Abstract
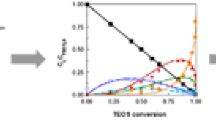
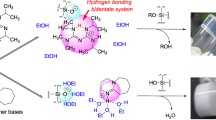
Three silica gel sample systems, modified with 3-aminopropyltriethoxy silane (APTS), were prepared by sequentially sampling the reaction mixture at various time intervals. The concentrations of 3-aminopropylsilyl groups (APS) bound on the silica surface were determined by elemental analysis. For the same sample systems, 29Si NMR intensities of an (-O)4Si species belonging only to the silica gel particles and corrected by a cross-polarization correction factor were also measured. Both the APS-concentrations and the corrected 29Si NMR intensities depended upon reaction time, reflecting the rate of the APTS-silica gel reaction. Kinetic analysis of these data was made by use of the Gauss-Newton method, and the overall reaction was found to consist of three reaction processes (an initial fast reaction, a slower second reaction and a much slower third reaction). In particular, the conversion of (-O)3SiOH to (-O)4Si is predominant in the second reaction process and the pore size of a silica gel particle affects the reaction mechanism.
Similar content being viewed by others
References
Hockey JA (1965)Chem Ind (London) 57–63;
Arkles B (1977) Chemtech 7:766–778
Davydov VY, Kiselev AV, Zhuravlev LT (1960) Trans Faraday Soc 60:2254–2264
Snyder LR, Ward JW (1966) J Phys Chem 70:3941–3952
Hair ML, Hertl W (1969) J Chem Phys 73:2372–2378
Armistead CG, Tyler AJ, Hambleton FH, Mitchell SA, Hockey JA (1969) J Phys Chem 73:3947–3952
Tripp CP, Hair ML (1991) Langmuir 7:923–927;
Tripp CP, Hair ML (1993) J Phys Chem 97:5693–5698
Morrow BA, McFarian AJ (1992) J Phys Chem 96:1395–1400
Van Der Voort P, Gillis-D’Hamers I, Vrancken KC, Vansant EF (1991) J Chem Soc Faraday Trans 87:3899–3905
Vrancken KC, Van Der Voort P, Gillis-D’Hamers I, Vansant EF, Grobet JP (1992) J Chem Soc Faraday Trans 88:3197–3200
Vrancken KC, Coster LD, Van Der Voort P, Grobet JP, Vansant EF (1995) J Colloid Interface Sci 170:71–77
Morrow BA, Cody IA, Lee LSM (1976) J Phys Chem 80:2761–2767
Chiang C-H, Ishida H, Koenig JL (1980) J Colloid Interface Sci 74:396–404
Murthy Shreedhara RS, Leyden DE (1986) Anal Chem 58:1228–1233
Murthy Shreedhara RS, Blitz JP, Leiden DE (1986) Anal Chem 58:3167–3172
Moses PR, Wier LM, Lennos JC, Finklea HO, Lenhard JR, Murray RW (1978) Anal Chem 50:576–585
DiBencdetfo AT, Scola DA (1978) J Colloid Interface Sci 64:480–500
Cain JF, Sacher E (1978) J Colloid Interface Sci 67:538–540
Maciel GE, Sindorf DW (1980) J Am Chem Soc 102:7606–7607
Sindorf DW, Maciel GE (1983) J Am Chem Soc 105:1487–1493
Caravajal SG, Leyden DE, Quinting GR, Maciel GE (1988) Anal Chem 60:1776–1791
Furusawa K (1996) Colloid Science IV; Tokyo Kagaku Dojin Inc. Tokyo, Chapter 10,266–267
Kaneko K (1996) Colloid Science IV; Tokyo Kagaku Dojin Inc. Tokyo, Chapter 11,313
Shimizu I, Yoshino A, Okabayashi H, Taga K, Nishio E, O’Connor CJ (1996) J Chem Soc Faraday Trans, submitted
Kuester JL (1973) Optimization Techniques with Fortran. McGraw-Hill, New York, Ch 6, pp203–271
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yoshino, A., Okabayashi, H., Shimizu, I. et al. Kinetics of interaction of 3-aminopropyltriethoxysilane with silica gel using elemental analysis and 29Si NMR spectra. Colloid Polym Sci 275, 672–680 (1997). https://doi.org/10.1007/s003960050134
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s003960050134