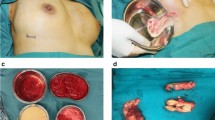
Abstract
From 1997 to 2006, polyacrylamide hydrogel (PAAG) was approved for use in China as a permanent filler for breast augmentation, and it is estimated that 200,000 women have undergone PAAG injection since then. After injection, complications such as pain, mass, hematoma, asymmetry, migration, infection, and even cancer continue to emerge. Because of the potential toxicity and unstable nature of the material and the nonstandardized injection layers, complications after PAAG injection breast augmentation are often complex and difficult to treat. The only treatment for these complications is debridement surgery, which includes PAAG evacuation, capsule remove, lesion excision, and mastectomy. Currently, although there are a variety of surgical methods for complications after PAAG injection, there is a lack of consensus regarding the diagnosis and treatment. We systematically review the literature and summarize our experience of diagnosis and treatment of complications after PAAG injection in our hospital over the past 10 years. To date, this is the first attempt to propose a diagnostic classification for PAAG injection breast augmentation and to set out a treatment strategy based on this classification. Although the China Food and Drug Administration withdrew its approval in 2006, PAAG is still being used illegally in some areas, and the patient population is widespread. This study aims to provide a more comprehensive understanding of PAAG complications to drive the standard diagnosis and treatment based on clinical classification, and to provide references for the future development of safer injectable products.
Level of Evidence IV This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.







Similar content being viewed by others
References
Broder KW, Cohen SR (2006) An overview of permanent and semipermanent fillers. Plast Reconstr Surg 118(3S):7S–14S
Lemperle G, Morhenn V, Charrier U (2003) Human histology and persistence of various injectable filler substances for soft tissue augmentation. Aesthet Plast Surg 27(5):354–366
Hedén P, Sellman G, von Wachenfeldt M et al (2009) Body shaping and volume restoration: the role of hyaluronic acid. Aesthet Plast Surg 33(3):274–282
Hivernaud V, Lefourn B, Guicheux J et al (2015) Autologous fat grafting in the breast: critical points and technique improvements. Aesthet Plast Surg 39(4):547–561
Cohen O, Lam G, Karp N, Choi M (2017) Determining the oncologic safety of autologous fat grafting as a reconstructive modality: an institutional review of breast cancer recurrence rates and surgical outcomes. Plastic Reconstr Surg 140(3):e382–e392
Zemskov VS, Zavgorodniĭ IA, Roshchina LA et al (2000) Endoprosthesis of mammary glands using hydrogel prosthesis PAAG Interfall. Klin Khir 6:23–24
Hen NB, Yuan WZ, Shuai XR (2010) Clinical analysis of removal of injecting polyacrylamide hydrogel through the mammary areolar incision in 68 cases Chin J Aesthet Plastic Surg (10):617–618
Shi HS, Cao C, Chen L, Li XG, Li SR (2015) Clinical analysis and management of complications of breast augmentation with injected polyacrylamide hydrogel. J Reg Anat Oper Surg 24(3):263–266
Chen L, Sha L, Huang SP et al (2015) Treatment for displacement of PAAG mixture after injection augmentation mammoplasty. Int J Clin Exp Med 8(3):3360
Ono S, Ogawa R, Hyakusoku H (2010) Complications after polyacrylamide hydrogel injection for soft-tissue augmentation. Plast Reconstr Surg 126(4):1349–1357
Shen H, Lv Y, Xu J et al (2012) Complications after polyacrylamide hydrogel injection for facial soft-tissue augmentation in China: twenty-four cases and their surgical management. Plast Reconstr Surg 130(2):340e–348e
Cheng NX, Liu LG, Hui L et al (2009) Breast cancer following augmentation mammaplasty with polyacrylamide hydrogel (PAAG) injection. Aesthet Plast Surg 33(4):563
Xiao Z, Liu Y (2008) The relationship between breast cancer and breast augmentation with injected polyacrylamide gel: two case reports. J Plast Reconstr Aesthet Surg 61(8):981–982
Liu HL, Cheung WY (2010) Complications of polyacrylamide hydrogel (PAAG) injection in facial augmentation. J Plast Reconstr Aesthet Surg 63(1):e9–e12
Pallua N, Wolter TP (2010) A 5-year assessment of safety and aesthetic results after facial soft-tissue augmentation with polyacrylamide hydrogel (Aquamid): a prospective multicenter study of 251 patients. Plast Reconstr Surg 125(6):1797–1804
Unukovych D, Khrapach V, Wickman M et al (2012) Polyacrylamide gel injections for breast augmentation: management of complications in 106 patients, a multicenter study. World J Surg 36(4):695–701
Luo SK, Chen GP, Sun ZS et al (2011) Our strategy in complication management of augmentation mammaplasty with polyacrylamide hydrogel injection in 235 patients. J Plast Reconstr Aesthet Surg JPRAS 64(6):731–737
Lin DC, Yurke B, Langrana NA (2004) Mechanical properties of a reversible, DNA-crosslinked polyacrylamide hydrogel. J Biomech Eng 126(1):104–110
Leung KM, Yeoh GPS, Chan KW (2007) Breast pathology in complications associated with polyacrylamide hydrogel (PAAG) mammoplasty. Hong Kong Med J 13(2):137
King DJ, Noss RR (1989) Toxicity of polyacrylamide and acrylamide monome. Rev Environ Health 8(1–4):3–16
Huo M, Huang J, Qi K (2002) Experimental study on the toxic effects of hydrophilic polyacrylamide gel. Chin J Plast Surg 18(2):79
Andersen FA (2005) Amended final report on the safety assessment of polyacrylamide and acrylamide residues in cosmetics. Int J Toxicol 24:21–50
Smith EA, Oehme FW (1991) Acrylamide and polyacrylamide: a review of production, use, environmental fate and neurotoxicity. Rev Environ Health 9(4):215–228
Cheng N, Wang Y, Wang J et al (2002) Complications of breast augmentation with injected hydrophilic polyacrylamide gel. Aesthet Plast Surg 26(5):375–382
Zhao Y, Yuan NA, Li K et al (2015) Bilateral breast cancer following augmentation mammaplasty with polyacrylamide hydrogel injection: a case report. Oncol Lett 9(6):2687–2693
Chen G, Wang Y, Huang JL (2016) Breast cancer following polyacrylamide hydrogel injection for breast augmentation: a case report. Mol Clin Oncol 4(3):433
Joint FAO, Ng J, WHO Expert Committee on Food Additives (2010) Joint FAO/WHO Expert Committee on Food Additives Seventy-second meeting: Summary and conclusions. FAO/WHO
Lin WC, Hsu GC, Hsu YC et al (2008) A late complication of augmentation mammoplasty by polyacrylamide hydrogel injection: ultrasound and magnetic resonance imaging findings of huge galactocele formation in a puerperal woman with pathological correlation. Breast J 14(6):584–587
Wang ZX, Luo DL, Dai X et al (2012) Polyacrylamide hydrogel injection for augmentation mammaplasty: loss of ability for breastfeeding. Ann Plast Surg 69(2):123–128
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Electronic supplementary material
Below is the link to the electronic supplementary material.

266_2017_1006_MOESM1_ESM.jpg
Sup. Figure 1. Preoperative photographs of case 1. A. Preoperative anterior view; B. Preoperative lateral view; C. MRI showing that the injection layer is mainly under the breast, and the capsule is intact (JPEG 34 kb)

266_2017_1006_MOESM2_ESM.jpg
Sup. Figure 2. Pre and postoperative photographs of case 2. A, B are preoperative anterior and lateral views and C, D are postoperative anterior and lateral views (JPEG 54 kb)
Rights and permissions
About this article
Cite this article
Jin, R., Luo, X., Wang, X. et al. Complications and Treatment Strategy After Breast Augmentation by Polyacrylamide Hydrogel Injection: Summary of 10-Year Clinical Experience. Aesth Plast Surg 42, 402–409 (2018). https://doi.org/10.1007/s00266-017-1006-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-017-1006-9