Abstract
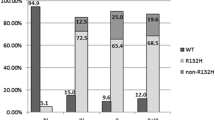
A recent study reported on mutations in the active site of the isocitrate dehydrogenase (IDH1) gene in 12% of glioblastomas. All mutations detected resulted in an amino acid exchange in position 132. We analyzed the genomic region spanning wild type R132 of IDH1 by direct sequencing in 685 brain tumors including 41 pilocytic astrocytomas, 12 subependymal giant cell astrocytomas, 7 pleomorphic xanthoastrocytomas, 93 diffuse astrocytomas, 120 adult glioblastomas, 14 pediatric glioblastomas, 105 oligodendrogliomas, 83 oligoastrocytomas, 31 ependymomas, 58 medulloblastomas, 9 supratentorial primitive neuroectodermal tumors, 17 schwannomas, 72 meningiomas and 23 pituitary adenomas. A total of 221 somatic IDH1 mutations were detected and the highest frequencies occurred in diffuse astrocytomas (68%), oligodendrogliomas (69%), oligoastrocytomas (78%) and secondary glioblastomas (88%). Primary glioblastomas and other entities were characterized by a low frequency or absence of mutations in amino acid position 132 of IDH1. The very high frequency of IDH1 mutations in WHO grade II astrocytic and oligodendroglial gliomas suggests a role in early tumor development.

Similar content being viewed by others
References
Geisbrecht BV, Gould SJ (1999) The human PICD gene encodes a cytoplasmic and peroxisomal NADP(+)-dependent isocitrate dehydrogenase. J Biol Chem 274:30527–30533
Ichimura K, Bolin MB, Goike HM et al (2000) Deregulation of the p14ARF/MDM2/p53 pathway is a prerequisite for human astrocytic gliomas with G1-S transition control gene abnormalities. Cancer Res 60:417–424
Jennings GT, Minard KI, McAlister-Henn L (1997) Expression and mutagenesis of mammalian cytosolic NADP+-specific isocitrate dehydrogenase. Biochemistry 36:13743–13747
Kim SY, Lee SM, Tak JK et al (2007) Regulation of singlet oxygen-induced apoptosis by cytosolic NADP+-dependent isocitrate dehydrogenase. Mol Cell Biochem 302:27–34
Lee SM, Koh HJ, Park DC et al (2002) Cytosolic NADP(+)-dependent isocitrate dehydrogenase status modulates oxidative damage to cells. Free Radic Biol Med 32:1185–1196
Meyer-Puttlitz B, Hayashi Y, Waha A et al (1997) Molecular genetic analysis of giant cell glioblastomas. Am J Pathol 151:853–857
Moreno CS, Evans CO, Zhan X et al (2005) Novel molecular signaling and classification of human clinically nonfunctional pituitary adenomas identified by gene expression profiling and proteomic analyses. Cancer Res 65:10214–10222
Mueller W, Hartmann C, Hoffmann A et al (2002) Genetic signature of oligoastrocytomas correlates with tumor location and denotes distinct molecular subsets. Am J Pathol 161:313–319
Parsons DW, Jones S, Zhang X et al (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321:1807–1812
Peraud A, Watanabe K, Plate KH et al (1997) p53 mutations versus EGF receptor expression in giant cell glioblastomas. J Neuropath Exp Neurol 56:1236–1241
Raff MC, Miller RH, Noble M (1983) A glial progenitor cell that develops in vitro into an astrocyte or an oligodendrocyte depending on culture medium. Nature 303:390–396
Scherer HJ (1940) Cerebral astrocytomas and their derivatives. Am J Cancer 40:159–198
Schmidt M, Antweiler S, Urban N et al (2002) Impact of genotype and morphology on the prognosis of glioblastoma. J Neuropathol Exp Neurol 61:321–328
Soundar S, Danek BL, Colman RF (2000) Identification by mutagenesis of arginines in the substrate binding site of the porcine NADP-dependent isocitrate dehydrogenase. J Biol Chem 275:5606–5612
von Deimling A, Fimmers R, Schmidt MC et al (2000) Comprehensive allelotype and genetic analysis of 466 human nervous system tumors. J Neuropathol Exp Neurol 59:544–558
von Deimling A, von Ammon K, Schoenfeld D et al (1993) Subsets of glioblastoma multiforme defined by molecular genetic analysis. Brain Pathol 3:19–26
Watanabe K, Tachibana O, Sato K et al (1996) Overexpression of the EGF receptor and p53 mutations are mutually exclusive in the evolution of primary and secondary glioblastomas. Brain Pathol 6:217–223
Acknowledgments
This work was supported by the Bundesministerium für Bildung und Forschung (BMBF). We wish to thank K. Lindenberg, U. Ernst and F. Mössler for skillful assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balss, J., Meyer, J., Mueller, W. et al. Analysis of the IDH1 codon 132 mutation in brain tumors. Acta Neuropathol 116, 597–602 (2008). https://doi.org/10.1007/s00401-008-0455-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-008-0455-2