Abstract
Purpose
The treatment of gastroschisis (GS) using our collaborative clinical pathway, with immediate attempted abdominal closure and bowel irrigation with a mucolytic agent, was reviewed.
Methods
A retrospective review of the past 20 years of our clinical pathway was performed on neonates with GS repair at our institution. The clinical treatment includes attempted complete reduction of GS defect within 2 h of birth. In the operating room, the bowel is evaluated and irrigated with mucolytic agent to evacuate the meconium and decompress the bowel. No incision is made and a neo-umbilicus is created. Clinical outcomes following closure were assessed.
Results
150 babies with gastroschisis were reviewed: 109 (77%) with a primary repair, 33 (23%) with a spring-loaded silo repair. 8 babies had a delayed closure and were not included in the statistical analysis. Successful primary repair and time to closure had a significant relationship with all outcome variables—time to extubation, days to initiate feeds, days to full feeds, and length of stay.
Conclusion
Early definitive closure of the abdominal defect with mucolytic bowel irrigation shortens time to first feeds, total TPN use, time to extubation, and length of stay.
Similar content being viewed by others
Background
The prevalence of gastroschisis has increased from an overall rate of 3.6 per 10,000 births during 1995–2005 to 4.9 per 10,000 births during 2006–2012 [1]. Yet, there is a lack of consensus about the optimal course of medical and surgical management for mothers and neonates with gastroschisis. From the surgeon’s standpoint, the anatomical defect is relatively simple to repair. But the effect on intestinal length and function is unpredictable. Thus, management is complicated and requires determination of the proper obstetrical and surgical approach and maintenance of nutrition.
Management approaches have evolved over time but the goal has essentially remained the same: reduce the viscera safely and close the defect with a cosmetically acceptable neo-umbilicus. The literature remains divided on a number of factors related to gastroschisis including optimal gestational age (timing of delivery), mode of delivery and utilization of a silo versus primary repair. Some studies have shown gastroschisis managed with a silo and delayed closure increased the neonate’s time on the ventilator, time to initiate enteral feeding, time to full enteral feeding, length of hospital stay, and hospital charges [2,3,4]. Conversely, similar retrospective studies have demonstrated silo placement improved outcomes [5,6,7,8,9] while still other studies have demonstrated no difference of outcomes between the repair techniques [10, 11]. Not surprisingly, studies conflict with each other on optimal timing of delivery [12,13,14,15,16,17,18,19]. The disagreement among published results warrants the need for comparative analysis of different standardized protocols of management to offer more definitive insights into the factors contributing to improved outcomes for neonates with gastroschisis.
With the divergence of results as discussed above, our goal was to standardize the care of the neonates as much as possible to minimize the variability in their care. To accomplish this we agreed upon the following protocol: (1) multi-specialty prenatal counseling, (2) monthly follow-up ultrasounds, (3) suggested cesarean delivery at 36–38 weeks, (4) gastroschisis repair scheduled for < 2 h post-delivery, (5) irrigating and decompressing the bowel with 5% Mucomyst, (6) bowel reduction and attempted primary repair via native defect, (7) measure urinary bladder pressure during closure, (8) creation of an neo-umbilicus with the native umbilical cord stump, (9) and monitor bladder pressure in acute postoperative period. All the steps were agreed upon and implemented prospectively at the beginning of the study in 1998 and have been used continuously by the perinatologists, neonatologists, and pediatric surgeons. The primary outcome was the total number of days on parenteral nutrition representing the time to full enteral feeds. The secondary outcomes were hospital length of stay and time on mechanical ventilation. We hypothesized earlier definitive closure of the gastroschisis defect utilizing the mucolytic irrigation of the meconium leads to earlier progression to enteral feeding and a shorter hospital length of stay.
Methods
Study design
An Institutional Review Board (IRB #0000068) approved retrospective chart review of all neonates that underwent gastroschisis (GS) repair between September 1998 and November 2017 at our institution was performed. All babies born with gastroschisis and treated at our institution were included whether born in our institution or elsewhere.
Clinical pathway
Babies born with gastroschisis were received by a neonatologist and taken to the NICU. Concurrently, a pediatric surgeon examined the neonate. The operating room was scheduled and waiting to ensure an attempt at primary repair was made within 2 h of birth. Initial management included rapid physical assessment, placement of the bowel in the midline to prevent venous compromise and distension, placement of a nasogastric tube to low intermittent suction, IV fluid resuscitation, and intravenous administration of ampicillin and gentamicin.
Neonates were then transported to the operating room. Nine board certified pediatric surgeons performed the surgeries in the series, ranging from 1 to 69 operations each. All surgeons had performed gastroschisis repair prior to this study, but except for the senior surgeon, none had previously implemented this technique and therefore received OR mentoring for standardization of the protocol.
Under general anesthesia in the operating room, the colon was irrigated with 5% n-acetyl cysteine (Mucomyst) diluted in warm normal saline to evacuate meconium and ensure that there were no bowel atresias before primary repair was attempted via the native GS defect. A Foley catheter was placed into the urinary bladder and connected to a pressure transducer to measure intra-cystic pressures as a surrogate of intra-abdominal pressures. Bladder pressures were maintained at less than 20 mmHg throughout closure of the defect. Ventilator settings used to maintain satisfactory oxygenation and ventilation were noted. Tidal volume achieved and end-tidal carbon dioxide with a set peak inspiratory pressure were followed throughout the course of the operation. The bowel was then reduced through the primary defect without extension by incision. The fascia was separated from the skin and closure performed in layers. The umbilical cord stump was utilized to create a neo-umbilicus to allow for a cosmetically appealing result, which was anatomically correct. Primary repair (PR) was defined as complete abdominal reduction with closure.
Attempted primary repair was defined as any record of the pediatric surgeon attempting reduction of the viscera into the abdomen. Primary repair was not always successful due to thickened or unpliable bowel, elevated ventilator pressures, or elevated abdominal pressure (bladder pressure > 20 mmHg). In those cases, at the attending surgeon’s discretion, the defect was partially closed and then fully reduced within 24–48 h. If an attempt at repair was not made, the surgeon documented reasons for a spring-loaded silo placement. This was done most frequently in situations of thick-walled non-compliant bowel or complicated GS, with intestinal atresia, stenosis, and/or perforation.
Post-operatively, IV sedation ± muscle paralysis was maintained for 12–48 h and bladder pressures were checked every 4 h for 24 h and maintained below 20 mmHg. Early in the protocol, paralysis was maintained routinely. However, as our data accumulated, we weaned and stopped paralysis much earlier post-operatively using decreasing bladder pressures as an indicator of safety. The Foley catheter was removed when bladder pressure monitoring was no longer needed. Neonates were extubated when deemed clinically ready using standard weaning parameters. NG suction was maintained until return of bowel function. Parenteral nutrition was provided until adequate enteral nutrition was tolerated.
Data collection and analysis
Demographic data for the babies with gastroschisis was collected, including indication for delivery, gender, birth weight, and gestational age. Variables of immediate management included time to the operating room, surgeon evaluation, and type of repair. Outcomes measured were feeding status, ventilator management, and length of hospital stay, complications, and mortality.
All statistical analysis was performed using the open source statistical program R (version 3.4.1) against a two-sided alternative hypothesis with a significance level of 5% (p = 0.05). Data were assessed for normality. Parametric variables are reported as mean ± standard deviation. The data tended to be right skewed with most observations grouped towards the lower end of the distribution with some going into a higher number of days. This led to the use of non-parametric Wilcoxon rank sum tests to compare groups and the group median.
Results
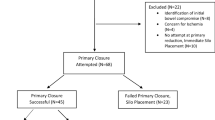
150 babies with gastroschisis were reviewed; eight patients (5%) had a delayed closure without a silo. These eight patients were partially reduced and had patch or Tegaderm coverage for several days and then returned to the operating room (OR) for definitive closure. They were neither primary nor silo repair and, therefore, were not included in either group for further analysis. Of the remaining 142 patients, 53% were males, the mean birth weight was 2348 ± 490 g, and the median time from birth to OR was 1.8 h. 14 (10%) had complicated gastroschisis (Table 1). Primary repair was achieved in 109 (77%) patients and 33 (23%) had silo placement. The mean gestational age for primary and silo repair was 35.33 weeks and 35.08 weeks, respectively; the difference was not significant. Comparing primary repair and silo placement, the dependent variables of this study were days to initiate feeding, days to enteral feeds, days to extubation, and days to discharge. Babies who underwent a primary repair of the GS defect without any comorbidities (subtract 12 patients, Table 1) had significantly better outcomes for all dependent variables (Table 2).
Of all the neonates taken to the OR for attempted primary repair, 66% were taken to the OR in less than 2 h and all in less than 6 h. The median time to the OR was 1.8 h. Regression analysis indicated a significant relationship between the number of hours to closure and overall length of stay (p < 0.01). For each 1-h increase in time to closure, there is a documented 5.5% increase in length of stay. A graph of the estimated LOS based on time to closure in hours for primary repair is shown in Fig. 1. Furthermore, there was a significant negative relationship between time to closure in hours and all outcome variables including length of stay, days on ventilator, days to initiate feeds, and days to full feeds. Comparison data for other contemporary reported gastroschisis outcomes and our data are presented in Table 3.
Discussion
Gastroschisis is a neonatal condition associated with prolonged hospitalization, need for parenteral nutritional support, and mechanical ventilation. Even in patients with excellent outcomes there is a disproportionate utilization of resources consumed by neonates with gastroschisis when compared to other Neonatal Intensive Care Unit patient groups [20]. Numerous retrospective studies have been performed to try to analyze the relationship between mode of delivery, gestational age at delivery, and neonatal outcomes in babies born with gastroschisis [12,13,14,15,16,17,18,19]. However, these have proven to be difficult questions to answer due to the interdependent nature of all these clinical variables. We created a standardized protocol for management controlled gestational age, spontaneous labor, delivery mode, time to closure, primary repair, intra-abdominal pressure, and relaxation in first 24 h post-operatively. By eliminating much of this variability we were able to better understand factors which might contribute to improved outcomes. Factors most suggestive of contributing to our excellent outcomes are: high rate of attempted primary repair, mucolytic bowel irrigation, and safely expediting time to closure in the OR. Our data suggest that this is what caused the markedly decreased time to first feeds, TPN use, time to extubation, and hospital length of stay in our study as compared to previously reported series [21,22,23,24,25].
Our patients had an attempted primary repair rate of 95%, with success 77% of the time, 66% of these patients in the OR in less than 2 h, and all patients in the OR in less than 6 h. Our results showed successful primary repair and time to closure having a significant linear relationship with all outcomes measured. Other recent studies have also showed improvement in outcomes with less time to closure [21,22,23, 26,24,25,]. Of note, we demonstrated a direct relationship between time to the OR and LOS with every hour past 2 h adding 5.5% of time to the LOS in days. This is the first study that has documented that expediting these babies to the operating room could impact the LOS in such a direct fashion. Additionally, our patients had markedly shorter time to enteral feeding, time to extubation, and overall length of stay when compared to all other recently published reports. Other similar contemporary works reported lengths of stay of 36.5–55.6 days for primary repairs and 44.5–74.4 for silo repairs, 1.5–3 times greater than our patient cohort’s lengths of stay (Table 3) [21, 22]. We attribute our high primary repair rate to a disciplined adherence to our multidisciplinary protocol and intraoperative management techniques.
One of these intraoperative techniques we innovated was the use of Mucomyst for bowel irrigation and evacuation. For decades, Mucomyst, alongside Gastrografin, has become the treatment of choice for the simple meconium ileus [27]. It works by reducing the viscosity of the meconium allowing for more thorough evacuation [28]. For each patient in our series, the Mucomyst was diluted to approximately 5% concentration with warm saline and the colon was thoroughly irrigated and manually evacuated. This resulted in significantly decompressed bowel, which we believe contributed to the high success rate of primary closure. Furthermore, the irrigation allowed us to inspect for any bowel atresias that may have been present.
Development of abdominal compartment syndrome (ACS) is a related concern with primary closure. Other studies have suggested increased complication rates associated with primary repair due to tight abdominal closure [3, 8]. In our cohort, no patients developed ACS. As outlined in the methods, peak airway pressures, tidal volume, and bladder pressures were all closely monitored and controlled in both the intra-operative and post-operative settings to prevent this complication. Placement of a bladder catheter for both intra-operative and early post-operative monitoring of bladder pressure clearly was critical to this result. Again, it was part of the protocol and contributed to the high primary closure rate being done safely.
The financial implications of reduced LOS nationally are substantial. Available data on national daily NICU costs average $3000 [30]. A financial review of the three large contemporary series we evaluated revealed their increased LOS of primary repair patients created multiple millions of dollars of increased estimated charges to the health care system. Different techniques for gastroschisis closure such as the suture-less flap closure have proposed cost savings due to lack of operating room needs [31]. However, a large meta-analysis study showed no significant difference in cost for flap versus fascial closure, and in fact, there was an increased umbilical hernia rate for flap closure with associated second operation and additional cost. Length of stay as well was not seen to be shorter [31]. We show the estimated savings of our series versus other recent contemporary reports calculated at Table 3.
This study is not without its limitations. Inherent limits exist due to the retrospective, non-randomized design of the study and the lack of a control group. Yang et al. posited it is spontaneous onset of labor, rather than route of delivery, that is associated with lower gestational age, lower birth weight, lower rate of primary closure, and longer LOS [15]. To better understand the role of timing and mode of delivery, a randomized controlled study is warranted [32]. An upcoming prospective randomized trial, the “The Gastroschisis Outcome of Delivery (GOOD) Study,” will evaluate mothers with fetal gastroschisis with a controlled delivery between 36 0/7–36 6/7 weeks and should offer more definitive guidelines on delivery method and timing [33] and may validate our data on timing and method.
A related factor is the reliance on surgeon judgment to dictate the treatment method and/or their comfort on the learning curve with the protocol. Patients in the silo group were categorized accordingly due to lack of documentation of attempted reduction. Although some patients were placed in this group due to the surgeon’s assessment of the appearance of the bowel, others were placed in the silo due to the surgeon’s lack of comfort with the protocol initially.
Conclusion
Our data demonstrate that attempted primary repair with bowel irrigation using a mucolytic agent on all simple cases of gastroschisis with bowel irrigation and early definitive closure of the abdominal defect shortens time to first feeds, total TPN use, time to extubation, and length of stay and is recommended.
References
Jones AM, Isenburg J, Salemi JL, Arnold KE, Mai CT, Aggarwal D et al (2016) Increasing prevalence of gastroschisis—14 States, 1995–2012. Morb Mortal Wkly Rep 65(2):23–26
Eggink BH, Richarson CJ, Malloy MH, Angel CA (2006) Outcome of gastroschisis: a 20-year case review of infants with gastroschisis born in Galveston, Texas. J Pediatr Surg 41(6):1103–1108
Kidd JN, Jackson RJ, Smith SD, Wagner CW (2003) Evolution of staged versus primary closure of gastroschisis. Ann Surg 237(6):758–765
Driver CP, Bruce J, Bianchi A, Doig CM, Dickson AP, Bowen J (2000) The contemporary outcome of gastroschisis. J Pediatr Surg 35(12):1719–1723
Lansdale N, Hill R, Gull-Zamir S, Drewett M, Parkinson E, Davenport M et al (2009) Staged reduction of gastroschisis using preformed silos: practicalities and problems. J Pediatr Surg 44(11):2126–2129
Allotey J, Davenport M, Njere I, Charlesworth P, Greenough A, Ade-Ajayi N et al (2007) Benefit of preformed silos in management of gastroschisis. Pediatr Surg Int 23(11):1065–1069
Owen A, Marven S, Jackson L, Antao B, Roberts J, Walker J et al (2006) Experience of bedside preformed silo staged reduction and closure for gastroschisis. J Pediatr Surg 41(11):1830–1835
Chiu B, Lopoo J, Hoover JD, Almond PS, Arensman R, Madonna MB (2006) Closing arguments for gastroschisis: management with silo reduction. J Perinat Med 34(3):243–245
Schlatter M, Norris K, Uitvlugt N, DeCou J, Connors R (2003) Improved outcomes in the treatment of gastroschisis using a preformed silo and delayed repair approach. J Pediatr Surg 38(3):459–464
Tsai MH, Huang HR, Chy SM, Yang PH, Lien R (2010) Clinical features of newborns with gastroschisis and outcomes of different initial interventions: primary closure versus staged repair. Pediatr Neonatol 51(6):320–325
Owen A, Marven S, Johnson P, Kurinczuk J, Spark P, Draper ES et al (2010) Gastroschisis: a national cohort study to describe contemporary surgical strategies and outcomes. J Pediatr Surg 45(9):1808–1816
Baud D, Lausman A, Algaraj MA, Seaward G, Kingdom J, Windrim R et al (2013) Expectant management compared with elective delivery at 37 weeks for gastroschisis. Obstet Gynecol 121(5):990–998
Cain MA, Salemi JL, Paul Tanner J, Mogos MF, Kirby RS, Whiteman VE et al (2014) Perinatal outcomes and hospital costs in gastroschisis based on gestational age at delivery. Obstet Gynecol 124(3):543–550
Overcash RT, DeUgarte DA, Stephenson ML, Gutkin RM, Norton ME, Parmar S et al (2014) Factors associated with gastroschisis outcomes. Obstet Gynecol 124(3):551–557
Yang EY, Davies LM, Buchanan P, Kling C, Banyard DA, Ramones T (2014) Spontaneous onset of labor, not route of delivery, is associated with prolonged length of stay in babies with gastroschisis. J Pediatr Surg 49(12):1776–1781
Baerg J, Kaban G, Tonita P, Pahwa P, Reid D (2003) Gastroschisis: a sixteen-year review. J Pediatr Surg 38(5):771–774
Moir CR, Ramsey PS, Ogbum PL, Johnson RV, Ramini KD (2004) A prospective trial of elective preterm delivery for fetal gastroschisis. Am J Perinatol 21(5):289–294
Charlesworth P, Njerea I, Allotey J, Dimitrou G, Ade-Ajayi N, Devane S et al (2007) Postnatal outcome in gastroschisis: effect of birth weight and gestational age. J Pediatr Surg 42(5):815–818
Langer JC, Longaker MT, Crombleholme TM, Bond SJ, Finkbeiner WE, Rudolph CA et al (1989) Etiology of intestinal damage in gastroschisis I; effects of amniotic fluid exposure and bowel constriction in a fetal lamb model. J Pediatr Surg 24(10):992–997
Sydorak RM, Nijagal A, Sbragia L, Hilrose S, Tsao K, Phibbs RH et al (2002) Gastroschisis: small hole, big cost. J Pediatr Surg 37(12):1669–1672
Islam S, St. Peter S, Downard CD, Qureshi F, Bruch S, Renaud E et al (2014) Immediate vs. silo closure in gastroschisis: results from a large multicenter study. In: American academy of pediatrics section on surgery national conference, October 2014
Stanger J, Mohajerani N, Skarsgard ED (2014) Practice variation in gastroschisis: factors influencing closure technique. J Pediatr Surg 49(5):720–723
Banyard D, Ramones T, Phillips SE, Leys CM, Rauth T, Yang EY (2009) Method to our madness: an 18-year retrospective analysis on gastroschisis. J Pediatr Surg 45:579–584
Boutros J, Regier M, Skarsgard ED (2009) Is timing everything? The influence of gestational age, birth weight, route, and intent of delivery on outcome in gastroschisis. J Pediatr Surg 44(5):912–917
Carnaghan H, Pereira S, James CP, Charlesworth PB, Ghionzoli M, Mohomed E et al (2014) Is early delivery beneficial in gastroschisis? J Pediatr Surg 49:928–933
Harris J, Poirier J, Sellip D, Pillai S, Shah AN, Jackson C et al (2015) Early closure of gastroschisis after silo placement correlates with earlier enteral feeding. J Neonatal Surg 4(3):28
Shaw A (1969) Safety of N-acetylcysteine in treatment of meconium obstruction of the newborn. J Pediatr Surg 4(1):119–124
Burke R et al (2002) New strategies in nonoperative management of meconium ileus. J Pediatr Surg 37(5):760–764
Gurien LA, Dassinger MS, Burford JM, Saylors ME, Smith SD (2017) Does timing of gastroschisis repair matter? A comparison using the ACS NSQIP pediatric database. J Pediatr Surg 52(11):1741–1754
Kornhauser M, Schneiderman R (2010) How plans can improve outcomes and cut costs for preterm infant care. Manag Care 19:28–30
Youssef F, Gorgy A, Arbash G, Puligandla PS, Baird RJ (2016) Flap versus fascial closure for gastroschisis: a systematic review and meta analysis. J Pediatr Surg 51:718–725
Logghe HL, Mason GC, Thornton JG, Stringer MD (2005) A randomized controlled trial of elective preterm delivery of fetuses with gastroschisis. J Pediatr Surg 40:1726–1731
ClinicalTrials.gov (2016) Gastroschisis outcomes of delivery (GOOD) study. https://clinicaltrials.gov/ct2/show/NCT02774746?term=Gastroschisis+Outcomes+of+Delivery+%28GOOD%29+Study&rank=1. Accessed 16 Aug 2016
Acknowledgements
We thank Jeremey S. McGarvey, statistician, OSF Healthcare Analytics, for assistance with analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interests.
Rights and permissions
About this article
Cite this article
Pearl, R.H., Esparaz, J.R., Nierstedt, R.T. et al. Single center protocol driven care in 150 patients with gastroschisis 1998–2017: collaboration improves results. Pediatr Surg Int 34, 1171–1176 (2018). https://doi.org/10.1007/s00383-018-4349-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-018-4349-7