Abstract
Objective
To assess antibody response to inactivated COVID-19 vaccine in patients with immune-mediated diseases (IMD) among hospital workers and people aged 65 and older.
Methods
In this cross-sectional study, we studied 82 hospital workers with IMD (mean age: 42.2 ± 10.0 years) and 300 (mean age: 41.7 ± 9.9 years) controls. Among + 65 aged population, we studied 22 (mean age: 71.4 ± 4.5 years) patients and 47 controls (mean age: 70.9 ± 4.8 years). All study subjects had a negative history for COVID-19. Sera were obtained after at least 21 days following the second vaccination. Anti-spike IgG antibody titers were measured quantitatively using a commercially available immunoassay method.
Results
Patients with IMD were significantly less likely to have detectable antibodies than healthy controls both among the hospital workers (92.7% vs 99.7%, p < 0.001) and elderly population (77.3% vs 97.9%, p = 0.011). Among patients with IMD, those using immunosuppressive or immune-modulating drugs (64/75, 85.3%) were significantly less likely to have detectable antibodies compared to those off treatment (29/29, 100%) (p = 0.029). Additionally, a negative association between age and the antibody titer categories among patients (r = − 0.352; p < 0.001) and controls (r = − 0.258; p < 0.001) were demonstrated.
Conclusions
Among hospital workers, the vast majority of patients with IMD and immunocompetent controls developed a significant humoral response following the administration of the second dose of inactivated COVID-19 vaccine. This was also true for the elderly population, albeit with lower antibody titers. Immunosuppressive use, particularly rituximab significantly reduced antibody titers. Antibody titers were significantly lower among those aged ≥ 60 years both in patient and control populations. Whether these individuals should get a booster dose warrants further studies.
Similar content being viewed by others
Introduction
The COVID-19 pandemic has been globally responsible for 169,118,995 confirmed cases and 3,398,302 deaths as of 30 May 2021 [1]. Multiple potential vaccines against COVID-19 have been developed at an unprecedented speed and as of December 2020, mass vaccination programs were started worldwide. As a result, within 6 months a total of 1,546,316,352 vaccine doses have been administered [1].
As of January 13, 2021, Turkey granted emergency use authorization to ‘CoronaVac’ produced by Chinese biopharmaceutical company Sinovac [2,3,4]. CoronaVac is a chemically inactivated vaccine developed against SARS-CoV-2 [5, 6]. As the phase III trial of CoronaVac was still ongoing the authorization was based on the published results on phase I and II trials [5, 6] and unpublished results on “interim analyses” [7]. When the current study took place Turkey had made agreement with only Sinovac and CoronaVac was the only vaccine available in the country [2,3,4].
Current guidelines [8] recommend vaccinating patients with autoimmune and inflammatory rheumatic diseases (AIIRD) whether or not they are using immuno-modulatory agents based on their risk for COVID-19 and these patients are now prioritized for vaccination in many countries [9,10,11]. Since registration trials of vaccines against SARS-CoV-2 did not include patients with AIIRD [12, 13] and current evidence from pre COVID-19 era literature is scarce, there are many unknown questions at this time. Major concern is the uncertainty about whether these patients can mount a protective immune response to vaccine either because of their inherent immunocompromised status or because of the effect of commonly used biological and non-biological disease modifying anti-rheumatic drugs (DMARDs) [14, 15]. The same concerns are also true for the elderly population who are at increased risk for severe COVID-19 [16] and usually excluded from the vaccine trials.
We, therefore, in this study evaluated the humoral immune response of patients with immune-mediated diseases (IMD) to inactivated COVID-19 vaccine compared to immunocompetent controls. Healthcare workers of a single university hospital and elderly population who were prioritized for vaccination during the first months of the vaccination campaign in Istanbul, Turkey constituted our study population.
Patients and methods
Vaccination program in Turkey
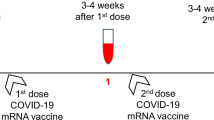
On January 14, 2021, a mass vaccination campaign started first with healthcare workers and then continued with the elderly after January 28 (Fig. 1). Vaccine inoculation was done with two shots of CoronaVac 4 weeks apart.
Study population
This cross-sectional study was conducted at Cerrahpasa Medical Faculty of Istanbul University-Cerrahpasa during the first months of the vaccination roll-out, between January 14 and May 2, 2021. Hospital workers (aged between 21 and 64) and elderly people (aged between 65 and 90) who attended the COVID-19 vaccine center for their scheduled vaccine appointment constituted the source of our study population. Those who volunteered to participate in the study fulfilled a standardized survey (electronic supplement material) that assessed demographic and health-related characteristics. The survey form was distributed to eligible participants at the day of the first dose of vaccination. The survey included questions about basic demographic information, body and height measures, smoking status, current medications, previous COVID-19 diagnosis, and history of rheumatic diseases, inflammatory disease, malignancy (solid organ or hematologic), transplantation, or immune-suppressive use. Comorbid diseases were identified according to Charlson index list [17]. Those who had contracted COVID-19 verified with positive PCR test or those who were considered symptomatic for COVID-19 infection but had negative PCR test were not included in the study. Moreover, only those who got two shots of CoronaVac were studied.
Figure 1 shows the flowchart of the enrollment of patients and controls. Patients with IMD were defined as all those who had a history of inflammatory disease including rheumatic diseases, psoriasis, inflammatory bowel diseases and multiple sclerosis currently under medical treatment or off treatment or any condition that requires chronic immunosuppressive use. These patients with IMD were later interviewed with telephone to collect information about disease duration and current and previous treatments. Immunosuppressive use during the vaccination period and the last three months before the vaccination was specifically sought.
Thus, we identified 98 patients with IMD among the hospital workers population and 25 patients with IMD among the elderly population. We assigned two separate age and gender matched control groups for the two patient groups with an allocation ratio of 1:3 to study age effect better in a large control population. Therefore, as control groups, we planned to study 300 and 75 individuals with no IMD selected consecutively among the eligible candidates of the hospital workers and elderly populations, respectively. Patients and controls were invited for blood collection 3 weeks after their second dose of vaccination. A total of 104 patients (82 hospital workers and 22 elderly) and 347 controls (300 hospital workers and 47 elderly) accepted our invitation. It has to be noted that most of the elderly who refused our invitation expressed their concerns about coming to the hospital because of the ongoing pandemic. All blood samples were collected between March 9 and May 2, 2021.
Description of disease subgroups and treatment regimens
We preferred to label the patient population as patients with IMD although all rheumatic or inflammatory diseases included here are not immune mediated. To study the effect of different disease subgroups and immunosuppressive agents we combined all 104 patients with IMD (hospital workers and elderly) and categorized into subgroups. We defined disease subsets as rheumatoid arthritis (RA) (n = 19); connective tissue diseases (CTD) (n = 17), spondylarthropathies (SPA)/inflammatory bowel diseases (IBD) (n = 29), Behcet’s syndrome (BS) (n = 16), familial Mediterranean fever (FMF) (n = 10), multiple sclerosis (MS) (n = 5) and vasculitis (n = 7). One kidney transplant recipient was excluded from these analyses.
We categorized treatment regimens as rituximab (RTX)-based treatment (n = 7), non-RTX biological agents-based treatment (n = 25), conventional DMARDs-based treatment (n = 27), colchicine (n = 13) and other immune-modulating drugs (such as fingolimod: n = 1 and dimethyl fumarate: n = 2) and no treatment (n = 29).
Antibody testing
All sera samples were aliquoted after centrifugation of peripheral blood tubes at 800 × g for 12 min and sera samples were kept in – 20 ℃ until the study day. Minimum 100 μL of serum was required for analysis. For detection of antibodies against receptor binding domain of the S1 spike protein (anti-spike SARS-CoV-2 IgG) we used the commercially available Elecsys® Anti-SARS-CoV-2 assay (Roche Diagnostics International Ltd, Rotkreuz, Switzerland) [18]. Quantitative results were determined using in vitro immunoassay. A cut-off value of ≥ 0.8 U/mL was accepted as positive according to the manufacturer’s instructions [18]. The upper limit of detection by the assay was 250 U/mL.
Statistical methods
Numeric results were expressed as mean ± standard deviation or median (minimum–maximum), and categorical results were expressed as n (%). Categorical variables were compared using Pearson, Yates or Fisher chi-square test and continuous variables with Student’s t test or Mann–Whitney U test. Spearmen correlation test was used to investigate association between antibody titer and age. We calculated age adjusted Charlson comorbidity index as reported [17]. We analyzed antibody titers categorically. In the current study, among 300 controls (< 65 years of age), 109 (36.3%) had antibody titers > 250 U/mL, 88 (29.3%) had between 250 and 117 U/mL and 103 (34.3%) < 117 U/mL. Therefore, we identified high, medium and low titers as upper 35th (> 250 U/mL), middle 30th (between 250 and 117 U/mL) and lower 35th percentile (< 117 U/mL). The effect of age (≥ 60 years), gender, body mass index, smoking and having IMD on positive antibody test was investigated using multivariate logistic regression analysis. A p value of less than 0.05 was accepted as statistical significance. IBM SPSS Statistics for Windows, v.20.0 (IBM Corp., Armonk, NY, USA) was used in the statistical analysis.
Results
Among hospital workers, we studied 82 (29 M/ 53 F; mean age: 42.2 ± 10.0 years) patients with IMD and 300 (107 M/ 193 F; mean age: 41.7 ± 9.9 years) controls (Table 1 and 2). Among + 65 aged population, on the other hand, we studied 22 (6 M/ 16 F; mean age: 71.4 ± 4.5 years) patients with IMD and 47 controls (23 M/ 24 F; mean age: 70.9 ± 4.8 years).
Clinical characteristics and immunosuppressive treatment of the patients with IMD
Patients with IMD had various diagnoses with the majority being rheumatic diseases as shown in Table 1. Apart from rheumatic diseases, patients have psoriasis (n = 3), inflammatory bowel diseases (n = 5), and multiple sclerosis (n = 5). There was one kidney transplant recipient included in the study because of his using immunosuppressive drugs. At the time of vaccination and for at least 3 months before vaccination, 32.9% and 9.1% of the patients among the hospital workers and elderly, respectively, were not using any immunosuppressive, corticosteroids or colchicine. Biological disease modifying anti-rheumatic drugs (DMARD’s) and conventional DMARD’s were the most commonly used drugs by the two patient groups (Table 1).
Antibody results
-
a)
Study groups
Mean time interval after the second dose of vaccination to blood collection was 30.7 ± 9.0 days for the whole study population (patients: 27.3 ± 7.6; min–max: 19–68; controls: 31.7 ± 9.1; min–max: 19–57).
Patients with IMD and their respective controls were similar with regards to age, gender and body mass index (Table 2). Age adjusted Charlson comorbidity index was significantly higher among patients with IMD compared to controls in both hospital workers and elderly population (Table 2).
Among hospital workers, patients with IMD were less likely to have detectable antibodies than controls (92.7% vs 99.7%, p < 0.001) (Table 2). This was also true for those patients with IMD in the elderly population (77.3% vs 97.9%, p = 0.011) (Table 2). While categorical antibody titers did not differ between patients and controls among hospital workers; elderly patients had significantly lower levels compared to elderly controls (p = 0.045) as shown in Table 2 and Fig. 2.
-
b)
Patients with IMD (hospital workers and elderly population combined)
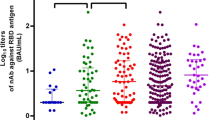
The frequency of those with detectable antibody did not differ between different subgroups (RA: 15/19, 78.9%; CTD: 14/17, 82.4%; SPA/IBD: 28/29, 96.6%; BS: 15/16, 93.8%; FMF: 10/10, 100%; MS: 5/5, 100% and vasculitis: 5/7, 71.4%; p = 0.170). On the other hand, categorized antibody titers showed significant difference across disease subgroups (p = 0.037), as shown in Fig. 3A. While patients with vasculitis and CTD had the lowest titers, patients with FMF, BS and MS had the highest titers (Fig. 3A).
Analysis between different treatment regimen subgroups showed significant differences with regard to positivity (p < 0.001) and categorized antibody titers (p < 0.001) (Table 3). Those who were receiving rituximab-based treatment (1/7, 14.3%), were significantly less likely to have detectable antibodies compared to those who were using non-RTX biological agents-based treatment (22/25, 88%), conventional DMARDs-based treatment (25/27, 92.6%), colchicine and MS drugs (16/16, 100%) or no treatment (29/29, 100%) (p < 0.001). Categorized antibody titers are shown in Table 3 and Fig. 3B. It is apparent that those who are using either biological agents (particularly RTX) or conventional DMARDs had significantly reduced antibody titers compared to those who are not using any immunosuppressive treatment (Table 3 and Fig. 3B). On the other hand, colchicine and immune-modulating drugs used in MS seemed to not affect antibody titers.
-
c)
Age effect on categorized antibody titers
Additionally, we found a negative association between age and categorized antibody titers among patients with IMD (r = − 0.352; p < 0.001) as well as among the controls (r = − 0.258; p < 0.001). As shown in Fig. 4, both in patients (Fig. 4A) and controls (Fig. 4B), antibody titers started to decrease after third decade of life and dropped significantly after the sixth (p < 0.001 for both patients and controls).
-
d)
Multivariate logistic regression analysis
The effect of age (≥ 60 years), gender, body mass index, smoking and having IMD were investigated using multivariate logistic regression analysis in the whole study cohort. Being diagnosed as IMD [OR 17.31; 95% CI (3.57–85.95), p < 0.001] and being 60 years or more [OR 4.32; 95% CI (1.20–15.50), p = 0.025] were found to be independently associated with having negative antibody result.
Categorized antibody titers between disease subgroups (A) and different treatment regimens (B). RA rheumatoid arthritis, CTD connective tissue disease, SPA spondyloarthropathies, IBD inflammatory bowel diseases, BS Behcet’s syndrome, FMF familial Mediterranean fever, MS multiple sclerosis. Patient with kidney transplant recipient was excluded from disease subgroup analysis RTX rituximab, DMARD disease modifying anti-rheumatic drug. aOther immune-modulating drugs such as fingolimod and dimethyl fumarate
Discussion
In this study of the antibody response to two doses of inactivated SARS-CoV-2 vaccine among patients with IMD and a large cohort of controls, the majority had detectable anti-spike antibody responses. This was also true for the elderly. However, those who were using immunosuppressives (either biological or conventional DMARD’s) were found to have significantly lower levels of antibody titers than those who were off treatment. Additionally, the study revealed a significant negative association between age and antibody titers in both patient and control populations. Finally, having IMD and being older (≥ 60 years) were found to be independently associated with a negative antibody response in the whole study population.
Antibody response to vaccine against SARS-CoV-2 in patients with IMD or patients on immunosuppression has been investigated in a few studies recently [19,20,21,22,23,24] and notably in all, mRNA vaccine was studied. To our knowledge, ours is the first with chemically inactivated vaccine (CoronaVac). The results of the current study are important in that, we were able to investigate patients with IMD and elderly two different populations primarily at risk for severe COVID-19 and usually omitted from the vaccine trials [12, 13, 16].
Our study revealed that patients with vasculitis and CTD had significantly lower levels of antibody titers compared to those with RA, SPA/IBH, BS, FMF and MS. Those who were using any immunosuppressive drug (either conventional or biological) were significantly less likely to develop high antibody titers compared to those who were not. This effect was more pronounced among those receiving RTX-based regimens of whom only one in seven patients developed positive antibody response. Colchicine and immune-modulating drugs used in MS on the other hand did not seem to have a negative impact on the antibody response.
Our results are in line with the recent literature [19,20,21,22,23,24]. Reduced antibody titers and negative effect of DMARDs among patients with IMD were reported in the majority of the studies investigating mRNA vaccine response. It appears that, almost all DMARDs including Janus kinase inhibitors and methotrexate blunt humoral responses, while B cell depletion and glucocorticoids exert the strongest effect. On the other hand, Simon et al. suggested that the delayed and reduced antibody response was associated with inherent disease itself rather than the concomitant treatment [24]. Moreover, it appears that the situation is much worse for the solid organ transplant recipients [25,26,27]. An inadequate antibody response to mRNA SARS-CoV-2 vaccination was demonstrated in several patient populations [25,26,27].
Similar observations can be found in other inactivated vaccine studies such as those against influenza or hepatitis [28,29,30,31,32,33]. These vaccines could be able to induce significant humoral responses in patients with autoimmune diseases. However, the antibody response was observed to be blunted in those treated with methotrexate, azathioprine, mycophenolate mofetil or with CD20-depleting antibodies.
Immunosenescence, described as the impairment of both innate and adaptive immune functions in the elderly is rather well known [34,35,36]. It results in increased susceptibility to infectious diseases and a diminished vaccine effectiveness. Compared to younger individuals, in older people, the amount of post-vaccination antibody titers is significantly reduced. Moreover, the titers decline more rapidly. Comorbidities such as cardiovascular disease, diabetes, and obesity could also contribute to the lower humoral response in the elderly. It has to be noted that, age adjusted Charlson comorbidity index was also significantly higher in the elderly in our study.
It has to be reminded that the presence of antibodies does not explain whether they ensure protection from COVID-19. There is also a need to establish a threshold antibody titer for protective immunity.
This study has several limitations. We did not assess the neutralizing activity of the vaccinee serum against SARS-CoV-2 or determine T cell responses. We could not quantitate exact antibody titers of those with upper limit (> 250 U/mL) determined by the immunoassay. This led us to do categoric analysis of antibody titers, which may have resulted loss of data. Sample size of the patients with IMD and elderly was small. All patients and controls had a negative history for COVID-19, however, we did not test anti–SARS-CoV-2 antibodies on the day of the first vaccine injection. This is especially important as the great majority of the study population was composed of hospital workers. Nevertheless, it has to be said that the most of the hospital workers have been tested for COVID-19 more frequently than the general population throughout the pandemic. Finally, we did not assess formally side effects of the vaccine nor evaluated exacerbations after vaccination in patients with IMD or the elderly.
Conclusions
We found a robust antibody response to inactivated vaccine against SARS-CoV-2 among patients with IMD as well as immunocompetent controls. This was also true for the elderly. However, those who were using immunosuppressive treatment (particularly rituximab) and elderly had reduced antibody titers. Caution, close surveillance and perhaps earlier revaccination could be recommended for those using immunosuppressives and the elderly. Further studies should assess whether the low antibody titers are associated with diminished protection against COVID-19.
References
https://covid19.who.int/. Accessed 30 May 2021
https://www.aa.com.tr/en/turkey/turkey-grants-approval-for-chinese-covid-19-vaccine/2108980
https://www.genengnews.com/covid-19-candidates/covid-19-keeping-an-eye-on/sinovac-biotech/
Gao Q, Bao L, Mao H, Wang L, Xu K, Yang M, Li Y, Zhu L, Wang N, Lv Z, Gao H, Ge X, Kan B, Hu Y, Liu J, Cai F, Jiang D, Yin Y, Qin C, Li J, Gong X, Lou X, Shi W, Wu D, Zhang H, Zhu L, Deng W, Li Y, Lu J, Li C, Wang X, Yin W, Zhang Y, Qin C (2020) Development of an inactivated vaccine candidate for SARS-CoV-2. Science 369(6499):77–81. https://doi.org/10.1126/science.abc1932
Zhang Y, Zeng G, Pan H, Li C, Hu Y, Chu K, Han W, Chen Z, Tang R, Yin W, Chen X, Hu Y, Liu X, Jiang C, Li J, Yang M, Song Y, Wang X, Gao Q, Zhu F (2021) Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial. Lancet Infect Dis 21(2):181–192. https://doi.org/10.1016/S1473-3099(20)30843-4
Bueno SM, Abarca K, González PA, Gálvez NMS, Soto JA, Duarte LF, Schultz BM et al (2021) Interim report: safety and immunogenicity of an inactivated vaccine against SARS-CoV-2 in healthy chilean adults in a phase 3 clinical trial. medRxiv. https://doi.org/10.1101/2021.03.31.21254494
Curtis JR, Johnson SR, Anthony DD, Arasaratnam RJ, Baden LR, Bass AR, Calabrese C, Gravallese EM, Harpaz R, Kroger A, Sadun RE, Turner AS, Anderson Williams E, Mikuls TR (2021) American College of Rheumatology Guidance for COVID-19 vaccination in patients with rheumatic and musculoskeletal diseases-Version 1. Arthritis Rheumatol. https://doi.org/10.1002/art.41734
https://www.gov.uk/government/publications/covid-19-vaccination-care-home-and-healthcare-settings-posters/covid-19-vaccination-first-phase-priority-groups. Accessed 20 May 2021
https://www.cdc.gov/mmwr/volumes/69/wr/mm695152e2.htm. Accessed 20 May 2021
https://doh.gov.ph/Vaccines/when-will-the-COVID-19-available-to-me. Accessed 20 May 2021
Logunov DY, Dolzhikova IV, Shcheblyakov DV, Tukhvatulin AI, Zubkova OV, Dzharullaeva AS, Kovyrshina AV, Lubenets NL, Grousova DM, Erokhova AS, Botikov AG, Izhaeva FM, Popova O, Ozharovskaya TA, Esmagambetov IB, Favorskaya IA, Zrelkin DI, Voronina DV, Shcherbinin DN, Semikhin AS, Simakova YV, Tokarskaya EA, Egorova DA, Shmarov MM, Nikitenko NA, Gushchin VA, Smolyarchuk EA, Zyryanov SK, Borisevich SV, Naroditsky BS, Gintsburg AL, Gam-COVID-Vac Vaccine Trial Group (2021) Safety and efficacy of an rAd26 and rAd5 vector-based heterologous prime-boost COVID-19 vaccine: an interim analysis of a randomised controlled phase 3 trial in Russia. Lancet 397(10275):671–681. https://doi.org/10.1016/S0140-6736(21)00234-8
Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, Perez JL, Pérez Marc G, Moreira ED, Zerbini C, Bailey R, Swanson KA, Roychoudhury S, Koury K, Li P, Kalina WV, Cooper D, Frenck RW Jr, Hammitt LL, Türeci Ö, Nell H, Schaefer A, Ünal S, Tresnan DB, Mather S, Dormitzer PR, Şahin U, Jansen KU, Gruber WC, C4591001 Clinical Trial Group (2020) Safety and efficacy of the BNT162b2 mRNA Covid-19 Vaccine. N Engl J Med 383(27):2603–2615. https://doi.org/10.1056/NEJMoa2034577
Hyrich KL, Machado PM (2021) Rheumatic disease and COVID- 19: epidemiology and outcomes. Nat Rev Rheumatol 17(2):71–72. https://doi.org/10.1038/s41584-020-00562-2(PMID:33339986;PMCID:PMC7747184)
Felten R, Dubois M, Ugarte-Gil MF et al (2021) Vaccination against COVID-19: expectations and concerns of patients with autoimmune and rheumatic diseases. Lancet Rheumatol. 16. https://doi.org/10.1016/S2665-9913(21)00039-4. [Epub ahead of print: 22 Feb 2021]. http://www.ncbi.nlm.nih.gov/pubmed/33655219
Dorjee K, Kim H, Bonomo E, Dolma R (2020) Prevalence and predictors of death and severe disease in patients hospitalized due to COVID-19: a comprehensive systematic review and meta-analysis of 77 studies and 38,000 patients. PLoS ONE. https://doi.org/10.1371/journal.pone.0243191(PMID:33284825;PMCID:PMC7721151)
Dessai SB, Fasal R, Dipin J, Adarsh D, Balasubramanian S (2018) Age-adjusted charlson comorbidity index and 30-day morbidity in pelvic surgeries. South Asian J Cancer. 7(4):240–243. https://doi.org/10.4103/sajc.sajc_241_17 (PMID: 30430092; PMCID: PMC6190402)
Riester E, Findeisen P, Hegel JK, Kabesch M, Ambrosch A, Rank CM, Langen F, Laengin T, Niederhauser C (2021) Performance evaluation of the Roche Elecsys Anti-SARS-CoV-2 immunoassay. medRxiv. https://doi.org/10.1101/2021.03.02.21252203
Braun-Moscovici Y, Kaplan M, Markovits D, Giryes S, Toledano K, Tavor Y, Dolnikov K, Balbir-Gurman A (2021) Humoral response to Pfizer mRNA vaccine against SARS CoV2, in patients with autoimmune inflammatory rheumatic diseases and the impact on the rheumatic disease activity. medRxiv. https://doi.org/10.1101/2021.04.02.21254493
Haberman RH, Herati R, Simon D et al (2021) Methotrexate hampers immunogenicity to BNT162b2 mRNA COVID-19 vaccine in immune-mediated inflammatory disease. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2021-220597 (Published Online First)
Deepak P, Kim W, Paley MA, Yang M, Carvidi AB, El-Qunni AA, Haile A, Huang K, Kinnett B, Liebeskind MJ, Liu Z, McMorrow LE, Paez D, Perantie DC, Schriefer RE, Sides SE, Thapa M, Gergely M, Abushamma S, Klebert M, Mitchell L, Nix D, Graf J, Taylor KE, Chahin S, Ciorba MA, Katz P, Matloubian M, O’Halloran JA, Presti RM, Wu GF, Whelan SPJ, Buchser WJ, Gensler LS, Nakamura MC, Ellebedy AH, Kim AHJ (2021) Glucocorticoids and B Cell depleting agents substantially ımpair immunogenicity of mRNA vaccines to SARS-CoV-2. medRxiv [Preprint]. https://doi.org/10.1101/2021.04.05.21254656 (PMID: 33851176; PMCID: PMC8043473)
Park JK, Lee EB, Shin K, Sung YK, Kim TH, Kwon SR, Lee MS, Hong SJ, Choi BY, Lee SS, Back HJ, on behalf of the Korean College of Rheumatology Task Force for COVID-19 Vaccine Guidance for Patients with Autoimmune Inflammatory Rheumatic Diseases (2021) COVID-19 vaccination in patients with autoimmune inflammatory rheumatic diseases: clinical guidance of the korean college of rheumatology. J Korean Med Sci 36(12):e95. https://doi.org/10.3346/jkms.2021.36.e95
Geisen UM, Berner DK, Tran F et al (2021) Immunogenicity and safety of anti-SARS-CoV-2 mRNA vaccines in patients with chronic inflammatory conditions and immunosuppressive therapy in a monocentric cohort. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2021-220272 (Published Online First)
Simon D, Tascilar K, Fagni F, Krönke G, Kleyer A, Meder C, Atreya R, Leppkes M, Kremer AE, Ramming A, Pachowsky ML, Schuch F, Ronneberger M, Kleinert S, Hueber AJ, Manger K, Manger B, Berking C, Sticherling M, Neurath MF, Schett G (2021) SARS-CoV-2 vaccination responses in untreated, conventionally treated and anticytokine-treated patients with immune-mediated inflammatory diseases. Ann Rheum Dis. https://doi.org/10.1136/annrheumdis-2021-220461
Boyarsky BJ, Werbel WA, Avery RK et al (2021) Immunogenicity of a single dose of SARS-CoV-2 messenger RNA vaccine in solid organ transplant recipients. JAMA 325(17):1784–1786
Rabinowich L, Grupper A, Baruch R et al (2021) Low immunogenicity to SARS-CoV-2 vaccination among liver transplant recipients. J Hepatol. https://doi.org/10.1016/j.jhep.2021.04.020
Benotmane I, Gautier-Vargas G, Cognard N, Olagne J, Heibel F, Braun-Parvez L, Martzloff J, Perrin P, Moulin B, Fafi-Kremer S, Caillard S (2021) Weak anti-SARS-CoV-2 antibody response after the first injection of an mRNA COVID-19 vaccine in kidney transplant recipients. Kidney Int S0085–2538(21):00348–00353. https://doi.org/10.1016/j.kint.2021.03.014
Kostianovsky A, Charles P, Alves JF, Goulet M, Pagnoux C, Le Guern V, Mouthon L, Krivine A, Villiger P, Launay O, Guillevin L, French Vasculitis Study Group (2012) Immunogenicity and safety of seasonal and 2009 pandemic A/H1N1 influenza vaccines for patients with autoimmune diseases: a prospective, monocentre trial on 199 patients. Clin Exp Rheumatol 30(1 Suppl 70):S83–S89
Elkayam O, Yaron M, Caspi D (2002) Safety and efficacy of vaccination against hepatitis B in patients with rheumatoid arthritis. Ann Rheum Dis 61(7):623–625. https://doi.org/10.1136/ard.61.7.623
Erkek E, Ayaslioglu E, Erkek AB, Kurtipek GS, Bagci Y (2005) Response to vaccination against hepatitis B in patients with Behcet’s disease. J Gastroenterol Hepatol 20(10):1508–1511. https://doi.org/10.1111/j.1440-1746.2005.03903.x
McMahan ZH, Bingham CO 3rd (2014) Effects of biological and non-biological immunomodulatory therapies on the immunogenicity of vaccines in patients with rheumatic diseases. Arthritis Res Ther 16(6):506. https://doi.org/10.1186/s13075-014-0506-0
Bingham CO 3rd, Looney RJ, Deodhar A, Halsey N, Greenwald M, Codding C, Trzaskoma B, Martin F, Agarwal S, Kelman A (2010) Immunization responses in rheumatoid arthritis patients treated with rituximab: results from a controlled clinical trial. Arthritis Rheum 62(1):64–74. https://doi.org/10.1002/art.25034
Park JK, Lee YJ, Shin K et al (2018) Impact of temporary methotrexate discontinuation for 2 weeks on immunogenicity of seasonal influenza vaccination in patients with rheumatoid arthritis: a randomised clinical trial. Ann Rheum Dis 77:898–904
Stiasny K, Aberle JH, Keller M, Grubeck-Loebenstein B, Heinz FX (2012) Age affects quantity but not quality of antibody responses after vaccination with an inactivated flavivirus vaccine against tick-borne encephalitis. PLoS ONE 7(3):34145. https://doi.org/10.1371/journal.pone.0034145 (Epub 2012 Mar 26. PMID: 22461903; PMCID: PMC3312914)
Crooke SN, Ovsyannikova IG, Poland GA, Kennedy RB (2019) Immunosenescence and human vaccine immune responses. Immun Ageing 13(16):25. https://doi.org/10.1186/s12979-019-0164-9.PMID:31528180;PMCID:PMC6743147
Müller L, Andrée M, Moskorz W, Drexler I, Walotka L, Grothmann R, Ptok J, Hillebrandt J, Ritchie A, Rabl D, Ostermann PN, Robitzsch R, Hauka S, Walker A, Menne C, Grutza R, Timm J, Adams O, Schaal H (2021) Age-dependent immune response to the Biontech/Pfizer BNT162b2 COVID-19 vaccination. Clin Infect Dis 27:ciab381. https://doi.org/10.1093/cid/ciab381
Acknowledgements
We would like to thank our students, nurses and staff who contributed to the work including Dilsen Cevirgen, Havva Yazici, Salih Erkan, Bulent Ahmet Kart, Serife Altunbay, Damla Erdas, Merve Karayigit, Betul Ozcaglayan, Alphan Yesilbag, Busra Mirzamli, Kerem Yusuf Kanoglu, Utku Yıldırım, Zühre Tarlak, Aysegul Hamza, Furkan Sevim, Hayriye Koseleci, Filiz Comert, Ayten Dag and Sukran Yuksel Findikoglu.
Funding
This study was supported by the Turkish Rheumatology Society (26.03.2021/5).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by GB, MO, MAK, YT, NS, GO, AO, IIB, NS, FT, VH, ES. The first draft of the manuscript was written by ES and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Guldaran Bakhdiyarli, MD, Mert Oztas, MD, Mert Ahmet Kuskucu, MD, Yesim Tok, MD, Necdet Sut, PhD, Guzin Ozcifci, Ms, Ali Ozcaglayan, Mr, Ilker Inanc Balkan, MD, Nese Saltoglu, MD, Fehmi Tabak, MD, Vedat Hamuryudan, MD, Emire Seyahi, MD, declare that they have no conflict of interest.
Ethical statement
The study protocol was approved by the Ministry of Health (2021-01-25T15_35_02) and by the Ethics Committee of Istanbul University-Cerrahpasa, Cerrahpasa Medical Faculty (10.03.2021-49051). Written informed consent was obtained from each enrolled participant. All study procedures were carried out in accordance with the ethical standards of the Helsinki Declaration.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Seyahi, E., Bakhdiyarli, G., Oztas, M. et al. Antibody response to inactivated COVID-19 vaccine (CoronaVac) in immune-mediated diseases: a controlled study among hospital workers and elderly. Rheumatol Int 41, 1429–1440 (2021). https://doi.org/10.1007/s00296-021-04910-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-021-04910-7