Abstract
Key message
We cloned a dehydrins gene CaDHN1 from pepper and the expression of CaDHN1 was markedly upregulated by cold, salt, osmotic stresses and salicylic acid (SA) treatment.
Abstract
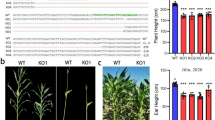
Dehydrins (DHNs) are a subfamily of group 2 late embryogenesis-abundant (LEA) proteins that are thought to play an important role in enhancing abiotic stress tolerance in plants. In this study, a DHN EST (Expressed Sequence Tag) was obtained from 6 to 8 true leaves seedlings of pepper cv P70 (Capsicum annuum L.) by our laboratory. However, the DHN gene in pepper was not well characterized. According to this EST sequence, we isolated a DHN gene, designated as CaDHN1, and investigated the response and expression of this gene under various stresses. Our results indicated that CaDHN1 has the DHN-specific and conserved K- and S- domain and encodes 219 amino acids. Phylogenetic analysis showed that CaDHN1 belonged to the SKn subgroup. Tissue expression profile analysis revealed that CaDH N1 was expressed predominantly in fruits and flowers. The expression of CaDHN1 was markedly upregulated in response to cold, salt, osmotic stresses and salicylic acid (SA) treatment, but no significant change by abscisic acid (ABA) and heavy metals treatment. Loss of function of CaDHN1 using the virus-induced gene silencing (VIGS) technique led to decreased tolerance to cold-, salt- and osmotic-induced stresses. Overall, these results suggest that CaDHN1 plays an important role in regulating the abiotic stress resistance in pepper plants.







Similar content being viewed by others
References
Allagulova CR, Gimalov FR, Shakirova FM, Vakhitov VA (2003) The plant dehydrins: structure and putative functions. Biochemistry 68(9):945–951
Amara I, Odena A, Oliveira E, Moreno A, Masmoudi K, Pages M, Goday A (2012) Insights into maize LEA proteins: from proteomics to functional approaches. Plant Cell Physiol 53:312–329
Arkus KAJ, Cahoon EB, Jez JM (2005) Mechanistic analysis of wheat chlorophyllase. Arch Biochem Biophys 438:146–155
Artlip TS, Callahan AM, Bassett CL, Wisniewski ME (1997) Seasonal expression of a dehydrin gene in sibling deciduous and evergreen genotypes of peach (Prunus persica). Plant Mol Biol 33(1):61–70
Borovskii GB, Stupnikova IV, Antipina AI, Vladimirova SV, Voinikov VK (2002) Accumulation of dehydrin-like proteins in the mitochondria of cereals in response to cold, freezing, drought and ABA treatment. BMC Plant Biol. doi:10.1186/1471-2229-2-5
Brini F, Hanin M, Lumbreras V, Amara I, Khoudi H, Hassairi A, Pages M, Masmoudi K (2007) Overexpression of wheat dehydrin DHN-5 enhances tolerance to salt and osmotic stress in Arabidopsis thaliana. Plant Cell Rep 26:2017–2026
Campbell SA, Close TJ (1997) Dehydrins: genes, proteins, and associations with phenotypic traits. New Phytol 137:61–74
Chen RG, Guo WL, Yin YX, Gong ZH (2014) A novel F-Box protein CaF-Box is involved in responses to plant hormones and abiotic stress in pepper (Capsicum annuum L.). Int J Mol Sci 15:2413–2430
Chini A, Grant JJ, Seki M, Shinozaki K, Loake GJ (2004) Drought tolerance established by enhanced expression of the CC-NBS-LRR gene, ADR1, requires salicylic acid, EDS1 and ABI1. Plant J 38:810–822
Choi DW, Close TJ (2000) A newly identified barley gene, Dhn12, encoding an YSK2 DHN, is located on chromosome 6H and has embryo-specific expression. Theor Appl Genet 100:1274–1278
Choudhury S, Panda P, Sahoo L, Panda SK (2013) Reactive oxygen species signalingin plants under abiotic stress. Plant Signal Behav 8(4):e23681. doi:10.4161/psb.23681
Chung E, Kim SY, Yi SY, Choi D (2003) Capsicum annuum dehydrin, an osmotic-stress gene in hot pepper plants. Mol Cells 15(3):327–332
Chung E, Seong E, Kim YC, Chung EJ, Oh SK, Lee S, Park JM, Joung YH, Choi D (2004) A method of high frequency virus-induced gene silencing in chili pepper (Capsicum annuum L.cv.Bukang). Mol Cells 17:377–380
Close TJ (1997) Dehydrins: a commonalty in the response of plants to dehydration and low temperature. Physiol Plant 100:291–296
Cutler SR, Rodriguez PL, Finkelstein RR, Abrams SR (2010) Abscisic acid: emergence of a core signaling network. Annu Rev Plant Biol 61:651–679
Dhanaraj AL, Slovin JP, Rowland LJ (2005) Isolation of a cDNA clone and characterization of expression of the highly abundant, cold acclimation-associated 14 kDa dehydrin of blueberry. Plant Sci 168:949–957
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot 32:93–101
Dionisio-Sese ML, Tobita S (1998) Antioxidant responses of rice seedlings to salinity stress. Plant Sci 135:1–9
Fujita Y, Fujita M, Shinozaki K, Yamaguchi-Shinozaki K (2011) ABA-mediated transcriptional regulation in response to osmotic stress in plants. J Plant Res 124:509–525
Guo WL, Chen RG, Gong ZH, Yin YX, Li DW (2013) Suppression subtractive hybridization analysis of genes regulated by application of exogenous abscisic acid in pepper plant (Capsicum annuum L.) leaves under chilling stress. PLoS ONE 8(6):e66667. doi:10.1371/journal.pone.0066667
Hara M, Terashima S, Fukaya T, Kuboi T (2003) Enhancement of cold tolerance and inhibition of lipid peroxidation by citrus dehydrin in transgenic tobacco. Planta 217:290–298
Hara M, Fujinaga M, Kuboi T (2004) Radical scavenging activity and oxidative modification of citrus dehydrin. Plant Physiol Biochem 42:657–662
Hara M, Fujinaga M, Kuboi T (2005) Metal binding by citrus dehydrin with histidine-rich domains. J Exp Bot 56:2695–2703
Hirayama T, Shinozaki K (2010) Research on plant abiotic stress responses in the post-genome era: past, present and future. Plant J 61:1041–1052
Koag MC, Fenton RD, Wilkens S, Close TJ (2003) The binding of maize DHN1 to lipid vesicles. Gain of structure and lipid specificity. Plant Physiol 131:309–316
Kosova K, Vitamvas P, Prasil IT (2007) The role of dehydrins in plant response to cold. Biol Plantarum 51:601–617
Kovacs D, Agoston B, Tompa P (2008) Disordered plant LEA proteins as molecular chaperones. Plant Signal Behav 3:710–713
Kurkela S, Franck M (1990) Cloning and characterization of a cold- and ABA-inducible Arabidopsis gene. Plant Mol Biol 15:137–144
Li ZG, Zhao LX, Kai GY, Yu SW, Cao YF, Pang YZ, Sun XF, Tang KX (2004) Cloning and expression analysis of a water stress-induced gene from Brassica oleracea. Plant Physiol Biochem 42:789–794
Li Q, Yu B, Gao Y, Dai AH (2011) Cinnamic acid pretreatment mitigates chilling stress of cucumber leaves through altering antioxidant enzyme activity. Plant Physiol 168:927–934
Liu Y, Schiff M, Dinesh-Kumar SP (2002) Virus-induced gene silencing in tomato. Plant J 31:777–786
Liu H, Yu CY, Li HX, Ouyang B, Wang TT, Zhang JH, Wang X, Ye ZB (2015) Overexpression of ShDHN, a dehydrin gene from Solanum habrochaites enhances tolerance to multiple abiotic stresses in tomato. Plant Sci 231:198–211
Nguyen HT, Leipner J, Stamp P, Guerra-Peraza O (2009) Low temperature stress in maize (Zea mays L.) induces genes involved in photosynthesis and signal transduction as studied by suppression subtractive hybridization. Plant Physiol Biochem 47:116–122
Ochoa-Alfaro AE, Rodrıguez-Kessler M, Perez-Morales M, Delgado-Sanchez BP, Cuevas-Velazquez CL, Gomez-Anduro G, Jimenez-Bremont JF (2012) Functional characterization of an acidic SK3 dehydrin isolated from an Opuntia streptacantha cDNA library. Planta 235:565–578
Qiu HL, Zhang LH, Liu C, Li H, Wang AY, Liu HH, Zhu JB (2014) Cloning and characterization of a novel dehydrin gene, SiDhn2, from Saussurea involucrata Kar. et Kir. Plant Mol Biol 84:707–718
Rorat T (2006) Plant dehydrins-tissue location, structure and function. Cell Mol Biol Lett 11:536–556
Rorat T, Szabala BM, Grygorowicz WJ, Wojtowicz B, Yin Z, Rey P (2006) Expression of SK3-type dehydrin in transporting organs is associated with cold acclimation in Solanum species. Planta 224:205–221
Ruibal C, Salamó IP, Carballo V, Castro A, Bentancor M, Borsani O, Szabados L, Vidal S (2012) Differential contribution of individual dehydrin genes from Physcomitrella patens to salt and osmotic stress tolerance. Plant Sci 190:89–102
Santos AB, Mazzafera P (2012) Dehydrins are highly expressed in water-stressed plants of two coffee species. Tropical Plant Biol 5:218–232
Sun X, Lin HH (2010) Role of plant dehydrins in antioxidation mechanisms. Biologia 65:755–759
Svenson J, Ismail AM, Palva ET, Close TJ (2002) Dehydrins. In: Storey KB, Storey JM (eds) Sensing, signalling and cell adaptation. Elsevier Science, BV, pp 155–171
Szabala BM, Fudali S, Rorat T (2014) Accumulation of acidic SK3 dehydrins in phloem cells of cold- and drought-stressed plants of the Solanaceae. Planta 239:847–863
Thomashow MF (1999) Plant cold acclimation: freezing tolerance genes and regulatory mechanisms. Annu Rev Plant Physiol Plant Mol Biol 50:571–599
Wan HJ, Yuan W, Ruan M, Ye Q, Wang R, Li Z, Zhou G, Yao Z, Zhao J, Liu S, Yang Y (2011) Identification of reference genes for reverse transcription quantitative real-time PCR normalization in pepper (Capsicum annuum L.). Biochem Biophy Res Co 416:24–30
Wang JE, Liu KK, Li DW, Zhang YL, Zhao Q, He YM, Gong ZH (2013) A novel peroxidase CanPOD gene of pepper is involved in defense responses to Phytophthora capsici infection as well as abiotic stress tolerance. Int J Mol Sci 14:3158–3177
Welin BV, Olson A, Nylander M, Palva ET (1994) Characterization and differential expression of dhn/lea/rab-like genes during cold acclimation and drought stress in Arabidopsis thaliana. Plant Mol Biol 26:131–144
Wisniewski ME, Bassett CL, Renaut J, Farrell R, Tworkoski T, Artlip TS (2006) Differential regulation of two dehydrin genes from peach (Prunus persica) by photoperiod, low temperature and water deficit. Tree Physiol 26:575–584
Xu J, Zhang YX, Wei W, Han L, Guan ZQ, Wang Z, Chai TY (2008) BjDHNs confer heavy-metal tolerance in plants. Mol Biotechnol 38:91–98
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance, to dehydration and cold stresses. Annu Rev Plant Biol 57:781–803
Zhang YX, Li JM, Yu F, Cong L, Wang LY, Burkard G, Chai TY (2006) Cloning and expression analysis of SKn-type dehydrin gene from bean in response to heavy metals. Mol Biotechnol 32:205–218
Acknowledgments
This work was supported through funding from the National Natural Science Foundation of China (#31201615, #31272163), Jiangsu Agriculture Science and Technology Innovation Fund [CX(12)1004], the Natural Science Foundation of Shaanxi Province (#2011JQ3010), the Shaanxi Provincial Science and Technology Coordinating Innovative Engineering Project (#2012KTCL02-09), and the Northwest A&F University Cyrus Tang Seed Development Fund.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by A. Dhingra.
Ru-gang Chen, Hua Jing, Wei-li Guo and Shu-Bin Wang are contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, Rg., Jing, H., Guo, Wl. et al. Silencing of dehydrin CaDHN1 diminishes tolerance to multiple abiotic stresses in Capsicum annuum L.. Plant Cell Rep 34, 2189–2200 (2015). https://doi.org/10.1007/s00299-015-1862-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-015-1862-1