Abstract
Purpose
Urethral strictures are a common disease of the lower urinary tract in men. At present, the use of buccal mucosa is the method of choice for long or recurrent strictures. However, autologous tissue-engineered grafts are still under investigation for reconstructive urological surgery. The aim of this pilot study was to evaluate the use of human urothelial cells (HUC) seeded on bovine collagen type I-based cell carriers (CCC) in an animal model and to evaluate short-term outcome of the surgical procedure.
Methods
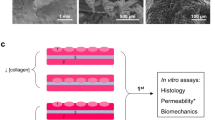
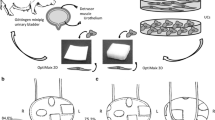
Four male Göttingen minipigs were used with immunosuppression (cyclosporine A) for this pilot xenograft study. HUC obtained from human benign ureteral tissue were stained by PKH26 and seeded on a collagen cell carrier (CCC). Seven weeks after urethral stricture induction and protective vesicostomy, cell-seeded CCC was implanted in the urethra with HUC luminal and antiluminal, respectively. After two weeks animals were euthanized, urethrography and histological assessment were performed.
Results
Surgery was technically feasible in all minipigs. Stricture was radiologically established 7 weeks after induction. CCC was visible after two weeks and showed good integration without signs of inflammation or rejection. In the final urethrography, no remaining stricture could be detected. Near porcine urothelium, PKH26-positive areas were found even if partially detached from CCC. Although diminished, immunofluorescence with pankeratin, CK20, E-cadherin and ZO-1 showed intact urothelium in several areas on and nearby CCC.
Conclusion
Finally, this study demonstrates that the HUC-seeded CCC used as a xenograft in minipigs is technically feasible and shows promising results for further studies.




Similar content being viewed by others
References
Barbagli G, Palminteri E, Lazzeri M, Guazzoni G (2003) Anterior urethral strictures. BJU Int 92(5):497–505
Gozzi C, Tritschler S, Bastian PJ, Stief CG (2008) Management of urethral strictures. Der Urologe Ausg A 47(12):1615–1622. doi:10.1007/s00120-008-1903-2
Rodder K, Olianas R, Fisch M (2006) [Urethral strictures–operative strategy]. Der Urologe Ausg A 45 (4):499–511; quiz 512–493. doi:10.1007/s00120-006-1031-9
Engel O, Fisch M (2010) Urethral reconstruction after failed primary surgery. Der Urologe Ausg A 49(7):822–826. doi:10.1007/s00120-010-2315-7
Bhargava S, Chapple CR (2004) Buccal mucosal urethroplasty: is it the new gold standard? BJU Int 93(9):1191–1193. doi:10.1111/j.1464-410X.2003.04860.x
Sinha RJ, Singh V, Sankhwar SN, Dalela D (2009) Donor site morbidity in oral mucosa graft urethroplasty: implications of tobacco consumption. BMC Urol 9:15. doi:10.1186/1471-2490-9-15
Mangera A, Chapple CR (2013) Tissue engineering in urethral reconstruction: an update. Asian J Androl 15(1):89–92. doi:10.1038/aja.2012.91
Idzenga T, Pel JJ, van Mastrigt R (2006) A biophysical model of the male urethra: comparing viscoelastic properties of polyvinyl alcohol urethras to male pig urethras. Neurourol Urodyn 25(5):451–460. doi:10.1002/nau.20282
Seibold J, Selent C, Feil G, Wiedemann J, Colleselli D, Mundhenk J, Gakis G, Sievert KD, Schwentner C, Stenzl A (2012) Development of a porcine animal model for urethral stricture repair using autologous urothelial cells. J Pediatr Urol 8(2):194–200. doi:10.1016/j.jpurol.2011.02.001
Sievert KD, Selent-Stier C, Wiedemann J, Greiner TO, Amend B, Stenzl A, Feil G, Seibold J (2012) Introducing a large animal model to create urethral stricture similar to human stricture disease: a comparative experimental microscopic study. J Urol 187(3):1101–1109. doi:10.1016/j.juro.2011.10.132
Feil G, Christ-Adler M, Maurer S, Corvin S, Rennekampff HO, Krug J, Hennenlotter J, Kuehs U, Stenzl A, Sievert KD (2006) Investigations of urothelial cells seeded on commercially available small intestine submucosa. Eur Urol 50(6):1330–1337. doi:10.1016/j.eururo.2006.05.041
Hennig C, Theuring F (1990) Physiologic regeneration of rat urothelium after pulse and continual labeling with 3H-thymidine at various ages. Acta Histochem Suppl 39:263–266
Bouhout S, Chabaud S, Bolduc S (2016) Organ-specific matrix self-assembled by mesenchymal cells improves the normal urothelial differentiation in vitro. World J Urol 34(1):121–130. doi:10.1007/s00345-015-1596-2
Zhu L, Fang Y, Liu Z, Wang P, Wang Y, Xu H (2008) Rabbit anti-human leukocyte polyclonal antibody inhibits xenogeneic cell-mediated immune responses. Transpl Proc 40(8):2760–2763. doi:10.1016/j.transproceed.2008.07.111
Katnik-Prastowska I, Lis J, Matejuk A (2014) Glycosylation of uroplakins: implications for bladder physiopathology. Glycoconj J 31(9):623–636. doi:10.1007/s10719-014-9564-4
Nagele U, Maurer S, Feil G, Bock C, Krug J, Sievert KD, Stenzl A (2008) In vitro investigations of tissue-engineered multilayered urothelium established from bladder washings. Eur Urol 54(6):1414–1422. doi:10.1016/j.eururo.2008.01.072
Raya-Rivera A, Esquiliano DR, Yoo JJ, Lopez-Bayghen E, Soker S, Atala A (2011) Tissue-engineered autologous urethras for patients who need reconstruction: an observational study. Lancet 377(9772):1175–1182. doi:10.1016/S0140-6736(10)62354-9
Acknowledgements
The study was supported by Viscofan Bioengineering, a business unit of Naturin Viscofan GmbH, Weinheim, Germany. We want to thank Prof. Dr. Wilhelm K. Aicher for his help in evaluation of experimental data.
Authors’ contribution
SA analysed the data, wrote and edited the manuscript. MV was involved in protocol/project development, data collection or management. AK analysed the data. SM was involved in data collection or management, data analysis. LG, JM and LD contributed to data collection or management. SB developed the protocol/project, analysed the data and edited the manuscript. AS and K-DS were involved in protocol/project development, manuscript writing/editing. BA was involved in data collection or management, data analysis, manuscript writing/editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. (Institutional ethics committee approval no. 379/2010BO2). All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Aufderklamm, S., Vaegler, M., Kelp, A. et al. Collagen cell carriers seeded with human urothelial cells for urethral reconstructive surgery: first results in a xenograft minipig model. World J Urol 35, 1125–1132 (2017). https://doi.org/10.1007/s00345-016-1959-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-016-1959-3