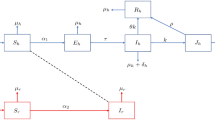
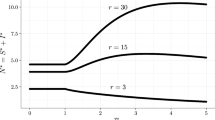
Abstract
We applied optimal control theory to an SI epidemic model to identify optimal culling strategies for diseases management in wildlife. We focused on different forms of the objective function, including linear control, quadratic control, and control with limited amount of resources. Moreover, we identified optimal solutions under different assumptions on disease-free host dynamics, namely: self-regulating logistic growth, Malthusian growth, and the case of negligible demography. We showed that the correct characterization of the disease-free host growth is crucial for defining optimal disease control strategies. By analytical investigations of the model with negligible demography, we demonstrated that the optimal strategy for the linear control can be either to cull at the maximum rate at the very beginning of the epidemic (reactive culling) when the culling cost is low, or never to cull, when culling cost is high. On the other hand, in the cases of quadratic control or limited resources, we demonstrated that the optimal strategy is always reactive. Numerical analyses for hosts with logistic growth showed that, in the case of linear control, the optimal strategy is always reactive when culling cost is low. In contrast, if the culling cost is high, the optimal strategy is to delay control, i.e. not to cull at the onset of the epidemic. Finally, we showed that for diseases with the same basic reproduction number delayed control can be optimal for acute infections, i.e. characterized by high disease-induced mortality and fast dynamics, while reactive control can be optimal for chronic ones.



Similar content being viewed by others
References
Abakuks A (1973) An optimal isolation policy for an epidemic. J Appl Probab 10:247–262
Abakuks A (1974) Optimal immunization policies for epidemics. Adv Appl Probab 6:494–511
Ainseba B, Iannelli M (2012) Optimal screening in structured SIR epidemics. Math Model Nat Phenom 7:12–27
Anderson R, Jackson H, May R, Smith A (1981) Population dynamics of rabies in Europe. Nature 289: 765–771
Anderson R, May R (1979) Population biology of infectious diseases: Part I. Nature 280:361–367
Asano E, Gross L, Lenhart S, Real L (2008) Optimal control of vaccine distribution in a rabies metapopulation model. Math Biosci Eng 5:219–238
Barlow N (1996) The ecology of wildlife disease control: simple models revisited. J Appl Ecol 33:303–314
Beeton N, MacCallum H (2011) Models predict that culling is not a feasible strategy to prevent extinction of tasmanian devils from facial tumour disease. J Appl Ecol 48:1315–1323
Behncke H (2000) Optimal control of deterministic epidemics. Optim Control Appl Methods 21:269–285
Bolzoni L, De Leo G (2007) A cost analysis of alternative culling strategies for the eradication of classical swine fever in wildlife. Environ Dev Econ 12:653–671
Bolzoni L, De Leo G (2013) Unexpected consequences of culling on the eradication of wildlife diseases: the role of virulence evolution. Am Nat 181:301–313
Bolzoni L, Real L, De Leo G (2007) Transmission heterogeneity and control strategies for infectious disease emergence. Plos ONE 2:e747
Bolzoni L, De Leo G, Gatto M, Dobson A (2008) Body-size scaling in an SEI model of wildlife diseases. Theoret Popul Biol 73:374–382
Bonds M (2006) Host life-history strategy explains pathogen-induced sterility. Am Nat 168:281–293
Bortins R, Boustany NM, Powers WF (1980) The infinite-order singular problem. Optim Control Appl Methods 1:279–302
Choisy M, Rohani P (2006) Harvesting can increase severity of wildlife disease epidemics. Proc R Soc B 273:2025–2034
Clayton T, Duke-Sylvester S, Gross L, Lenhart S, Real L (2010) Optimal control of a rabies epidemic model with a birth pulse. J Biol Dyn 4:43–58
Cleaveland S, Laurenson K, Mlengeya T (2005) Impacts of wildlife infections on human and livestock health with special reference to Tanzania: implications for protected area management. In: Osofsky S, Cleaveland S, Karesh W, Kock M, Nyhus P, Starr L, Yang A (eds) Conservation and development interventions at the wildlife/livestock interface: implications for wildlife, livestock and human health. IUCN, Gland, pp 147–151
Coyne M, Smith G, McAllister F (1989) Mathematical model for the population biology of rabies in raccoons in the mid-atlantic states. Am J Vet Res 50:2148–2154
Daszak P, Cunningham A, Hyatt A (2000) Emerging infectious diseases of wildlife–threats to biodiversity and human health. Science 287:443–449
de Pillis L, Gu W, Fister K, Head T, Maples K, Murugan A, Neal T, Yoshida K (2007) Chemotherapy for tumors: an analysis of the dynamics and a study of quadratic and linear optimal controls. Math Biosci 209:292–315
DEFRA/DCMS (2002) Economic cost of foot and mouth disease in the UK. Tech. rep, Joint working paper of the Department for the Environment, Food and Rural Affairs and the Department of Culture, Media and Sport, UK
Dobson A, Meagher M (1996) The population dynamics of brucellosis in the Yellowstone national park. Ecology 77:1026–1036
Donaldson A (1997) Foot-and-mouth disease in Taiwan. Vet Rec 140:407
Donnelly C, Woodroffe R, Cox D, Bourne J, Gettinby G, Le Fevre A, McInerney J, Morrison W (2003) Impact of localized badger culling on tuberculosis incidence in British cattle. Nature 426:834–837
Donnelly C, Woodroffe R, Cox D, Cheeseman C, Clifton-Hadley R, Wei G, Gettinby G, Gilks P, Jenkins H, Johnston W, Le Fevre A, McInerney J, Morrison W (2006) Positive and negative effects of widespread badger culling on tubercolosis in cattle. Nature 439:843–846
Fenichel E, Horan R (2007) Jointly-determined ecological thresholds and economic trade-offs in wildlife disease management. Nat Resour Model 20:511–547
Ferguson N, Donnelly C, Anderson R (2001a) The foot-and-mouth epidemic in Great Britain: pattern of spread and impact of interventions. Science 292:1155–1160
Ferguson N, Donnelly C, Anderson R (2001b) Transmission intensity and impact of control policies on the foot and mouth epidemic in Great Britain. Nature 413:542–547
Fleming W, Rishel R (1975) Deterministic and stochastic optimal control. Springer, New York
Fonkwo P (2008) Pricing infectious disease. The economic and health implications of infectious diseases. EMBO Rep 8:S13–S17
Gaillard J, Festa-Bianchet M, Yoccoz N, Loison A, Toïgo C (2000) Temporal variation in fitness components and population dynamics of large herbivores. Annu Rev Ecol Evol Syst 31:367–393
Gibbens J, Sharpe C, Wilesmith J, Mansley L, Michalopoulou E, Ryan J, Hudson M (2001) Descriptive epidemiology of the 2001 foot-and-mouth disease epidemic in Great Britain: the first five months. Vet Rec 149:729–743
Handel A, Longini I, Antia R (2007) What is the best control strategy for multiple infectious disease outbreaks. Proc R Soc B 274:833–837
Hansen E, Day T (2011a) Optimal antiviral treatment strategies and the effects of resistance. Proc R Soc B 278:1082–1089
Hansen E, Day T (2011b) Optimal control of epidemics with limited resources. J Math Biol 62:423–451
Hofmann M, Thür B, Vanzetti T, Schleiss W, Schmidt J, Griot C (1999) Klassische schweinepest beim wildschwein in der schweiz. Schweizerisches Archiv für Tierheilkunde 141:185–190
Horan R, Wolf C (2005) The economics of managing infectious wildlife disease. Am J Agric Econ 87: 537–551
Horan R, Wolf C, Fenichel E, Mathews K (2005) Spatial management of wildlife disease. Rev Agric Econ 27:483–490
Howard J, Donnelly C (2000) The importance of immediate destruction in epidemics of foot and mouth disease. Res Vet Sci 69:189–196
Kaden V (1999) Bekämpfung der klassischen schweinepest beim schwarzwild. Zeitschrift für Jagdwissenschaften 45:45–59
Keeling M, Woolhouse M, May R, Davies G, Grenfell B (2003) Modelling vaccination strategies against foot-and-mouth disease. Nature 421:136–142
Krener A (1977) The high order maximal principle and its application to singular extremals. SIAM J Control Optim 15:256–293
Lachish S, McCallum H, Mann D, Pukk C, Jones M (2010) Evaluation of selective culling of infected individuals to control Tasmanian devil facial tumor disease. Conserv Biol 24:841–851
Laddomada A (2000) Incidence and control of CSF in wild boar in Europe. Vet Microbiol 73:121–130
Laddomada A, Patta C, Oggiano A, Caccia A, Ruiu A, Cossu P, Firinu A (1994) Epidemiology of classical swine fever in sardinia: a serological survey of wild boar and comparison with african swine fever. Vet Rec 134:183–187
Lee S, Chowell G, Chastillo-Chavez C (2010) Optimal control for pandemic influenza: the role of limited antiviral treatment and isolation. J Theoret Biol 265:136–150
Lenhart S, Workman J (2007) Optimal control applied to biological models. CRC Mathematical and Computational Biology Series. Chapman & Hall, Boca Raton
Lloyd-Smith J, Cross P, Briggs C, Daugherty M, Getz W, Latto J, Sanchez M, Smith A, Swei A (2005) Should we expect population thresholds for wildlife disease? Trends Ecol Evol 20:511–519
Maurer H (1977) On optimal control problems with bounded state variables and control appearing linearly. SIAM J Control Optim 15:345–362
McCallum H, Barlow N, Hone J (2001) How should pathogen transmission be modelled. Trends Ecol Evol 16:295–300
Meuwissen M, Horst H, Huirne R, Dijkhuizen A (1999) A model to estimate the financial consequences of classical swine fever outbreaks: principles and outcomes. Prevent Vet Med 42:249–270
Mollison D (1991) Dependence of epidemic and population velocities on basic parameters. Math Biosci 107:255–287
Morters M, Restif O, Hampson K, Cleaveland S, Woods J, Conlan A (2013) Evidence-based control of canine rabies: a critical review of population density reduction. J Anim Ecol 82:6–14
Morton R, Wickwire K (1974) On the optimal control of a deterministic epidemic. Adv Appl Probab 6:622–635
Neilan R, Lenhart S (2011) Optimal vaccine distribution in a spatiotemporal epidemic model with an application to rabies and raccoons. J Math Anal Appl 378:603–619
Okosun K, Ouifki R, Marcus N (2011) Optimal control analysis of a malaria disease transmission model that includes treatment and vaccination with waning immunity. Biosystems 106:136–145
Pontryagin L, Boltyanskii V, Gamkrelidze R, Mishchenko E (1962) The mathematical theory of optimal processes. Interscience Publishers, Los Angeles
Potapov A, Merrill E, Lewis M (2012) Wildlife disease elimination and density dependence. Proc R Soc B 279:3139–3145
Powers W (1980) On the order of singular optimal control problems. J Optim Theory Appl 32:479–489
Rosatte R, Donovan D, Allan M, Howes LA, Silver A, Bennett K, MacInnes C, Davies C, Wandeler A, Radford B (2001) Emergency response to raccoon rabies introduction into Ontario. J Wildl Dis 37: 265–279
Schnyder M, Stärk K, Vanzetti T, Salman M, Thor B, Schleiss W, Griot C (2002) Epidemiology and control of an outbreak of classical swine fever in wild boar in Switzerland. Vet Rec 150:102–109
Seierstad A, Sydsaeter K (1987) Optimal control theory with economic applications. North Holland, Amsterdam
Smith G, Bennett R, Wilkinson D, Cooke R (2007) A cost-benefit analysis of culling badgers to control bovine tubercolosis. Vet J 173:302–310
Smith G, Cheeseman C (2002) A mathematical model for the control of diseases in wildlife populations: culling, vaccination and fertility control. Ecol Modell 150:45–53
Volterra V (1927) Fluctuations in the abundance of a species considered mathematically. Nature 119:12–13
Wickwire K (1975) Optimal isolation policies for deterministic and stochastic epidemics. Math Biosci 26:325–346
Woodroffe R, Cleaveland S, Courtenay O, Laurenson M, Artois M (2004) Infectious disease in the management and conservation of wild canids. In: Macdonald D, Sillero-Zubiri C (eds) The biology and conservation of wild canids. Oxford University Press, Oxford, pp 124–142
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bolzoni, L., Tessoni, V., Groppi, M. et al. React or wait: which optimal culling strategy to control infectious diseases in wildlife. J. Math. Biol. 69, 1001–1025 (2014). https://doi.org/10.1007/s00285-013-0726-y
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-013-0726-y