Abstract
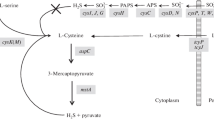
The A. nidulans cysD gene encoding homocysteine synthase (O-acetyl-L-homoserine sulphydrylase) has been isolated by functional complementation of a cysD11 mutation. The gene contains five short introns and codes for a protein of 437 amino acids. The protein shows homology with bacterial and yeast O-acetyl- and O-succinyl-homoserine sulphydrylases, particularly from Schizosaccharomyces pombe, Saccharomyces cerevisiae and Kluyveromyces lactis. The cysD cDNA is able to complement a S. cerevisiae mutation impairing homocysteine synthase. Synthesis of the cysD mRNA is down-regulated by a high concentration of methionine in growth medium without sulphate and up-regulated under sulphur limitation. A comparison of cysD genomic and cDNA copies, derived from different A. nidulans strains, revealed a marked DNA-sequence polymorphism manifested mostly by silent point mutations. There was, however, much less polymorphism in the protein sequence.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 18 July / 27 October 1997
Rights and permissions
About this article
Cite this article
Sieńko, M., Topczewski, J. & Paszewski, A. Structure and regulation of cysD, the homocysteine synthase gene of Aspergillus nidulans. Curr Genet 33, 136–144 (1998). https://doi.org/10.1007/s002940050319
Issue Date:
DOI: https://doi.org/10.1007/s002940050319