92(11):4150–4166
Terme M, Pernot S, Marcheteau E, Sandoval F, Benhamouda N, Colussi O et al (2013) VEGFA-VEGFR pathway blockade inhibits tumor-induced regulatory T-cell proliferation in colorectal cancer. Can Res 73(2):539–549. https://doi.org/10.1158/0008-5472.can-12-2325
Palazón A, Aragonés J, Morales-Kastresana A, de Landázuri MO, Melero I (2012) Molecular pathways: hypoxia response in immune cells fighting or promoting cancer. Clin Cancer Res 18(5):1207–1213. https://doi.org/10.1158/1078-0432.ccr-11-1591
Ohm JE, Gabrilovich DI, Sempowski GD, Kisseleva E, Parman KS, Nadaf S et al (2003) VEGF inhibits T-cell development and may contribute to tumor-induced immune suppression. Blood 101(12):4878–4886. https://doi.org/10.1182/blood-2002-07-1956
Chen PL, Roh W, Reuben A, Cooper ZA, Spencer CN, Prieto PA et al (2016) Analysis of immune signatures in longitudinal tumor samples yields insight into biomarkers of response and mechanisms of resistance to immune checkpoint blockade. Cancer Discov 6(8):827–837. https://doi.org/10.1158/2159-8290.cd-15-1545
McDermott DF, Huseni MA, Atkins MB, Motzer RJ, Rini BI, Escudier B et al (2018) Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat Med 24(6):749–757. https://doi.org/10.1038/s41591-018-0053-3
Molife C, Hess LM, Cui ZL, Li XI, Beyrer J, Mahoui M et al (2019) Sequential therapy with ramucirumab and/or checkpoint inhibitors for non-small-cell lung cancer in routine practice. Future Oncol 15(25):2915–2931. https://doi.org/10.2217/fon-2018-0876
Motzer RJ, Penkov K, Haanen J, Rini B, Albiges L, Campbell MT et al (2019) Avelumab plus Axitinib versus Sunitinib for Advanced Renal-Cell Carcinoma. N Engl J Med 380(12):1103–1115. https://doi.org/10.1056/NEJMoa1816047
Rini BI, Powles T, Atkins MB, Escudier B, McDermott DF, Suarez C et al (2019) Atezolizumab plus bevacizumab versus sunitinib in patients with previously untreated metastatic renal cell carcinoma (IMmotion151): a multicentre, open-label, phase 3, randomised controlled trial. Lancet 393(10189):2404–2415. https://doi.org/10.1016/s0140-6736(19)30723-8
Amin A, Plimack ER, Ernstoff MS, Lewis LD, Bauer TM, McDermott DF et al (2018) Safety and efficacy of nivolumab in combination with sunitinib or pazopanib in advanced or metastatic renal cell carcinoma: the CheckMate 016 study. J Immunother Cancer 6(1):109. https://doi.org/10.1186/s40425-018-0420-0
Makker V, Rasco D, Vogelzang NJ, Brose MS, Cohn AL, Mier J et al (2019) Lenvatinib plus pembrolizumab in patients with advanced endometrial cancer: an interim analysis of a multicentre, open-label, single-arm, phase 2 trial. Lancet Oncol 20(5):711–718. https://doi.org/10.1016/s1470-2045(19)30020-8
Arkenau HT, Martin-Liberal J, Calvo E, Penel N, Krebs MG, Herbst RS et al (2018) Ramucirumab plus pembrolizumab in patients with previously treated advanced or metastatic biliary tract cancer: nonrandomized, open-label, phase I trial (JVDF). Oncologist 23(12):1407–e1136. https://doi.org/10.1634/theoncologist.2018-0044
Li J, Zhao X, Chen L, Guo H, Lv F, Jia K et al (2010) Safety and pharmacokinetics of novel selective vascular endothelial growth factor receptor-2 inhibitor YN968D1 in patients with advanced malignancies. BMC Cancer 10:529. https://doi.org/10.1186/1471-2407-10-529
Fang S, Zhang M, Wei G, Lu KH (2018) Apatinib as a third- or further- line treatment in patients with advanced NSCLC harboring wild-type EGFR. Oncotarget 9(6):7175–7181. https://doi.org/10.18632/oncotarget.23612
Hu X, Zhang J, Xu B, Jiang Z, Ragaz J, Tong Z et al (2014) Multicenter phase II study of apatinib, a novel VEGFR inhibitor in heavily pretreated patients with metastatic triple-negative breast cancer. Int J Cancer 135(8):1961–1969. https://doi.org/10.1002/ijc.28829
Kong Y, Sun L, Hou Z, Zhang Y, Chen P, Cui Y et al (2017) Apatinib is effective for treatment of advanced hepatocellular carcinoma. Oncotarget 8(62):105596–105605. https://doi.org/10.18632/oncotarget.22337
Li F, Liao Z, Zhang C, Zhao J, Xing R, Teng S et al (2018) Apatinib as targeted therapy for sarcoma. Oncotarget 9(36):24548–24560. https://doi.org/10.18632/oncotarget.24647
Lin Y, Wang C, Gao W, Cui R, Liang J (2017) Overwhelming rapid metabolic and structural response to apatinib in radioiodine refractory differentiated thyroid cancer. Oncotarget 8(26):42252–42261. https://doi.org/10.18632/oncotarget.15036
Miao M, Deng G, Luo S, Zhou J, Chen L, Yang J et al (2018) A phase II study of apatinib in patients with recurrent epithelial ovarian cancer. Gynecol Oncol 148(2):286–290
Bindea G, Mlecnik B, Tosolini M, Kirilovsky A, Waldner M, Obenauf AC et al (2013) Spatiotemporal dynamics of intratumoral immune cells reveal the immune landscape in human cancer. Immunity 39(4):782–795. https://doi.org/10.1016/j.immuni.2013.10.003
Becht E, Giraldo NA, Lacroix L, Buttard B, Elarouci N, Petitprez F et al (2016) Estimating the population abundance of tissue-infiltrating immune and stromal cell populations using gene expression. Genome Biol 17(1):218. https://doi.org/10.1186/s13059-016-1070-5
Aslakson CJ, Miller FR (1992) Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Can Res 52(6):1399–1405
Corbett TH, Griswold DP Jr, Roberts BJ, Peckham JC, Schabel FM Jr (1975) Tumor induction relationships in development of transplantable cancers of the colon in mice for chemotherapy assays, with a note on carcinogen structure. Can Res 35(9):2434–2439
Alfei F, Kanev K, Hofmann M, Wu M, Ghoneim HE, Roelli P et al (2019) TOX reinforces the phenotype and longevity of exhausted T cells in chronic viral infection. Nature 571(7764):265–269. https://doi.org/10.1038/s41586-019-1326-9
Khan O, Giles JR, McDonald S, Manne S, Ngiow SF, Patel KP et al (2019) TOX transcriptionally and epigenetically programs CD8(+) T cell exhaustion. Nature 571(7764):211–218. https://doi.org/10.1038/s41586-019-1325-x
Kim CG, Jang M, Kim Y, Leem G, Kim KH, Lee H et al (2019) VEGF-A drives TOX-dependent T cell exhaustion in anti-PD-1-resistant microsatellite stable colorectal cancers. Sci Immunol. https://doi.org/10.1126/sciimmunol.aay0555
Wherry EJ (2011) T cell exhaustion. Nat Immunol 12(6):492–499. https://doi.org/10.1038/ni.2035
Petitprez F, Vano YA, Becht E, Giraldo NA, de Reynies A, Sautes-Fridman C et al (2018) Transcriptomic analysis of the tumor microenvironment to guide prognosis and immunotherapies. Cancer Immunol Immunother C: II 67(6):981–988. https://doi.org/10.1007/s00262-017-2058-z
Guo LY, Zhu P, Jin XP (2016) Association between the expression of HIF-1alpha and VEGF and prognostic implications in primary liver cancer. Genet Mol Res. https://doi.org/10.4238/gmr.15028107
Jantus-Lewintre E, Sanmartin E, Sirera R, Blasco A, Sanchez JJ, Taron M et al (2011) Combined VEGF-A and VEGFR-2 concentrations in plasma: diagnostic and prognostic implications in patients with advanced NSCLC. Lung Cancer 74(2):326–331. https://doi.org/10.1016/j.lungcan.2011.02.016
Wang J, Taylor A, Showeil R, Trivedi P, Horimoto Y, Bagwan I et al (2014) Expression profiling and significance of VEGF-A, VEGFR2, VEGFR3 and related proteins in endometrial carcinoma. Cytokine 68(2):94–100. https://doi.org/10.1016/j.cyto.2014.04.005
Wang X, Chen X, Fang J, Yang C (2013) Overexpression of both VEGF-A and VEGF-C in gastric cancer correlates with prognosis, and silencing of both is effective to inhibit cancer growth. Int J Clin Exp Pathol 6(4):586–597
Yamamoto S, Konishi I, Mandai M, Kuroda H, Komatsu T, Nanbu K et al (1997) Expression of vascular endothelial growth factor (VEGF) in epithelial ovarian neoplasms: correlation with clinicopathology and patient survival, and analysis of serum VEGF levels. Br J Cancer 76(9):1221–1227
Xiao K, Ouyang Z, Tang HH (2018) Inhibiting the proliferation and metastasis of hilar cholangiocarcinoma cells by blocking the expression of vascular endothelial growth factor with small interfering RNA. Oncol Lett 16(2):1841–1848. https://doi.org/10.3892/ol.2018.8840
Zeng FC, Zeng MQ, Huang L, Li YL, Gao BM, Chen JJ et al (2016) Downregulation of VEGFA inhibits proliferation, promotes apoptosis, and suppresses migration and invasion of renal clear cell carcinoma. Onco Targets Ther 9:2131–2141. https://doi.org/10.2147/ott.s98002
Bergers G, Hanahan D (2008) Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer 8(8):592–603. https://doi.org/10.1038/nrc2442
Movahedi K, Laoui D, Gysemans C, Baeten M, Stange G, Van den Bossche J et al (2010) Different tumor microenvironments contain functionally distinct subsets of macrophages derived from Ly6C(high) monocytes. Can Res 70(14):5728–5739. https://doi.org/10.1158/0008-5472.can-09-4672
Cooke VG, LeBleu VS, Keskin D, Khan Z, O'Connell JT, Teng Y et al (2012) Pericyte depletion results in hypoxia-associated epithelial-to-mesenchymal transition and metastasis mediated by met signaling pathway. Cancer Cell 21(1):66–81. https://doi.org/10.1016/j.ccr.2011.11.024
Jain RK (2005) Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307(5706):58–62. https://doi.org/10.1126/science.1104819
Meng MB, Zaorsky NG, Deng L, Wang HH, Chao J, Zhao LJ et al (2015) Pericytes: a double-edged sword in cancer therapy. Future Oncol 11(1):169–179. https://doi.org/10.2217/fon.14.123
Batchelor TT, Gerstner ER, Emblem KE, Duda DG, Kalpathy-Cramer J, Snuderl M et al (2013) Improved tumor oxygenation and survival in glioblastoma patients who show increased blood perfusion after cediranib and chemoradiation. Proc Natl Acad Sci USA 110(47):19059–19064. https://doi.org/10.1073/pnas.1318022110
Heist RS, Duda DG, Sahani DV, Ancukiewicz M, Fidias P, Sequist LV et al (2015) Improved tumor vascularization after anti-VEGF therapy with carboplatin and nab-paclitaxel associates with survival in lung cancer. Proc Natl Acad Sci USA 112(5):1547–1552. https://doi.org/10.1073/pnas.1424024112
Tolaney SM, Boucher Y, Duda DG, Martin JD, Seano G, Ancukiewicz M et al (2015) Role of vascular density and normalization in response to neoadjuvant bevacizumab and chemotherapy in breast cancer patients. Proc Natl Acad Sci USA 112(46):14325–14330. https://doi.org/10.1073/pnas.1518808112
Huang Y, Yuan J, Righi E, Kamoun WS, Ancukiewicz M, Nezivar J et al (2012) Vascular normalizing doses of antiangiogenic treatment reprogram the immunosuppressive tumor microenvironment and enhance immunotherapy. Proc Natl Acad Sci USA 109(43):17561–17566. https://doi.org/10.1073/pnas.1215397109
Herbst RS, Arkenau HT, Santana-Davila R, Calvo E, Paz-Ares L, Cassier PA et al (2019) Ramucirumab plus pembrolizumab in patients with previously treated advanced non-small-cell lung cancer, gastro-oesophageal cancer, or urothelial carcinomas (JVDF): a multicohort, non-randomised, open-label, phase 1a/b trial. Lancet Oncol 20(8):1109–1123. https://doi.org/10.1016/s1470-2045(19)30458-9
Chung AS, Kowanetz M, Wu X, Zhuang G, Ngu H, Finkle D et al (2012) Differential drug class-specific metastatic effects following treatment with a panel of angiogenesis inhibitors. J Pathol 227(4):404–416. https://doi.org/10.1002/path.4052
Wu S, Zhou J, Guo J, Hua Z, Li J, Wang Z (2019) Apatinib inhibits tumor growth and angiogenesis in PNET models. Endocr Connect 8(1):8–19. https://doi.org/10.1530/ec-18-0397
Tian S, Quan H, Xie C, Guo H, Lu F, Xu Y et al (2011) YN968D1 is a novel and selective inhibitor of vascular endothelial growth factor receptor-2 tyrosine kinase with potent activity in vitro and in vivo. Cancer Sci 102(7):1374–1380. https://doi.org/10.1111/j.1349-7006.2011.01939.x
Peng QX, Han YW, Zhang YL, Hu J, Fan J, Fu SZ et al (2017) Apatinib inhibits VEGFR-2 and angiogenesis in an in vivo murine model of nasopharyngeal carcinoma. Oncotarget 8(32):52813–52822. https://doi.org/10.18632/oncotarget.17264
Zhou K, Zhang JW, Wang QZ, Liu WY, Liu JL, Yao L et al (2019) Apatinib, a selective VEGFR2 inhibitor, improves the delivery of chemotherapeutic agents to tumors by normalizing tumor vessels in LoVo colon cancer xenograft mice. Acta Pharmacol Sin 40(4):556–562. https://doi.org/10.1038/s41401-018-0058-y
Liu ZJ, Zhou YJ, Ding RL, Xie F, Fu SZ, Wu JB et al (2018) In vitro and in vivo apatinib inhibits vasculogenic mimicry in melanoma MUM-2B cells. PLoS ONE 13(7):e0200845. https://doi.org/10.1371/journal.pone.0200845
Li B, Lalani AS, Harding TC, Luan B, Koprivnikar K, Huan TuG et al (2006) Vascular endothelial growth factor blockade reduces intratumoral regulatory T cells and enhances the efficacy of a GM-CSF-secreting cancer immunotherapy. Clin Cancer Res Off J Am Assoc Cancer Res 12(22):6808–6816. https://doi.org/10.1158/1078-0432.ccr-06-1558
Huang KW, Wu HL, Lin HL, Liang PC, Chen PJ, Chen SH et al (2010) Combining antiangiogenic therapy with immunotherapy exerts better therapeutical effects on large tumors in a woodchuck hepatoma model. Proc Natl Acad Sci USA 107(33):14769–14774. https://doi.org/10.1073/pnas.1009534107
Reck M, Mok TSK, Nishio M, Jotte RM, Cappuzzo F, Orlandi F et al (2019) Atezolizumab plus bevacizumab and chemotherapy in non-small-cell lung cancer (IMpower150): key subgroup analyses of patients with EGFR mutations or baseline liver metastases in a randomised, open-label phase 3 trial. Lancet Respir Med 7(5):387–401. https://doi.org/10.1016/s2213-2600(19)30084-0
Xu J, Zhang Y, Jia R, Yue C, Chang L, Liu R et al (2019) Anti-PD-1 antibody SHR-1210 combined with apatinib for advanced hepatocellular carcinoma, gastric, or esophagogastric junction cancer: an open-label, dose escalation and expansion study. Clin Cancer Res Off J Am Assoc Cancer Res 25(2):515–523. https://doi.org/10.1158/1078-0432.ccr-18-2484
Wan X, Luo X, Tan C, Zeng X, Zhang Y, Peng L (2019) First-line atezolizumab in addition to bevacizumab plus chemotherapy for metastatic, nonsquamous non-small cell lung cancer: a United States-based cost-effectiveness analysis. Cancer. https://doi.org/10.1002/cncr.32368
Choueiri TK, Larkin J, Oya M, Thistlethwaite F, Martignoni M, Nathan P et al (2018) Preliminary results for avelumab plus axitinib as first-line therapy in patients with advanced clear-cell renal-cell carcinoma (JAVELIN Renal 100): an open-label, dose-finding and dose-expansion, phase 1b trial. Lancet Oncol 19(4):451–460. https://doi.org/10.1016/s1470-2045(18)30107-4
Chen J, Hu G, Chen Z, Wan X, Tan C, Zeng X et al (2019) Cost-effectiveness analysis of pembrolizumab plus axitinib versus sunitinib in first-line advanced renal cell carcinoma in China. Clin Drug Investig. https://doi.org/10.1007/s40261-019-00820-6
Acknowledgements
We gratefully thank the Natural Science Foundation of China and School of Medicine, Shanghai Jiaotong University, for the funding.
Funding
This work is supported by grants from National Natural Science Foundation of China (81472843 to W. X.) and the study program of clinical capacity in urgently needed postgraduate majors in the School of Medicine, Shanghai Jiaotong University (JQ201703).
Author information
Authors and Affiliations
Contributions
XW and WD designed and supervised the study. QHW performed the experiments in vitro. QHW and JZG established the animal model and conducted the animal study. JZG was responsible for the TCGA and mRNA sequencing analysis. QHW and XW completed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
We state that animal experiment in our study was approved by the Institutional Animal Care and Use Committee of Shanghai Jiao Tong University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, Q., Gao, J., Di, W. et al. Anti-angiogenesis therapy overcomes the innate resistance to PD-1/PD-L1 blockade in VEGFA-overexpressed mouse tumor models. Cancer Immunol Immunother 69, 1781–1799 (2020). https://doi.org/10.1007/s00262-020-02576-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02576-x