Abstract
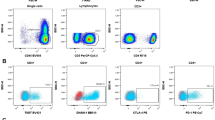
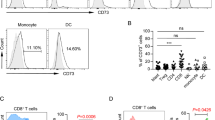
Malignant cells can increase in number using immune escape mechanisms such as immune checkpoints. In this study, we evaluated the expression of an immune checkpoint programmed death 1 (PD-1) on T-cell subsets in chronic myeloid leukemia (CML). We obtained bone marrow aspirate samples from CML patients and from individuals without evidence of hematologic malignancies (controls). PD-1 expression on T-cell subsets was measured using flow cytometric analysis. PD-1 expression levels on CD8+ T-cells were significantly lower in complete hematologic response (CHR) than in controls, chronic phase, and blast phase (BP). In CML patients receiving imatinib and dasatinib, PD-1 expression levels on CD8+ T-cells were lower than that at diagnosis. PD-1 expression levels on CD8+ T-cells were positively correlated with quantitative levels of the BCR/ABL fusion gene. PD-1 expression levels on CD4+ T-cells were higher in BP than in CHR. PD-1 expression levels on CD4+ T-cells did not differ significantly according to different medications or quantitative BCR/ABL1 fusion gene levels. Low PD-1 expression on CD8+ T-cells might play a role in maintaining CHR in CML patients. Immune monitoring of PD-1 expression on CD8+ T-cells may predict the disease course. In cases of refractory disease or resistance to imatinib or dasatinib, the use of PD-1 inhibitors would be helpful.







Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AP:
-
Accelerated phase
- BP:
-
Blastic phase
- CHR:
-
Complete hematologic response
- CP:
-
Chronic phase
References
Fife BT, Bluestone JA (2008) Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol Rev 224:166–182
Butte MJ, Keir ME, Phamduy TB, Sharpe AH, Freeman GJ (2007) Programmed death-1 ligand 1 interacts specifically with the B7–1 costimulatory molecule to inhibit T cell responses. Immunity 27:111–122
Francisco LM, Sage PT, Sharpe AH (2010) The PD-1 pathway in tolerance and autoimmunity. Immunol Rev 236:219–242
Keir ME, Butte MJ, Freeman GJ, Sharpe AH (2008) PD-1 and its ligands in tolerance and immunity. Annu Rev Immunol 26:677–704
Patel SP, Kurzrock R (2015) PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 14:847–856
Hughes A, Yong AS (2017) Immune effector recovery in chronic myeloid leukemia and treatment-free remission. Front Immunol 8:469
Christiansson L, Söderlund S, Svensson E, Mustjoki S, Bengtsson M, Simonsson B, Olsson-Strömberg U, Loskog AS (2013) Increased level of myeloid-derived suppressor cells, programmed death receptor ligand 1/programmed death receptor 1, and soluble CD25 in Sokal high risk chronic myeloid leukemia. PLoS ONE 8:e55818
Hughes A, Clarson J, Tang C, Vidovic L, White DL, Hughes TP, Yong AS (2017) CML patients with deep molecular responses to TKI have restored immune effectors, decreased PD-1 and immune suppressors. Blood:blood-2016–2010–745992
Pallera A, Altman JK, Berman E, Abboud CN, Bhatnagar B, Curtin P, DeAngelo DJ, Gotlib J, Hagelstrom RT, Hobbs G (2016) NCCN guidelines insights: chronic myeloid leukemia, version 1.2017. J Natl Compr Cancer Netw 14:1505–1512
WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (2017). Media centre—IARC news
Vonka V, Petráčková M (2015) Immunology of chronic myeloid leukemia: current concepts and future goals. Expert Rev Clin Immunol 11:511–522
Yong AS, Keyvanfar K, Eniafe R, Savani BN, Rezvani K, Sloand EM, Goldman JM, Barrett AJ (2008) Hematopoietic stem cells and progenitors of chronic myeloid leukemia express leukemia-associated antigens: implications for the graft-versus-leukemia effect and peptide vaccine-based immunotherapy. Leukemia 22:1721
Baccarani M, Pileri S, Steegmann J-L, Muller M, Soverini S, Dreyling M, Group EGW (2012) Chronic myeloid leukemia: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 23:vii72–vii77
Mumprecht S, Schürch C, Schwaller J, Solenthaler M, Ochsenbein AF (2009) Programmed death 1 signaling on chronic myeloid leukemia–specific T cells results in T-cell exhaustion and disease progression. Blood 114:1528–1536
Kreutzman A, Porkka K, Mustjoki S (2013) Immunomodulatory effects of tyrosine kinase inhibitors. Int Trends Immun 1:17–28
Balachandran VP, Cavnar MJ, Zeng S, Bamboat ZM, Ocuin LM, Obaid H, Sorenson EC, Popow R, Ariyan C, Rossi F (2011) Imatinib potentiates antitumor T cell responses in gastrointestinal stromal tumor through the inhibition of Ido. Nat Med 17:1094
Pauken KE, Juneja V, McGuire K, LaFleur M, Kuchroo J, Sage P, Chevrier N, Freeman GJ, Sharpe AH (2017) CD8+ T cell-independent mechanisms of PD-1-mediated suppression of anti-tumor immunity in mice. Am Assoc Immnol 198(205):11
Kreutzman A, Ladell K, Koechel C, Gostick E, Ekblom M, Stenke L, Melo T, Einsele H, Porkka K, Price D (2011) Expansion of highly differentiated CD8+ T-cells or NK-cells in patients treated with dasatinib is associated with cytomegalovirus reactivation. Leukemia 25:1587
Brück O, Blom S, Dufva O, Turkki R, Chheda H, Ribeiro A, Kovanen P, Aittokallio T, Koskenvesa P, Kallioniemi O (2018) Immune cell contexture in the bone marrow tumor microenvironment impacts therapy response in CML. Leukemia 32:1643–1656
Larmonier N, Janikashvili N, LaCasse CJ, Larmonier CB, Cantrell J, Situ E, Lundeen T, Bonnotte B, Katsanis E (2008) Imatinib mesylate inhibits CD4+ CD25+ regulatory T cell activity and enhances active immunotherapy against BCR-ABL− tumors. J Immunol 181:6955–6963
Fei F, Yu Y, Schmitt A, Rojewski MT, Chen B, Götz M, Döhner H, Bunjes D, Schmitt M (2009) Dasatinib inhibits the proliferation and function of CD4+ CD25+ regulatory T cells. Br J Haematol 144:195–205
Fei F, Yu Y, Schmitt A, Rojewski MT, Chen B, Greiner J, Götz M, Bunjes D, Schmitt M (2010) Effects of nilotinib on regulatory T cells: the dose matters. Mol Cancer 9:22
Lee M, Park C-J, Cho Y-U, Jang S (2019) Expression levels of PD-1 on T cells in chronic myeloid leukemia: PB1936. HemaSphere 3:880–881
Acknowledgements
The authors would like to thank Chan Hee Yoon, MT, and Sang Hee Han, MT, in the department of laboratory medicine in Asan Medical Center for technical assistance with flow cytometry. Portions of this study have been published in Abstract Book at the 24th Congress of the European Hematology Association, Amsterdam, The Netherlands, on June 13–16, 2019 [23].
Funding
This study was supported by a Grant (2016–635) from the Asan Institute for Life Science, Asan Medical Center, Seoul, Korea.
Author information
Authors and Affiliations
Contributions
CP and YC designed the study, and ML wrote the paper. ML and EY carried out the data management and analysis. SJ, CS, ES, EC, and JL provided study materials or patients. All authors have agreed with the final version of the paper.
Corresponding authors
Ethics declarations
Ethics approval
This study was approved by the institutional review board of Asan Medical Center (Approval number: 20161087) and was performed in accordance with the Declaration of Helsinki.
Conflicts of interest
No potential conflicts of interest relevant to this article were reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lee, M.Y., Park, CJ., Cho, YU. et al. Differences in PD-1 expression on CD8+ T-cells in chronic myeloid leukemia patients according to disease phase and TKI medication. Cancer Immunol Immunother 69, 2223–2232 (2020). https://doi.org/10.1007/s00262-020-02617-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02617-5